Abstract
Mitochondria are central to energy production and are crucial for the proper operation of the reproductive system. Mitochondrial function and capacity determine whether germ cells are fertilizable, and embryos can safely reach developmental expectations. Impaired mitochondrial biogenesis and defects in mitochondrial DNA result in low birth rates, infertility, and, more seriously, unhealthy offspring with inherited and irreversible metabolic diseases. In recent years, mitochondrial transfer and transplantation have contributed greatly to the field of assisted reproductive technology, especially with advances in biotechnology. Much effort has been invested in refining mitotherapy techniques, aiming at improving the safety, efficacy, and accessibility of the treatment, while reducing costs, labor, and ethical issues. Recent research has shown numerous changes in the approaches with innovative ideas and new materials. This review highlights the role of mitochondria in the reproductive system and the current efforts to improve the outcomes in ART cases with mitochondrial issues. We also summarize different types of mitochondrial transplantation techniques and emphasize the importance of mitochondria selection for reproductive purposes.
Introduction
A diagnosis of infertility is made if a couple cannot achieve or maintain a pregnancy after one year of attempting to conceive naturally. The World Health Organisation reported in 2023 that infertility affects about 17.5% of couples worldwide, threatening the stability of global population growth and the economy. Moreover, the age of first childbearing is reported to increase in various countries worldwide. According to the UNECE (United Nations Economic Commission for Europe) data, the mean age of women at the birth of their first child has increased by one year in the 10-year period from 2012 to 2022. In recent years, women have tended to delay their first childbearing plan, while their fertility age is limited, making the issue even more challenging1, 2. Infertility treatments can differ between men and women, which may include medications and surgery. However, with unexplained infertility, these treatments appear to be less effective. In that context, assisted reproductive technology (ART) is meeting increasing demand, contributing greatly to the world population, making up around 8% of the total population in some regions3, 4. The rates of success in ART are controversial and are correlated with the quality of the medical systems in different regions. Furthermore, the success rates also rely on different clinics, which may not always wish to share their data. Generally, depending on the patient's age, the indications could vary. For example, the use of intrauterine insemination (IUI) is suggested to be as effective as in vitro fertilization (IVF) in patients under 40, while IVF may be more effective for older patients5, 6, 7. In fact, both treatments may require several cycles, and even with failed attempts to conceive in the first three cycles in IVF, it was suggested that the success rate may increase, reaching near-natural rates with more repeated cycles8, 9, 10. However, it must be noted that the treatment of infertility can be costly and prolonged, putting an extra burden on the existing stress of the patients. Infertile individuals undergoing therapeutic support or ART often face mental health issues such as emotional adjustment, anxiety, and depression. Although these issues may not be directly related to the outcomes, they can sometimes prolong the time it takes to achieve success11, 12, 13, 14, 15, 16. Even though interventions that provide psychological support were shown to reduce depression in these women, they could not alleviate their anxiety levels and were not correlated with success rates17. Thus, the key to overcoming these challenges greatly relies on the innovation of new technology, as advancements in therapeutic development would provide better options and solutions for current biological and technical difficulties and narrow the gaps in approaching the treatments.
Infertility has various causes, such as genetic, hormonal, anatomical, immunological, infectious, environmental, and lifestyle factors18, 19. Among these factors, mitochondrial dysfunction or abnormality has been proposed as a potential factor contributing to infertility in both males and females20, 21. Mitochondria are responsible for producing and maintaining energy (or ATP) that is required for cellular activities via several metabolic pathways such as oxidative phosphorylation (OXPHOS), amino acid metabolism, beta-oxidation, and calcium homeostasis. Mitochondria contain their own DNA in a ring shape, which is generally referred to as mitochondrial DNA (mtDNA). Even though mtDNA is directly in contact with reactive oxygen species (ROS) produced by nearby metabolic processes, they are less efficiently repaired and thus, are more vulnerable to mutation. Mitochondria are essential for various aspects of reproductive function and processes, such as gamete maturation, fertilization, implantation, placental function, and embryonic development21. Mitochondrial dysfunction can result from mutations or deletions occurring in mtDNA or in nuclear DNA sequences that encode or regulate mitochondrial proteins, as a consequence of oxidative stress, inflammation, toxins, and aging. Mitochondrial dysfunction can affect different cell types involved in reproduction, including germ cells (sperm and oocytes), and embryonic cells, which can result in infertility. Given the importance of the mitochondria in the reproductive system, understanding the role of mitochondrial damage in the pathogenic progression of infertility is necessary for developing effective treatments and interventions. Various types of treatments have been clinically applied and developed, and the growth of different scientific fields has greatly improved the effectiveness of assisted reproductive technology (ART). One way to approach the issues with mitochondrial dysfunction in infertility is mitochondrial therapy (or mitotherapy), in which healthy mitochondria are transferred or transplanted into defective cells, which would be oocytes or zygotes22. The data from preclinical and clinical applications have shown that mitotherapy is a promising and potential method for treating infertility, raising hopes and opportunities for infertile patients; however, it is met with numerous concerns both technically and ethically23, 24, 25. In this review, we cover the biological causes and consequences of mitochondrial damage in various cells and tissues of the reproductive system and their impacts on fertility. We also discuss the current methods to improve the outcomes for mitochondria-related issues and the development of mitochondrial transplantation in ART. We also give examples of how the techniques have been refined over time and highlight the critical importance of mitochondrial selection, specifically for improving poor-quality oocytes and low fertilization rates.
MITOCHONDRIA IN THE REPRODUCTIVE SYSTEM
In reproductive cells, mitochondria serve a vital function in providing the energy necessary for various processes such as oocyte maturation, sperm motility, and fertilization26. Mitochondria in reproductive cells have unique characteristics, including differences in morphology, size, and distribution compared to those found in other cell types27. Additionally, mitochondrial DNA (mtDNA) is inherited maternally, with the egg providing the majority of the mtDNA in the developing embryo, which is a unique feature of mitochondria in reproductive cells28.
In female reproductive cells, mitochondria are especially critical for the growth of oocytes, which are the precursor cells of mature eggs29. Oocyte maturation features an increase in mitochondrial activity to meet the energy demands for developing cells30. Once the oocyte is fertilized, mitochondria maintain their critical role in supporting embryonic development in the early stages31. Oocyte mitochondria influence oocyte quality, particularly in terms of proper chromosomal segregation during oocyte maturation32. Indeed, mitochondrial malfunction can imperil fetal viability, particularly in older women, and mitochondrial defects can be transferred to the fetus regardless of the mother's age33, 34. Apart from generating energy for basic cellular demands, mitochondria also act as regulators of calcium homeostasis, which is critical for cell survival and function35. This balanced state is achieved through calcium storage and release interdependently, ensuring suitable free intracellular calcium levels throughout various stages of cell growth36. Moreover, mitochondria are also involved in controlling the epigenetic alterations in oocytes and embryos by regulating biochemical activities, including histone acetylation, and histone and DNA methylation-demethylation21. The involvement of mitochondria in oocyte function is summarized in Figure 1.
In male reproductive cells, mitochondria are mainly located in the midpiece (also called the neck) of the sperm, where they supply energy for the flagellum to propel the sperm forward37. This is critical for successful fertilization because sperm must be able to reach the egg by swimming through the female reproductive tract21. Furthermore, the quantity of active mitochondria in spermatogonia stem cells (SSCs) can vary depending on the development stage38. SSCs are a type of cell found in the testes responsible for the production of sperm39. The specific changes in the quantity of active mitochondria in SSCs during development stages can have implications for the energy requirements and metabolic activities of these cells. It could potentially affect their ability to divide, differentiate, and ultimately contribute to the production of sperm40. Mitochondria in spermatogonia are typically small and spherical, located in the basal structure of the seminiferous epithelium, and have access to the vasculature and interstitial fluid41, which supports low oxidative phosphorylation (OXPHOS) activity at this stage. When mitochondria cross the blood-testis barrier and enter the adluminal compartment, they undergo a process known as intermitochondrial cement (IMC), where they become elongated and cluster around the nuage42. Mitochondrial fragmentation occurs in post-meiotic spermatids42. Finally, as spermiogenesis progresses, mitochondria pack closely around the sperm midpiece43. Only a fraction of mitochondria line the sperm midpiece during elongation; the rest, along with other cellular components, are collected into residual bodies just before spermiation (sperm release) for phagocytic destruction by Sertoli cells44. Furthermore, the presence of exogenous mitochondria in male germ cells emphasizes the relevance of mitochondria in testicular metabolism45. Germ cell survival in the adult testis relies entirely on energy from carbohydrate metabolism, which is produced by both glycolytic and OXPHOS processes46. This is particularly relevant in germ cells undergoing complex and energy-demanding processes such as meiosis and spermatogenesis. Mitochondrial function in sperm biology is summarized in Figure 2.
Mitochondrial dysfunction and the effects on the male reproductive system
Mitochondrial dysfunction involves the impairment of bioenergetic processes inside the mitochondria compartment, resulting in a reduction in energy production and dysregulation of calcium, electron-proton, and substrates balance in the cell, leading to excessive formation of ROS and high rates of mtDNA mutation47, 48. Mitochondrial dysfunction can lead to infertility in several ways. Studies have shown that the capacity of sperm and oocytes in assisted reproductive technologies is determined by mitochondrial function49, 50. Mutations in genes critical for mitochondrial function, particularly those involved in maintaining mtDNA or mitochondrial protein translation, have been increasingly recognized as a contributing factor to infertility20. Variants in any part of these genes can lead to mitochondrial disorders, which can cause infertility. Mitochondrial dysfunction can affect various cell types within the reproductive system, including oocytes, sperm, and somatic cells such as cumulus cells (CCs) and granulosa cells in the ovary51, which can also contribute to infertility.
Debilitated mtDNA integrity and sperm motility
Mitochondrial dysfunction impacts the integrity of sperm DNA. Alterations affecting the mitochondrial genome can impair male reproductive potential52. Large deletions or single-nucleotide polymorphisms are two examples of mutations that influence sperm mtDNA53, 54. As a result, sperm with mutant mtDNA may have respiratory issues, which impact how energy is produced, as well as motility issues that affect how active they normally are55. Studies suggest a strong link between the alterations or deletions in mtDNA and male infertility56. Deletions in mtDNA that affect cellular equilibrium and energy production have been shown to impair sperm motility56. Moreover, mtDNA copy number and sperm DNA fragmentation (SDF) are both correlated with semen quality52. The study found that asthenozoospermic semen samples contained a higher mtDNA copy number, which was associated with reduced sperm concentration, low sperm number, and decreased motile spermatozoa. Thus, adequate mitochondrial genome content is required for efficient energy metabolism and hence, facilitates sperm motility. In addition, SDF was found to increase in asthenozoospermic samples, which resulted in abnormal forms, even though SDF and mtDNA copy number were not correlated57. The decrease in mtDNA copy number naturally occurring during the process of sperm development could imply alterations in the maturation process of sperm41. This increase in mitochondrial genome content could also be a result of a compensatory response triggered to counteract the mitochondrial dysfunction, which is highly probable to impact sperm quality of infertile males40. Overall, these findings underline the importance of mitochondrial integrity and mtDNA content in maintaining efficient energy metabolism, which ensures proper sperm function, motility, and ultimately, male reproductive potential.
The mitochondrial genome quantity is controlled by various molecules and factors to regulate the genetic material cloning process. The mitochondrial transcription factor A (TFAM) protein is involved in the creation of primers needed for mitochondrial DNA copy number, was found actively in lower total motile sperm and this high TFAM activity also coincided with a rise in abnormally shaped sperm, SDF, and mtDNA replication58. Another study found that sperm cells not only lack intact mtDNA, but they also lack TFAM protein, suggesting that TFAM gene expression is positively correlated with sperm motility59. Therefore, it is impossible to rule out the idea that a post-transcriptional regulatory mechanism underlies the distinct expression of the transcript and protein60. It must be noted that reduced sperm function and male infertility can also be a result of molecular modifications to the mtDNA, which affect sperm movement and shape51. For instance, asthenozoospermia has been linked to large mtDNA deletions, with sizes ranging from 4,977 to 7,599 base pairs61, 62. The typical 4977-bp mtDNA loss has been suggested as an efficient indicator for mtDNA damage because it increases in many organs with aging63 . The 4977-bp deletion in sperm mtDNA occurs more frequently in patients with asthenospermia and oligospermia than in healthy individuals64. Additionally, patients with primary infertility were more likely to experience the 4977-bp mtDNA loss in sperm than were those with secondary infertility65. Seven genes and five transfer RNAs are removed as a result of this loss, which affects the region of the mtDNA between 8483 and 13459 base pairs66. In infertile males with severe or ongoing and unexplained asthenozoospermia, abnormalities might be observed in the structure of the mitochondria within the middle section of their sperm67, 68. Asthenozoospermia may have an underlying etiology related to disruptions in energy synthesis and mitochondrial activity in sperm69. The quality of mtDNA and mitochondrial function are critically important in the male reproduction system, which ensures healthy maturation of sperm and their movement during fertilization. Thus, maintaining mitochondrial function and integrity is essential in male fertility.
Sperm apoptosis
Apoptosis is a cell death program, in which cells with compromised genetic materials are eliminated70. Apoptotic activation may occur in the absence of specific cell surface receptors as substances can enter the cell directly and modify the apoptotic cascade71. Heat shock, stressors, ROS, UV radiation, drugs, synthetic peptides, and poisons are a few examples of such variables72. Nowadays, it is acknowledged that human sperm exhibits and activates apoptotic signals in response to different types of stimuli73. A class of proteases known as caspases is crucial for controlling apoptosis. The mitochondria are crucial in the apoptotic cascade by providing key elements, such as those that activate caspase activity and DNA fragmentation74. Cytochrome c, a significant apoptosis component, facilitates caspase 9 and caspase 3 initiation, which results in cell apoptosis75. Disruptions in cellular homeostasis are known to induce the permeability transition pore (PTP), which is located in the outer membrane of mitochondria to open, which is involved in cell death signaling76. Mitochondrial ATP synthase dimers are in charge of PTP production77. The removal of cytochrome c via PTP activates the caspase cascade and apoptotic program76. Caspases 3 and 9 activity was detected in the midpiece of human sperm after ejaculation78, 79. When the apoptotic program was triggered in spermatozoa, caspase 9 and 3 activity increased, while mitochondrial membrane potential (MMP) decreased, which was associated with low sperm motility79, 80. Studies have established a detrimental link between caspase activation, sperm quality, and the absence of integrity in the plasma membrane81. This condition is evidenced by the presence of externalized phosphatidylserine, which is a marker of programmed cell death82. Understanding the complexities of sperm apoptosis is paramount for developing novel therapeutic interventions and improving assisted reproductive techniques. Furthermore, identifying the key regulators of sperm apoptosis may make way for innovative diagnostic tools and targeted therapies to address male infertility.
The phosphatidylinositol 3-kinase (PI3K)/AKT pathway is critical in regulating sperm apoptosis. This intracellular signaling system promotes a variety of cellular functions, which include cell survival, cell growth, proliferation, and migration83. AKT (the main protein in the PI3K pathway) activation improves sperm survival, especially under stress conditions84. Spermatozoa viability and function are dependent on the PI3K enzyme phosphorylating AKT1 (also known as RAC-alpha serine/threonine-protein kinase), which inhibits downstream effectors of the apoptotic pathway including the Bcl-2-associated death promoter85. Conversely, spermatozoa are driven toward the apoptosis process as a result of the inactivation of AKT1, when PI3K activity is suppressed86. This activates the caspase cascade in the cytosol, increases ROS production in the mitochondria, resulting in a considerable reduction of sperm motility and oxidative DNA damage73. The active endonucleases, however, are unable to cleave the nuclear DNA because the nucleus in the sperm head is well separated from the sperm midpiece, which contains the mitochondria and cytoplasm87. As a result, although DNA can be affected by oxidative reactions, apoptosis may not lead to DNA fragmentation in human spermatozoa73. The PI3K/AKT pathway expresses a critical role in controlling sperm function and quality, as evidenced by the complex interactions it has with the apoptotic apparatus.
Sperm apoptosis also comes from the dysregulation of mitochondrial dynamics, promoting oxidative stress and cell death. Mitochondrial fission and fusion are essential for maintaining mitochondrial function; excessive fission can lead to apoptosis by promoting mitochondrial outer membrane permeabilization (MOMP)88. Normal expression in fusion and fission genes allows spermatozoa to function successfully89. However, MOMP is regulated by the protein Bcl-2, which is an important factor influencing the release of apoptotic factors like cytochrome c, which activates caspases and initiates cell death90. Another study has shown that Hexavalent chromium-treated rat testis presents a decline in Sirtuin 1 (Sirt1), resulting in an increase in mitochondrial fission91. Moreover, Sirt1 then downregulates nuclear factor-erythroid-2-related factor 2 (Nrf2). These stages greatly activate OS and the expression of apoptotic genes such as Bcl-2, cytochrome c, and promote sperm apoptosis91. The overproduction of ROS could damage organelle structure and exacerbate downstream factors, leading to cellular apoptotic signs, meanwhile, those apoptotic features induced by exogenous factors could induce OS and disrupt mitochondrial dynamic homeostasis88, 92.
Oxidative stress
Sperm cells rely mainly on mitochondria to produce ATP in demand for energy to support their motility, capacitation, acrosome reaction, and fertilization ability through OXPHOS93. Mitochondrial dysfunction in sperm cells can cause male infertility by impairing sperm quality and function94. However, sperm mitochondria are also the main site of ROS production, which can damage cellular components if not scavenged by antioxidants95. Furthermore, ROS induces oxidative stress, negatively affecting the sperm membrane, DNA, proteins, and lipids96, 97.
Mitochondrial metabolism provides the energy required for sperm function98. Sperm movement requires an abundance of energy generated from OXPHOS. This process includes oxidative reactions and the generation of ROS99. According to Munro and Treberg (2017), ROS, namely superoxide radicals and hydrogen peroxide, are produced as a byproduct of the aerobic synthesis of ATP via OXPHOS100. Hydrogen peroxide (H2O2) generation in the mitochondrial matrix was increased through processes unrelated to MMP as a result of rotenone-induced suppression of Complex I101. Because of this, the sperm midpiece's lipids started to oxidize, which caused sperm motility to decline102. Nevertheless, using antioxidants such as tocopherol was found to reverse the harmful effects of rotenone103. In oligoasthenozoospermic patients, the co-incubation of spermatozoa with myoinositol improved sperm mobility and increased the number of spermatozoa with high MMP104. Lower MMP is also correlated with poor sperm mobility in infertile men105. When exposed to spermicidal agents, human sperm showed a significant reduction in motility and MMP106. Uncoupling the electron transport chain can lead to abundant ROS release, which is associated with reduced MMP and sperm mobility107. Finally, low sperm MMP can be an indicator of poor sperm quality, which results in lower fertilization rates in in vitro fertilization (IVF)108.
Sperm motility can significantly decrease due to the lack of energetic donation. There are two sources of energy providing ATP for sperm, including OXPHOS and glycolysis, primarily contributing ATP to sperm flagella for movement109. Meanwhile, the energy produced by mitochondrial OXPHOS is used for gluconeogenesis, which in turn produces raw sugar for glycolysis110. Studies have shown that disruption of glycolysis reduces sperm motility; in contrast, inhibition of oxidative phosphorylation does not significantly impair human sperm motility111. Additionally, exogenous pyruvate enhances glycolytic ATP production, motility, hyperactivation, and capacitation in human sperm, indicating a reliance on glycolysis112. Therefore, abnormalities in mitochondrial energy production do not directly affect sperm motility113. Although mitochondria are not the primary ATP donors for human spermatozoa, studies have suggested that mitochondrial activities are a significant source of ROS114. ROS exists in spermatozoa at a certain low controlled concentration, participating in several physiological processes of sperm. However, when ROS production becomes abnormal, the level of intrinsic ROS increases, thereby affecting sperm health115, 116. ROS in semen includes oxygen-centered radicals (e.g., superoxide anion (O2●−), hydroxyl radical(●OH)) and non-radical derivatives like hydrogen peroxide (H2O2)117. H2O2 and O2●− are two significant oxidants with key physiological roles118. They are continuously generated by mitochondrial NADH-dependent processes (located in the inner mitochondrial membrane) and extramitochondrial NADPH-dependent systems (located in the plasma membrane)119. O2●− can be converted into H2O2 through superoxide dismutase (SOD); furthermore, it can react with H2O2 to form highly reactive radicals (●OH), triggered by the presence of iron (Fe2+)120, 121. The overall reaction is simply described below:
O2●− + H2O2 → ●OH + OH⁻ (hydroxide ion) + O2
When the process of mitochondrial ROS generation is disrupted, leading to an increase in O2●− level122, 123. Such conditions can result in abnormal concentrations of the most highly reactive molecule, hydroxyl radicals124. ●OH can cause damage to cellular components, including lipids, proteins, and DNA, due to their unpaired electron125. Removing a hydrogen atom from membrane fatty acids leads to the oxidation of the spermatozoa membrane, called membrane lipid peroxidation115. This process produces an unstable lipid peroxyl radical, then initiate a series of reactions and generate harmful compounds, disrupting the membrane flexibility and fusion ability119. These properties are crucial for sperm movement, acrosome release, and successful fertilization119. Therefore, while mitochondria may not be the primary energy source for sperm motility, their role in ROS generation can significantly impact sperm quality and fertility potential.
Oxidative stress can also induce DNA fragmentation, chromosomal abnormalities126, and sperm epigenetic modifications, which can affect gene expression127. Moreover, sperm cells can transmit mtDNA mutations or deletions to the offspring through paternal inheritance128. These mutations or deletions can impair OXPHOS function and cause mitochondrial diseases that affect various organs and systems129. Oxidative imbalance can cause male infertility by the following mechanisms: (i) damaging sperm membrane, thereby reducing sperm motility and ability to fertilize; (ii) damaging sperm DNA leading to reduced fertilization ability and affecting embryonic development after fertilization; and (iii) increasing the process of sperm degradation130, 131. The increase of polyunsaturated fatty acid concentration in the sperm plasma membrane exposes them to oxidative stress, which results in lipid peroxidation and sperm membrane damage132. Oxidative stress-induced caspase activation can also cause cytochrome c to be released from the mitochondria, leading to the apoptosis of spermatozoa133. Various intrinsic and extrinsic factors can contribute to the development of SDF, including varicocele, infection, aging, heat stress, lifestyle, environmental toxins, and ionizing and non-ionizing radiation21. Antioxidants have been found to effectively reduce both ROS and SDF in infertile men with various conditions134. In conclusion, oxidative stress can compromise sperm health by damaging sperm components such as DNA and membrane, potentially leading to fertilization failure. Additional investigation is essential to elucidate the underlying pathways involved and to explore potential therapeutic strategies targeting OS in male infertility.
Mitochondria functions in oocyte development and aging
Approximately, sperm cells contain 70-80 mitochondria40, whereas a normal oocyte has around 100,000 mitochondria135. In female reproductive cells, mitochondria are especially critical for the growth of oocytes, which are the precursor cells of mature eggs29. Oocyte maturation features an increase in mitochondrial activity to supply energy demands for developing cells30. Once the oocyte is fertilized, mitochondria maintain their critical role in supporting embryonic development at the early stages31. Oocyte mitochondria influence oocyte quality, particularly in terms of proper chromosomal segregation during oocyte maturation32. Apart from generating energy for basic cellular demand, mitochondria also act as regulators of calcium homeostasis, which is critical for cell survival and balancing cell function35. This balanced state is achieved through calcium storage and release interdependently, ensuring suitable free intracellular calcium levels throughout numerous stages of cell growth36. Moreover, mitochondria are also involved in controlling the epigenetic alterations in oocytes and embryos by regulating biochemical activities including histone acetylation, histone and DNA methylation-demethylation21. Indeed, mitochondrial malfunction can imperil fetal viability, particularly in older women, and mitochondrial defects can be transferred to the fetus regardless of the mother's age33, 34. The involvement of mitochondria in oocyte function is summarized in Figure 3.
Oocyte aging and mitochondria DNA mutation
The accumulation of somatic mtDNA mutations, compromised quality control processes, and disrupted mitochondrial biogenesis are among the aspects of mitochondrial dysfunction that are related to aging136, 137. Ovarian aging manifests as a decline in ovarian reserve, a concept encompassing both the quality and oocyte number available for ovulation138. The reduction in primary follicle number indicates an ovarian quantitative decline whereas a qualitative decline is associated with decreased mtDNA content27, 139. Women's age is negatively correlated with mtDNA replication139. This can also be observed in women of similar age groups with diminished ovarian reserve140. Oocytes carrying mutations within their mtDNA can compromise mitochondrial function and potentially lead to reduced oocyte quality. Thus, mtDNA content is crucial to female fertilization. Not only mutations in mtDNA damage mitochondria, but mutations in genetic materials of the nucleus can also cause loss of mitochondrial function. Some mitochondrial proteins are translated from the nuclear genome when expressed abnormally. Allelic mutations of the nuclear-encoded catalytic subunit of the enzyme mtDNA polymerase gamma (PolgA), which encodes its catalytic components, have been linked to a variety of mitochondrial diseases affecting mtDNA stability141. POLG variants can lead to a variety of illnesses such as autosomal dominant and recessive progressive external ophthalmoplegia (PEO), which are defined by mtDNA deletions or depletion142. On the other hand, POLG recessive and dominant mutations are also linked to other conditions such as parkinsonism, and premature menopause143. Many women listed as having this illness had shown hypergonadotropic hypogonadism144. This condition was found to be related to a heterozygous p.Y955C mutation in three families with autosomal dominant inheritance20. Additionally, a family with a heterozygous Y955C mutation also showed cases of testicular atrophy145. A heterozygous POLG variation A was also reported in cases with ovarian dysgenesis, which could be associated with cataracts, muscle atrophy, and 3-methylglutaconic aciduria in females145. More studies on POLG should be undertaken to broaden the knowledge of the interaction between nuclear DNA and mtDNA, and prerequisite research could be conducted to develop disease treatment measures through genetic agents. It must be noted that in female mice with POLG mutator, fertility is reduced by mtDNA mutations that disrupt oocyte NADH/NAD+ redox state146, 147, which is an important factor to maintain mtDNA integrity.
Stress response pathways
Like sperm, oocyte mitochondria are susceptible to damage by reactive oxygen species (ROS). Maintained equilibrium of ROS level is necessary for normal physiology of the oocyte. This balance is disrupted by endogenous and exogenous factors such as apoptosis induction, cryoinjury, or exposure to disadvantageous factors, leading to mitochondrial dysfunction and impaired oocyte development148, 149. Oxidative stress affects oocyte development and survival, reducing oocyte quality and ability to fertilize. Particularly, oxidative stress can cause telomere shortening in oocytes, which consequently causes cell cycle arrest, DNA instability and abnormal spindle formation150, 151, 152. Oxidative stress is also correlated with ER stress, disrupting the import and exchange of calcium ion (Ca2+), which is important for calcium homeostasis in oocytes, supporting oocytes over maturation stages, especially during meiosis153, 154, 155, 156. Thus, preventing mitochondrial oxidative stress is necessary in early oocytes. In this context, early oocytes exquisitely retain low oxidative stress by repressing the expression of ETC complexes, especially complex I149. It was found that subunit assembling in Complex I was also significantly reduced, leading to the formation of inactive Complex I149. The suppression of Complex I was effective in maintaining low ROS production and redox state in early oocytes until oogenesis occurred and Complex I was activated149. These data indicate that low ROS and reduced redox status are important for oocyte function, as a high oxidative profile contributes to oocyte declining activity at age. Redox imbalance is also affected by aging in oocytes, in which the production of ROS increases while antioxidant levels decrease, disturbing the meiotic process and increasing aneuploidy in mature eggs157, 158, 159. It was shown in porcine oocytes that to overcome the damage from ROS, the cells activate Sirtuin 3 (SIRT3) protein expression, which in turn promotes the deacetylation of superoxide dismutase 2 (SOD2)160, 161. SOD2 is a strong antioxidant, responsible for eliminating ROS accumulation in the mitochondria, indicating that the SIRT3/SOD3 pathway is a mechanism for oocytes to retain low ROS levels160, 161. Therefore, strategies to maintain redox balance are crucial in oocyte function.
Additionally, maintaining a healthy proteome, especially those of long-lived proteins, is an important factor in ensuring the quality and development of oocytes during reproductive life162. Many of these long-lived proteins in women's oocytes are mitochondrial162. It has been shown that oocyte mitochondrial unfolded protein response (mtUPR) stands as a mechanism of cellular defense that helps resolve proteostatic stress163. mtUPR is activated to restore proteostasis and rescue mitochondrial function, whenever an increase in misfolded or unfolded proteins disrupts mitochondrial homeostasis163. This process includes a cascade of nuclear gene transcription to upregulate the expression of genes encoding specialized mitochondrial chaperones164. These molecular chaperones, such as heat shock protein (HSP) family, help misfolded proteins achieve their proper three-dimensional structure, crucial for protein function165. Furthermore, mtUPR also implements a negative feedback mechanism to limit the influx of newly synthesized mitochondrial proteins166. This serves to alleviate the burden on the already stressed protein folding machinery within the mitochondria167. Maintaining a healthy mitochondrial proteome necessitates the coordinated action of chaperones and mitochondrial proteases. Misfolded proteins beyond the capacity of the chaperone system are targeted for degradation by specialized mitochondrial proteases168. mtUPR ensures the proper function of mitochondria169. Any disruption to this quality control system can result in mitochondrial dysfunction169.
The primary regulator of mtUPR, mitochondrial matrix caseinolytic peptidase (Clpp), was discovered to be increased during mitochondrial stress produced by unfolded proteins170. This rise in Clpp protease activity promotes the degradation of misfolded proteins, generating peptide fragments that act as signaling molecules, which activate transcription factor ATFS1. ATFS1 then translocates from the mitochondrial matrix to the nucleus and interacts with two proteins: UBL5 (ubiquitin-like 5) and DVE1 (defective proventriculus in Drosophila homolog 1). This tripartite complex binds to specific nuclear DNA sequences, which regulate gene expression of essential mitochondrial chaperones, such as HSP60 and HSP1027. Some mitochondrial abnormal states such as increased mtDNA replication and ROS level, as well as disrupted MMP, are also determined in animals with Clpp deficiency, resulting in profound female infertility171. Furthermore, metabolic imaging employing fluorescence lifetime imaging (FLI) microscope revealed significant changes in Clpp-deficient oocyte metabolism, including those in the lifetimes of FAD (flavin adenine dinucleotide), NAD (nicotinamide adenine dinucleotide) and the protein-bound percentage of NADH, reflecting disruptions in mitochondrial electron transport chain activity172. Clpp-deficiency in animals was found to induce female infertility with poor oocyte maturation, low two-cell embryonic development, and inability to form blastocysts173. The data were correlated with increased spindle defects in eggs, which led to increased depletion of follicles in mice, which was linked to the initiation of the mammalian target of rapamycin (mTOR) pathway, which could be partially reversed by rapamycin, an inhibitor of mTOR174. In conclusion, it is essential to maintain the quality control mechanism of mitochondrial proteostasis, including mtUPR, to ensure reproductive capability. Elucidating the complicated regulatory networks that govern mitochondrial function has potential implications for understanding infertility and designing treatments.
Mitochondrial dynamics
Mitochondrial ATP production is critical during oocyte and early embryonic development175. These early stages of development exhibit a pronounced dependence on mitochondrial ATP (adenosine triphosphate) production to fuel vital cellular processes176. Beyond ATP generation, mitochondria orchestrate a multitude of cellular functions essential for proper development. These functions encompass the maintenance of mtDNA integrity, mitosis, and apoptosis177. It must be noted that immature oocytes rely on cumulus cells for energy and metabolic supply via gap junctions178. The ATP levels in oocytes fluctuate during the maturation and meiosis processes. ATP levels in oocytes increase during the resumption of meiosis I, then decrease after GV breakdown and reach maximal levels when the first polar body extrusion occurs179. The consumption of ATP also significantly increased during MI and MII179, indicating the need for ATP during oocyte maturation.
Any damaged mitochondria that are beyond repairing capacity or unnecessary will be removed via mitophagy180. To maintain the proper function of both mitochondria and cells, well-coordinated regulations must be in place to balance the formation of the new and the removal of damaged mitochondria181. Additionally, a recent study has shown that mitochondria redistribute during oocyte maturation and development, with mitochondria cluster area increased in the later stages (i.e. MI and MII oocytes)182. The increase in oocyte mitochondria number and clustering has also been shown in oocytes undergoing maturation, which was associated with an increase in ATP production183, most probably to prepare oocytes for energy demand through meiosis as deficiency in ATP production could lead to oocyte spindle disassembling in mice, resulting in apoptosis and arrested growth184. These data suggest that mitochondrial dynamics play an important role in regulating mitochondria biogenesis and energy distribution during oocyte development.
Furthermore, alterations in mitochondrial dynamics can result in mitochondrial dysfunction and cellular malfunction, which is linked to aging conditions and a variety of age-related illnesses185. The dynamin GTPase protein family mediates mitochondrial dynamics to fusion and fission186. Mitochondrial fusion is the process where two or more mitochondria join to form a single interconnected network, while fission is the division of one mitochondrion into separate units186. The balanced interaction between these two opposing processes of fusion and fission is important in maintaining mitochondrial function, inheritance, and quality control within cells187. In mammals, dynamin-related protein (DRP1), which can bind to various mitochondrial receptors, is the main protein involved in regulating the fission process188. Two proteins need to collaborate to run the fusion process, including mitofusin 1 (MFN1), which supports outer mitochondria membrane fusion, and optic atrophy 1 (OPA1), which assists the fusion of inner mitochondria membrane189. Mitochondrial dynamic-related proteins have been examined in germ cells and embryos using knockout mouse models190. DRP1 deletion in oocytes causes a cascade of detrimental effects, including defective mitochondrial dynamics and abnormalities in oocyte function191. During a state of stress in cells, mitochondrial fusion may provide additional cristae to maintain ATP production and hinder autophagy192. Deletion of the fusion protein Mfn1 is reported to cause female infertility in mice due to the developmental arrest of follicles at the secondary stage, leading to oocyte immaturity193. Furthermore, the lack of MFN1 caused ceramide buildup in oocytes and enhanced apoptosis, resulting in rapid follicular depletion138. Similarly, MFN2 deletion in oocytes results in oocyte immaturity, resulting in infertility194. Interestingly, these MFN2-deficient oocytes exhibited shorter telomeres suggesting accelerated cellular aging within the ovary27. Shorter telomeres are associated with a reduced oocyte pool available for ovulation, which contributes to the observed infertility phenotype195. These findings suggest that targeting mitochondrial dynamics could be a promising avenue for future research on female fertility treatments, particularly in cases associated with diminished ovarian reserve and age-related infertility.
Mitochondria and embryo
Mitochondrial DNA is uniparentally inherited in nearly all eukaryotes196. After sperm-oocyte interaction, the head of sperm transmits to the axoplasmic, and sperm mitochondria are trapped outside the oocyte plasma membrane. Research on animal embryos has shown the presence of sperm mitochondria197, which are eliminated by the paternal mitochondria selective autophagy mechanism in the cleavage-stage embryos198. However, it has been recently reported that mtDNA can be inherited from both parents199. Indeed, most of the mitochondria are of oocyte origin in the embryo. As mtDNA is unable to undergo replication before the blastocyst stage, the copy number of mtDNA remains at this level for the rest of the cleavage stage200. Therefore, the oocyte must increase mitochondria numbers from several hundred in primordial germ cells (PGCs) to about 100,000 in the metaphase II oocyte135. The embryo requires sufficient mitochondria count to divide into blastomeres and produce ATP, which is a primary source of energy before embryonic implantation148. During preimplantation, the embryo depends mainly on pyruvate for energy, which is produced via oxidative phosphorylation, while glycolysis is limited. Pyruvates are produced by cumulus cells surrounding oocytes; they are absorbed into the mitochondrial matrix oocyte and are oxidized by the pyruvate dehydrogenase to produce acetyl-CoA201. Cellular respiration produces about 15 ATP from each pyruvate molecule, supplying other organelles. The mechanism maintains a low oxidative phosphorylation pathway, allowing limiting oxidative stress202. ROS increase in poor-quality environmental conditions, that activates protein HIF of Krebs cycle. HIFs are key protein transcription genes that regulate cell adaptation in hypoxic conditions203. Mitochondrial dysfunction is correlated to fertilization failure and embryo development200, 204, 205. Indeed, mitochondrial function significantly impacts the sperm-triggered calcium signaling that activates oocytes and embryo development. Mitochondrial inhibitors such as FCCP or antimycin A uncoupler disrupt cytosolic calcium ions, generating Ca2+ waves206, 207. This event induces mitochondrial dysfunction, which leads to a sustained increase in the level of cytosolic Ca2+, subsequently inducing apoptotic cell death. In addition, both mitosis and meiosis require ATP to promote chromosome cohesion and segregation. Insufficient ATP can block the function of spindle organization, resulting in aneuploidy products. Consequently, this would result in poor-quality embryos and arrested embryo development208. Studies have shown that a different allocation of mitochondria in blastomeres can lead to abnormal cell division209. Mitochondrial dysfunction can be caused by insufficient mtDNA count and damaged mtDNA200. The mtDNA structure lacks histones and is not packed into chromosomes, which makes them more susceptible to damage than nuclear DNA. Without efficient mtDNA repair mechanisms, its mutation rate is approximately 20 times greater than nuclear genomes in vertebrates210. On the other hand, it was shown that cleavage-stage embryonic mtDNA mutation did not affect embryo quality211. Similarly to gametes, perhaps embryonic mitochondria may be damaged by endogenous and exogenous factors such as ROS, apoptosis, and drugs.
ART procedures can improve the reproductive function of infertility patients; however, research on repeated ovarian stimulation has presented an adverse impact on mtDNA. After repeated ovarian stimulation in mice, mtDNA copy number decreased, while oocyte number increased, bringing abnormally distributed mitochondria, which can be suppressed with an antioxidant212. More importantly, mtDNA diminishing increased after using recombinant gonadotropin in primate oocytes213. However, injecting recombinant gonadotropin in golden hamsters raised mitochondria number, leading to insufficient pyruvate, and producing high levels of ROS214. Redox is maintained in equilibrium in normal physiological conditions, which is crucial for embryo development. Specific events during fertilization and bovine embryo development have been associated with redox states. Increased ROS level after fertilization was related to better embryonic development215. For instance, a brief exposure to 50-100 µM hydrogen peroxide resulted in a higher blastocyst rate in comparison with control group216. Although pyruvate is an essential substrate of mitochondrial respiration, high pyruvate levels can scavenge hydrogen peroxide, resulting in reduced oxidative stress and low ATP production217. Thus, low-concentration pyruvate is provided in an in vitro culture medium to limit the effect on redox states. This evidence indicated an undeniable role of mitochondrial homeostasis during embryonic development, where mtDNA optimization and balance of mitochondrial metabolism have significant impacts on the rate of embryo quality and blastocyst formation. Thus, ensuring mitochondrial health and capacity remains critical after fertilization, which is essential to define the birth rate.
EFFORTS TO IMPROVE OUTCOMES OF ART TREATMENT
The mutation of mtDNA and mitochondrial dysfunction can lead to infertility and can be inherited by offspring21. ART is an effective tool for the treatment of infertility, including those caused by abnormal mitochondria. Several procedures involve the combination of different ART methods to improve the success rate and limit inherited mtDNA mutation.
Pharmacological Agents
The use of chemicals, especially antioxidants, has been proposed in ART to improve mitochondrial function by reducing oxidative stress. Coenzyme Q10 (CoQ10) is a common endogenous quinone present in biological membranes. The critical role of CoQ in the mitochondria includes two functions: bioenergetic and antioxidant. It is an essential cofactor of mitochondrial respiratory complexes contributing to the mitochondrial ATP synthesis by the electron transfer process. The reduced form of coenzyme Q10 (rCoQ10) possesses antioxidant activity218. Patients with varicocele and oligozoospermia frequently have low CoQ10 levels. Both CoQ10 monotherapy and compound combination supplementation have shown positive effects on semen parameters. Strong antioxidant properties have the capacity to protect sperm membranes to stay intact by controlling ROS levels and maintaining efficient energy production219. Similarly, oocytes and embryos have high energy demands in female fertility, and the aging of ovaries is associated with mitochondrial dysfunction. CoQ10 supplementation was shown to improve ART outcomes in females and prevent aging220. Expression of oocyte mitochondrial genes was restored, and mitochondrial activity was enhanced with CoQ10 treatment221, and sufficient energy and reduced ROS levels promoted oocyte maturation, fertilization process, embryo development, and embryo implantation222. However, in a recent study, it was shown that the use of some mitoquinone, such as TPP and MitoQ at the concentration of 0.5 µM and 1 µM, respectively, could reduce cleavage rate, 4-cell embryos, and blastocyst rate223, suggesting that the use of antioxidants may disrupt the balance of mitochondrial bioenergetics, which negatively affects embryonic development. The use of L-carnitine has also been investigated, which was effective in increasing antioxidant glutathione (GSH) levels and reducing ROS production in mouse oocytes224. The data also showed that L-carnitine treatment improved oocyte maturation and blastocyst development in mice224. Other benefits of L-carnitine have also been extensively reviewed elsewhere, with minor warnings of the side effects of the supplement being included225. Another direction is to target calcium homeostasis in oocytes. The use of Ruthenium Red (RR), an inhibitor of Ca2+ uptake into mitochondria from the cytosol was shown to effectively reduce Ca2+ levels of porcine zygotes 6 hours after in vitro fertilization (IVF), which was associated with increased ATP production and mitochondrial membrane potential, and reduced mitochondrial ROS226. RR treatment, especially at the concentration of 20 µM, also significantly increased blastocyst development and quality of the zygotes226. Despite the effectiveness of pharmacological reagents, the side effects of those supplements in ART remain to be clarified. Future directions may continue with evaluation and examination of their effects on different stages of oocyte and embryonic development.
Mitochondrial Replacement
Given that mitochondrial capacity and function of germ cells determine fertility and healthy embryonic development, ensuring mitochondrial health poses a critical role in the reproductive system and fertility rate. Mitotherapy has been proposed as a potential method in ART, where preclinical and clinical data showed that transferring healthy mitochondria can support oocyte function, fertilization, and live birth rates. As discussed above, since the fetus primarily inherits mtDNA from the mother, mitochondrial replacement is currently the only technique available to help women with primary mitochondrial diseases caused by mtDNA mutations have healthy babies. Research has shown that approximately one in 5,000 people carry either nuclear DNA or mtDNA mutations that can potentially lead to mitochondrial diseases227. In Australia, the prevalence of individuals carrying a disease that could result in mtDNA mutations is one in 250, which may be inherited and negatively impact their lifetime228. Although the mtDNA mutation rate is 25 times higher than that of nuclear DNA, it is generally accepted that mutation levels must exceed 70% to cause clinical symptoms229, 230. Nevertheless, the burden of diseases caused by mtDNA mutations is lifelong and can be inherited across generations.
Mitochondria replacement is a type of mitotherapy in ART, where preclinical and clinical data showed that the transfer of healthy mitochondria can improve oocyte function and fertilization and healthy live birth rates. The transfer of mitochondria into aged oocytes or oocytes with pathogenic mutation of mtDNA has been performed and resulted in the births of “three-parent babies,” providing hopes for women with infertility or inherited mtDNA diseases231. Several technologies have been developed to perform mitochondria transfer into eggs and embryos to improve the outcomes of ART. These techniques include partial or total cytoplasmic transfer in oocytes, germinal vesicle transfer (GCT), maternal spindle transfer (MST), pronuclear transfer (PNT), blastomere transfer (BT), and polar body transfer (PBT), where PNT and MST are two most applicable ones22, 232. In brief, MST involves the transfer of the spindle, where chromosomes are packed in the affected oocyte, into the healthy oocyte of the donor, whose spindle is discarded233. As a result, a healthy oocyte with the mother’s chromosomes will be fertilized and transferred to the mother's uterus. On the other hand, PNT is performed after the oocytes of the mother and donor are fertilized233. The pronuclear of the affected oocytes will then be transferred into the healthy fertilized oocyte of the donor, whose pronuclear is removed. Even though the technology can help with infertile females in general, it must be noted that these techniques are mostly applied to female patients with mitochondrial disorders, in which mutations in mtDNA may affect the health of the offspring. Having newborns with mtDNA disorders is presented as a highly concerned aspect rather than infertility itself since mtDNA mutations are more likely to affect male than female fertility147. The implications associated with these techniques have been thoroughly analyzed but remain controversial from various perspectives, including science, economic, social, ethical, cultural, and possibly religious issues, which warrant further investigation and the involvement of multiple parties and disciplines234, 235.
Consequently, mitochondria replacement therapy requires more research to clarify the mechanism of mitochondria replacement throughout the body, as well as to assess its efficacy and safety regarding the welfare of the offspring. These biological challenges have raised ethical concerns for clinical applications and have limited the legalization of mitochondria transplantation to certain countries and thus, require considerable effort235, 236, 237.
Mitochondria Transplantation
Current advancements in ART have provided hope for infertile patients without mtDNA mutations, whose oocytes of low quality could not be fertilized, or the embryos could not reach the full developmental expectation, making the technology more approachable in terms of ethical and legal issues. Mitochondria transplantation involves the transfer of mitochondria into eggs or embryos of low quality, without the need to completely separate the existing mitochondria. Adding healthy mitochondria to low-quality oocytes was suggested to supplement the cells with fresh and functional mitochondria to meet the developmental requirements. In fact, mitochondria transfer, and transplantation have been utilized to rescue oxidative stress in multiple pathological models, proposing the potential application in human health238, 239, 240. Supplementing reproductive cells with functional mitochondria, either heterologously or autologously, is also discussed elsewhere in the fertility aspect22. However, since mtDNA and nuclear DNA mismatch appears to be the biggest ethical and practical issues in oocyte regeneration, autologous mitochondria transfer has been proposed to minimize these effects. Autologous stem cells were shown as an effective source of mitochondria for oocyte rejuvenation in both animal and human models and the direct injection of mitochondria together with sperm during intracytoplasmic sperm injection (ICSI) has been successfully applied. For instance, direct injection of autologous mitochondria from adipose-derived stem cells (ADSCs) was able to improve mtDNA numbers and quality of mature MII oocytes in aged mice241. The number of pups also increased after mitochondria were co-injected with ICSI into MII oocytes241. The transfer of mitochondria isolated from ADSCs has also been applied to enhance the quality of cryopreserved oocytes in mouse models242. The data showed that injecting isolated mitochondria from ADSCs into cryopreserved oocytes significantly enhanced ATP production in embryos of the 2-cell stage and increased the rates of blastulation in both oocytes and 2-cell embryos242. However, not all types of mitochondria can rescue poor-quality oocytes. In a mouse model, neither fresh (14 hours) nor aged (20-24 hours) post-ovulation oocytes showed any improvement in fertilization rate and embryonic development after the transfer of liver-derived mitochondria, even though oxygen consumption rate significantly increased243. Another study indicated that mitochondria derived from aged ADSCs to oocytes collected from young or aged (10-month-old) mice neither increased fertilization rates nor embryonic development244. These data emphasize the critical importance of mitochondrial selection for oocytes specifically and in mitochondrial transplantation technique in general. The compatibility and competence of donor mitochondria have a great impact on the cells and thus, define the outcomes of the therapy.
In fact, the effort has also been invested into finding the matching mitochondria for oocyte development. Oocyte mitochondria are immature and featured with spherical shape and little cristae formation, while early oocytes show low ROS production and low MMP, most probably to protect DNA integrity. The shape and metabolic profile of oocytes change with oocyte fertilization and embryonic development. Thus, the selection of mitochondria compatible with oocyte mitochondria would be beneficial for the recovery of low-quality oocytes. Based on this, Zhang and colleagues have shown that the transplantation of mitochondria from iPSCs, which contained mitochondria similar to oocytes, were efficient to improve egg quality, embryo quality, and live birth rates in aged mice245. A recent study by Jiang and colleagues has shown that the mitochondria from urine-derived MSCs (USCs) were efficient to improve mitochondria function in early embryonic development of age-oocytes after ICSI procedure246. The data emphasized that mitochondria from USCs were the most representative of oocyte mitochondria with spherical shape and immature cristae, while relatively low ROS and high MMP (mitochondrial membrane potential) and a high metabolic profile identified the competency in metabolism246. Transferring mitochondria purified from endometrial mesenchymal stem cells (EnMSCs), isolated from the endometrium, to improve oocyte quality and fertilization has also been investigated in mouse models247. Mitochondria purified from EnMSCs showed high MMP and low ROS when compared to endometrial stromal cells (ESCs), and positively improved oocyte quality, pregnancy rate, embryonic growth, and number of live births247. Compared to ADSC mitochondria, the isolation of iPSCs, USCs, and EnMSCs appears to be more challenging to perform. Overall, these data indicate the continuous efforts being made in mitotherapy, suggesting that the approach is promising and potential.
Another technique in mitochondrial replacement is autologous germline mitochondrial energy transfer (AUGMENT), introducing a technological advancement for rescuing infertility in aged women and has been tested clinically248. The method is based on the transfer of stem cell mitochondria from the female germline, which were referred to as oogonial stem cells (OSCs), and showed competency and similarity to egg mitochondria. Clinical trials of the method have been reported with positive results in improving fertilization rate and live births in women with repeatedly failed IVF, proposing a new method for ART. Based on this technique, different clinics have successfully reproduced the positive results in women with repeated IVF failure249, 250. However, a randomized pilot study involving infertile women, which employed AUGMENT from egg precursor cells to metaphase oocytes (MII oocytes), failed to improve the outcomes in the subject population251, which was argued to be generated from technical misunderstanding252. These results highlight that the accuracy in selecting cells for mitochondrial transplantation is important to the success of ART in women with repeated IVF failure. Given that different technologies have been shown successful with positive results in infertile women, the effort might also be contributed to make it even better feasible for a broader translation in different clinics, as different clinical setups might be met with difficulty in performing particular procedures.
It must be emphasized that autologous mitochondria supplementary techniques are only suitable for aged and low-quality eggs, where mtDNA is not affected by pathogenic mutations. The current technology in mitochondrial replacement into oocytes or embryos mostly involves the discarding of donor oocytes or fertilized eggs, which are encountered for ethical issues22. However, recent research has shown that this issue may not be a concern in the future as mitoception procedures could effectively transfer isolated mitochondria into both oocytes and zygotes253. In detail, the authors indicated that low temperature centrifugation at 500g for 5 minutes was efficient to transfer 50 ng/µL of isolated human mitochondria from peripheral blood mononuclear cells into mouse oocytes253. Even though the data could not give a conclusion on the retention of human mitochondria in mouse pups, it has proposed a non-invasive technique for transferring healthy mitochondria into oocytes253. Tang and colleagues have developed a non-invasive technique for autologous mitochondria transfer by using umbilical cord-derived mesenchymal stem cells (UC-MSCs)254. In the experiments, UC-MSCs collected since birth of the female mice were cryopreserved. When those female mice were aged, UC-MSCs were thawed and induced to form granulosa cells, which aggregated around aged oocytes. The authors observed mitochondria from the induced granulosa being transferred into oocytes via transzonal filopodia, which significantly improved oocyte quality, embryonic development, and live births in aged mice254. Even though the technique is promising, the need to cryopreserve UC-MSCs at birth might be difficult in application, and further investigations would be warranted to understand the mechanism of mitochondria transfer in those mice.
| Species | Transplantation methods | Donor cells | Host cells | Outcomes | Reference |
| Mice | Autologous Mito-ICSI | ADSCs | MII oocytes | -Improved mtDNA number, quality of mature oocytes -Increased blastocyst and birth rate | 241 |
| Mice | Autologous Mito-ICSI | ADSC | Cryopreserved oocytes | Improved fertilization rate and embryonic development | 242 |
| Mice | Autologous microinjection | iPSC | Fertilized oocytes | Improve blastocyst development, preimplantation rate, and birth rate | 245 |
| Mice | Autologous Mito-ICSI | EnMSC | GV oocytes | -Improve oocyte maturation and quality in vivo embryonic development and birth rate | 247 |
| Human | Autologous Mito-ICSI | USC | MI oocytes | -Improved mitochondrial function in early embryos | 246 |
| Human | Autologous Mito-ICSI | OPCs/ OSCs | Oocytes | -Improve pregnancy and live birth rates | 249 , 250 , 255 |
Embryo selection
Embryo selection is the method to evaluate embryo quality and select embryos with biomarkers potential for development and implantation. The approaches can involve direct analysis of mtDNA blastomeres by biopsy or indirect analysis of somatic cells and cell-free mtDNA. Although embryonic mitochondria have critical embryo functions, analysis of mtDNA blastomeres is still under discussion. A few studies have shown appreciation of mtDNA analysis, which this biomarker may serve as a predictive indicator for embryo development. Some studies investigated the relative mtDNA numbers on oocyte and embryo quality, and treatment outcomes. Oocytes with low mtDNA content are associated with high fertilization failure200, 256, 257. In contrast, lower trophectoderm (TE) mtDNA levels were correlated with higher trophoblast quality. In day-5 transferred embryonic outcomes, higher implantation rate and live birth were associated with low TE mtDNA content258, 259. Nevertheless, no relationship was found between inner cell mass and mtDNA content260. The levels of mtDNA might be dependent on the biopsied date, with higher levels observed on day 5 compared to day 6261. Contrasting results have been reported that the biopsy on day 6 or day 7 was higher than day 5 mtDNA levels262. Furthermore, no correlation was found between TE mtDNA levels and embryonic implantation ability, which disagreed with the ability to use mtDNA content as an embryo selection criterion to transfer263. As a result, TE biopsy might be an advantageous approach for mtDNA analysis, however, biopsy still poses a risk of injury, which can lead to embryonic damage.
Cumulus cells (CCs) originate from granulosa cells, which surround oocytes, creating the oocyte-cumulus complex (OCC). Cumulus cell function is a metabolic cooperation during oocyte development, meiosis, fertilization, and early embryo development264. Besides producing cell cycle signals to oocytes, the cumulus cell glycolysis produces pyruvate to supply oocyte OXPHOS265. Equally, cumulus cell metabolism is incapable in the absence of the oocytes. Several studies have demonstrated factors of cumulus cells that can predict the oocyte quality266, 267. The genes PFKP, HAS2, TNFIP6, PTGS, and PTX3 of CCs have been shown to be correlated with embryo potential development and implantation268, 269. Moreover, the levels of mtDNA in CCs are not significant in predicting embryo development and outcome treatment270. Thus, evaluation of CC metabolism and gene expression could be a potential method for embryo selection.
Cell-free mtDNA (cf-mtDNA) is also a possible non-invasive marker for investigating mitochondrial health. Mitochondria communicate adaptively with stress by signaling cf-mtDNA molecules. Most mtDNA mutations in embryos have the potential to be identified from mtDNA in conditioned media271. The cf-mtDNA collected from media culture positively correlated with morphokinetic parameters from the beginning of blastulation to full blastocyst stage, where higher cf-mtDNA content reflects poorer quality blastocyst272. On the other hand, high cf-mtDNA content from cleavage-stage embryo culture medium is a positive predictive marker of good quality embryos273, 274.
CONCLUSION
Mitotherapy has been applied in ART and infertility for decades, and the innovation in research and technology is contributing greatly to the development of better and more effective procedures and approaches. Some of the techniques have been shown successful in clinical application, providing chances for women with low fertility. It is shown that ART with repeated effort, may potentially replace the natural birth rate, solving the issues of declining population worldwide [8-10]. With the current advancement in mitotherapy, a broader range of patients and customers will benefit, especially women with mtDNA disorders, or with aged, low-quality, and unknown infertility. The development of mitochondrial transplantation techniques has shifted ART toward a less invasive and more ethical approach, where the isolation and removal of the donor’s oocyte and embryonic nuclei are no longer required. The selection of cell sources for mitochondria isolation has been extensively investigated, and the combination of mitotherapy with stem cell therapy has shown positive results in tested procedures. However, there is still room for further refinement of ART employing mitotherapy. Exploring different aspects of mitochondria selection, which involves new insights into mitochondrial biology and metabolism, will lead to further improvements.
Current data showed that mitochondria transplantation might increase the risk of heart defects and other morphological concerns; however, it must be noted that ART, and particularly IVF technology itself, has been shown to raise health issues in IVF babies, including cardiovascular disease (CHD)275, 276. Perhaps the issues related to mito-nuclear mismatch are additional to the already-present risks of exposing germ cells to the in vitro environment. Therefore, future research should focus on examining various factors and chemicals in the in vitro environment, as well as the effects of the therapeutic procedures used in the current technologies. Furthermore, it should be distinguished between the investigation of additional mitochondria in germ cells and the current challenges. A deeper understanding of the mechanisms underlying mitochondrial transplantation and the involvement of donor mitochondria in host cell biology and function should be highlighted. Whether the safety and effectiveness of mitotherapy can be sustained over time throughout a human lifetime are critical questions. Thus, a longitudinal study of individuals conceived with mitotherapy is essential. This will require long-term commitment and collaboration from research, biological, and medical communities. Although concerns regarding the identification and acceptance of parenthood or religious beliefs are important and may be related to the ethics and accessibility of mitotherapy, they are beyond the scope of this work and should be thoroughly elaborated in collaboration with other fields such as humanities or philosophy. Overall, these questions should not hinder the progress in therapeutic development, with the focus remaining on safety, efficacy, and innovation.
Other potential directions have also been considered for age-related infertility, such as using a cell-free scaffold seeded with stem cells for ovarian restoration, which can be possibly performed in vivo277. It must be noted that the majority of the present mitochondria transplantation and transfer technologies are focused on female rather than male reproduction, most probably because sperms carry a limited number of mitochondria, which are strictly removed at the fertilization stages, and are difficult to manipulate when compared to eggs. The targeting of mitochondrial issues in male infertility is currently directed to the use of chemicals and medicines to improve mitochondrial function278, 279. Other directions involve the regeneration of testicular tissues and supporting spermatogenesis such as stem cell therapy280 and making testicular organoids281. With the continuous effort being made in research and medicine, it is promising that more refined treatments will be accessible to infertile patients.
Abbreviations
ADSCs - Adipose-Derived Stem Cells, AKT - Protein Kinase B (also known as RAC-alpha serine/threonine-protein kinase), ART - Assisted Reproductive Technology, ATP - Adenosine Triphosphate, AUGMENT - Autologous Germline Mitochondrial Energy Transfer, BT - Blastomere Transfer, CCs - Cumulus Cells, cf-mtDNA - Cell-Free Mitochondrial DNA, CHD - Congenital Heart Disease, CoQ10 - Coenzyme Q10, EnMSCs - Endometrial Mesenchymal Stem Cells, Fe2+ - Ferrous Iron, GCT - Germinal Vesicle Transfer, GSH - Glutathione, H2O2 - Hydrogen Peroxide, ICSI - Intracytoplasmic Sperm Injection, iPSCs - Induced Pluripotent Stem Cells, IUI - Intrauterine Insemination, IVF - In Vitro Fertilization, MMP - Mitochondrial Membrane Potential, MOMP - Mitochondrial Outer Membrane Permeabilization, MST - Maternal Spindle Transfer, mtDNA - Mitochondrial DNA, NADH - Nicotinamide Adenine Dinucleotide (Reduced Form), NADPH - Nicotinamide Adenine Dinucleotide Phosphate (Reduced Form), Nrf2 - Nuclear Factor Erythroid 2-Related Factor 2, O2●− - Superoxide Anion, OCC - Oocyte-Cumulus Complex, OSCs - Oogonial Stem Cells, OXPHOS - Oxidative Phosphorylation, PBT - Polar Body Transfer, PI3K - Phosphatidylinositol 3-Kinase, PNT - Pronuclear Transfer, PTP - Permeability Transition Pore, rCoQ10 - Reduced Coenzyme Q10, ROS - Reactive Oxygen Species, RR - Ruthenium Red, SDF - Sperm DNA Fragmentation, Sirt1 - Sirtuin 1, SOD - Superoxide Dismutase, TE - Trophectoderm, TFAM - Mitochondrial Transcription Factor A, UC-MSCs - Umbilical Cord-Derived Mesenchymal Stem Cells, USCs - Urine-Derived Mesenchymal Stem Cells, ●OH - Hydroxyl Radical
Acknowledgments
None.
Author’s contributions
Oanh TH Phan: Conceptualization, Visualization, Writing - Original draft. Tran TC Trinh: Conceptualization, Writing- Original draft, Writing- Review and editing. Van TT Nguyen: Conceptualization, Supervision, Visualization, Writing -original draft, Writing- Review and editing. All authors read and approved the final manuscript.
Funding
None.
Availability of data and materials
Not applicable.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
-
Croix
D. de la,
Pommeret
A.,
Childbearing postponement, its option value, and the biological clock. Journal of Economic Theory.
2021;
193
:
105231
.
View Article Google Scholar -
Goisis
A.,
Maternal Age at First Birth and Parental Support: Evidence From the UK Millennium Cohort Study. Population Research and Policy Review.
2023;
42
(5)
:
75
.
View Article Google Scholar -
Wyns
C.,
Bergh
C.,
Calhaz-Jorge
C.,
Geyter
C. De,
Kupka
M.S.,
Motrenko
T.,
Reproduction
European IVF-monitoring Consortium (EIM)\textdaggerdbl for the European Society of Human,
(ESHRE)
Embryology,
ART in Europe, 2016: results generated from European registries by ESHRE. Human Reproduction Open.
2020;
2020
(3)
:
hoaa032
.
View Article PubMed Google Scholar -
Smeenk
J.,
Wyns
C.,
De Geyter
C.,
Kupka
M.,
Bergh
C.,
Cuevas Saiz
I.,
European IVF Monitoring Consortium (EIM) for the European Society of Human Reproduction
Embryology (ESHRE)
ART in Europe, 2019: results generated from European registries by ESHRE\textdagger. Human Reproduction (Oxford, England).
2023;
38
(12)
:
2321-38
.
View Article PubMed Google Scholar -
Lai
S.,
Wang
R.,
van Wely
M.,
Costello
M.,
Farquhar
C.,
Bensdorp
A.J.,
IVF versus IUI with ovarian stimulation for unexplained infertility: a collaborative individual participant data meta-analysis. Human Reproduction Update.
2024;
30
(2)
:
174-85
.
View Article PubMed Google Scholar -
Man
J.K.,
Parker
A.E.,
Broughton
S.,
Ikhlaq
H.,
Das
M.,
Should IUI replace IVF as first-line treatment for unexplained infertility? A literature review. BMC Women's Health.
2023;
23
(1)
:
557
.
View Article PubMed Google Scholar -
Gunn
D.D.,
Bates
G.W.,
Evidence-based approach to unexplained infertility: a systematic review. Fertility and Sterility.
2016;
105
(6)
:
1566-1574.e
.
View Article PubMed Google Scholar -
Wang
N.,
Yin
X.,
Tao
Y.,
Wang
Y.,
Zhu
Q.,
Cumulative live birth rates over multiple complete cycles of in vitro fertilisation cycles: 10-year cohort study of 20,687 women following freeze-all strategy from one single centre. Archives of Gynecology and Obstetrics.
2022;
305
(1)
:
251-9
.
View Article PubMed Google Scholar -
Gnoth
C.,
Maxrath
B.,
Skonieczny
T.,
Friol
K.,
Godehardt
E.,
Tigges
J.,
Final ART success rates: a 10 years survey. Human Reproduction (Oxford, England).
2011;
26
(8)
:
2239-46
.
View Article PubMed Google Scholar -
Dong
X.,
Xue
X.,
Live birth rate following a failed first in vitro fertilization cycle with no embryos for transfer. Scientific Reports.
2023;
13
(1)
:
8343
.
View Article PubMed Google Scholar -
Spoletini
R.,
Trani
M. Di,
Renzi
A.,
Fedele
F.,
Scaravelli
G.,
Psychological care for infertile couples undergoing assisted reproductive technology: a national study on the characteristics of counselling services. Annali dell'Istituto Superiore di Sanità.
2022;
58
(1)
:
46-54
.
PubMed Google Scholar -
Langher
V.,
Fedele
F.,
Caputo
A.,
Marchini
F.,
Aragona
C.,
Extreme Desire for Motherhood: Analysis of Narratives From Women Undergoing Assisted Reproductive Technology (ART). Europe's Journal of Psychology.
2019;
15
(2)
:
292-311
.
View Article PubMed Google Scholar -
Jansen
C.,
Kuhlmann
E.,
Scharli
P.,
Schick
M.,
Ditzen
B.,
Langer
L.,
`A sorrow shared …': a qualitative content analysis of what couples with recurrent miscarriages expect from one another and their families and friends. Human Reproduction Open.
2022;
2022
(3)
:
hoac032
.
View Article PubMed Google Scholar -
Moura-Ramos
M.,
Gameiro
S.,
Canavarro
M.C.,
Soares
I.,
Almeida-Santos
T.,
Does infertility history affect the emotional adjustment of couples undergoing assisted reproduction? the mediating role of the importance of parenthood. British Journal of Health Psychology.
2016;
21
(2)
:
302-17
.
View Article PubMed Google Scholar -
Scaravelli
G.,
Fedele
F.,
Spoletini
R.,
Monaco
S.,
Renzi
A.,
Di Trani
M.,
Toward a Personalized Psychological Counseling Service in Assisted Reproductive Technology Centers: A Qualitative Analysis of Couples' Needs. Journal of Personalized Medicine.
2022;
13
(1)
:
73
.
View Article PubMed Google Scholar -
Nicoloro-SantaBarbara
J.,
Busso
C.,
Moyer
A.,
Lobel
M.,
Just relax and you'll get pregnant? Meta-analysis examining women's emotional distress and the outcome of assisted reproductive technology. Social Science & Medicine.
2018;
213
:
54-62
.
View Article PubMed Google Scholar -
Kremer
F.,
Ditzen
B.,
Wischmann
T.,
Effectiveness of psychosocial interventions for infertile women: A systematic review and meta-analysis with a focus on a method-critical evaluation. PLoS One.
2023;
18
(2)
:
e0282065
.
View Article PubMed Google Scholar -
Leslie
S.W.,
Soon-Sutton
T.L.,
Khan
M.A.,
Male Infertility, in StatPearls. 2024, StatPearls Publishing. 2024
.
-
Vander Borght
M.,
Wyns
C.,
Fertility and infertility: definition and epidemiology. Clinical Biochemistry.
2018;
62
:
2-10
.
View Article PubMed Google Scholar -
Demain
L.A.,
Conway
G.S.,
Newman
W.G.,
Genetics of mitochondrial dysfunction and infertility. Clinical Genetics.
2017;
91
(2)
:
199-207
.
View Article PubMed Google Scholar -
Tesarik
J.,
Mendoza-Tesarik
R.,
Mitochondria in Human Fertility and Infertility. International Journal of Molecular Sciences.
2023;
24
(10)
:
8950
.
View Article PubMed Google Scholar -
Rodríguez-Varela
C.,
Herraiz
S.,
Labarta
E.,
Mitochondrial enrichment in infertile patients: a review of different mitochondrial replacement therapies. Therapeutic Advances in Reproductive Health.
2021;
15
:
26334941211023544
.
View Article PubMed Google Scholar -
Dobler
R.,
Dowling
D.K.,
Morrow
E.H.,
Reinhardt
K.,
A systematic review and meta-analysis reveals pervasive effects of germline mitochondrial replacement on components of health. Human Reproduction Update.
2018;
24
(5)
:
519-34
.
View Article PubMed Google Scholar -
Chinnery
P.F.,
Mitochondrial Replacement in the Clinic
Mitochondrial Replacement in the Clinic. The New England Journal of Medicine.
2020;
382
(19)
:
1855-7
.
View Article PubMed Google Scholar -
Dimond
R.,
Social and ethical issues in mitochondrial donation. British Medical Bulletin.
2015;
115
(1)
:
173-82
.
View Article PubMed Google Scholar -
Tatone
C.,
Di Emidio
G.,
Mitochondria Biology in Reproductive Function. Antioxidants.
2022;
11
(10)
:
1978
.
View Article PubMed Google Scholar -
Kasapoğlu
I.,
Seli
E.,
Mitochondrial Dysfunction and Ovarian Aging. Endocrinology.
2020;
161
(2)
:
bqaa001
.
View Article PubMed Google Scholar -
Allen
J.F.,
de Paula
W.B.,
Mitochondrial genome function and maternal inheritance. Biochemical Society Transactions.
2013;
41
(5)
:
1298-304
.
View Article PubMed Google Scholar -
Igarashi
H.,
Takahashi
T.,
Nagase
S.,
Oocyte aging underlies female reproductive aging: biological mechanisms and therapeutic strategies. Reproductive Medicine and Biology.
2015;
14
(4)
:
159-69
.
View Article PubMed Google Scholar -
Sturmey
R.G.,
Reis
A.,
Leese
H.J.,
McEvoy
T.G.,
Role of fatty acids in energy provision during oocyte maturation and early embryo development. Reproduction in Domestic Animals.
2009;
44
(s3)
:
50-8
.
View Article PubMed Google Scholar -
Miao
Y.L.,
Williams
C.J.,
Calcium signaling in mammalian egg activation and embryo development: the influence of subcellular localization. Molecular Reproduction and Development.
2012;
79
(11)
:
742-56
.
View Article PubMed Google Scholar -
Homer
H.A.,
The Role of Oocyte Quality in Explaining "Unexplained" Infertility. Seminars in Reproductive Medicine.
2020;
38
(1)
:
21-8
.
PubMed Google Scholar -
Kirillova
A.,
Smitz
J.E.,
Sukhikh
G.T.,
Mazunin
I.,
The Role of Mitochondria in Oocyte Maturation. Cells.
2021;
10
(9)
:
2484
.
View Article PubMed Google Scholar -
Jansen
R.P.,
Germline passage of mitochondria: quantitative considerations and possible embryological sequelae. Human Reproduction (Oxford, England).
2000;
15
:
112-28
.
View Article PubMed Google Scholar -
Romero-Garcia
S.,
Prado-Garcia
H.,
Mitochondrial calcium: transport and modulation of cellular processes in homeostasis and cancer (Review). International Journal of Oncology.
2019;
54
(4)
:
1155-67
.
View Article PubMed Google Scholar -
Carafoli
E.,
Intracellular calcium homeostasis. Annual Review of Biochemistry.
1987;
56
(1)
:
395-433
.
View Article PubMed Google Scholar -
du Plessis
S.S.,
Agarwal
A.,
Mohanty
G.,
van der Linde
M.,
Oxidative phosphorylation versus glycolysis: what fuel do spermatozoa use?. Asian Journal of Andrology.
2015;
17
(2)
:
230-5
.
View Article PubMed Google Scholar -
Ryu
B.Y.,
Orwig
K.E.,
Kubota
H.,
Avarbock
M.R.,
Brinster
R.L.,
Phenotypic and functional characteristics of spermatogonial stem cells in rats. Developmental Biology.
2004;
274
(1)
:
158-70
.
View Article PubMed Google Scholar -
Singh
S.R.,
Burnicka-Turek
O.,
Chauhan
C.,
Hou
S.X.,
Spermatogonial stem cells, infertility and testicular cancer. Journal of Cellular and Molecular Medicine.
2011;
15
(3)
:
468-83
.
View Article PubMed Google Scholar -
Costa
J.,
Braga
P.C.,
Rebelo
I.,
Oliveira
P.F.,
Alves
M.G.,
Mitochondria Quality Control and Male Fertility. Biology (Basel).
2023;
12
(6)
:
827
.
View Article PubMed Google Scholar -
Varuzhanyan
G.,
Chan
D.C.,
Mitochondrial dynamics during spermatogenesis. Journal of Cell Science.
2020;
133
(14)
:
jcs235937
.
View Article PubMed Google Scholar -
Varuzhanyan
G.,
Mitochondrial dynamics and mitophagy during male germline developmentCalifornia Institute of Technology 2020.
Google Scholar -
Oko
R.,
Clermont
Y.,
Controls of Serm MotilityCRC Press 2020.
View Article Google Scholar -
Sun
X.,
Yang
W.X.,
Mitochondria: transportation, distribution and function during spermiogenesis. Advances in Bioscience and Biotechnology.
2010;
1
(02)
:
97-109
.
View Article Google Scholar -
Voigt
A.L.,
Kondro
D.A.,
Powell
D.,
Valli-Pulaski
H.,
Ungrin
M.,
Stukenborg
J.B.,
Unique metabolic phenotype and its transition during maturation of juvenile male germ cells. The FASEB Journal.
2021;
35
(5)
:
e21513
.
View Article PubMed Google Scholar -
Amaral
S.,
Amaral
A.,
Ramalho-Santos
J.,
Aging and male reproductive function: a mitochondrial perspective. Frontiers in Bioscience (Scholar Edition).
2013;
5
(1)
:
181-97
.
View Article PubMed Google Scholar -
Kusminski
C.M.,
Scherer
P.E.,
Mitochondrial dysfunction in white adipose tissue. Trends in Endocrinology and Metabolism.
2012;
23
(9)
:
435-43
.
View Article PubMed Google Scholar -
Diaz-Vegas
A.,
Sanchez-Aguilera
P.,
Krycer
J.R.,
Morales
P.E.,
Monsalves-Alvarez
M.,
Cifuentes
M.,
Is Mitochondrial Dysfunction a Common Root of Noncommunicable Chronic Diseases?. Endocrine Reviews.
2020;
41
(3)
:
bnaa005
.
View Article PubMed Google Scholar -
Bevilacqua
A.,
Carlomagno
G.,
Gerli
S.,
Montanino Oliva
M.,
Devroey
P.,
Lanzone
A.,
Results from the International Consensus Conference on myo-inositol and D-chiro-inositol in Obstetrics and Gynecology\textemdashassisted reproduction technology. Gynecological Endocrinology.
2015;
31
(6)
:
441-6
.
View Article PubMed Google Scholar -
Cecchino
G.N.,
Seli
E.,
Alves da Motta
E.L.,
García-Velasco
J.A.,
The role of mitochondrial activity in female fertility and assisted reproductive technologies: overview and current insights. Reproductive Biomedicine Online.
2018;
36
(6)
:
686-97
.
View Article PubMed Google Scholar -
Durairajanayagam
D.,
Singh
D.,
Agarwal
A.,
Henkel
R.,
Causes and consequences of sperm mitochondrial dysfunction. Andrologia.
2021;
53
(1)
:
e13666
.
View Article PubMed Google Scholar -
Faja
F.,
Carlini
T.,
Coltrinari
G.,
Finocchi
F.,
Nespoli
M.,
Pallotti
F.,
Human sperm motility: a molecular study of mitochondrial DNA, mitochondrial transcription factor A gene and DNA fragmentation. Molecular Biology Reports.
2019;
46
(4)
:
4113-21
.
View Article PubMed Google Scholar -
Holyoake
A.J.,
McHugh
P.,
Wu
M.,
O'Carroll
S.,
Benny
P.,
Sin
I.L.,
High incidence of single nucleotide substitutions in the mitochondrial genome is associated with poor semen parameters in men. International Journal of Andrology.
2001;
24
(3)
:
175-82
.
View Article PubMed Google Scholar -
Saleh Jaweesh
M.,
Hammadeh
M.E.,
Dahadhah
F.W.,
Al Zoubi
M.S.,
Amor
H.,
Association between the single nucleotide variants of the mitochondrial cytochrome B gene (MT-CYB) and the male infertility. Molecular Biology Reports.
2022;
49
(5)
:
3609-16
.
View Article PubMed Google Scholar -
Nakada
K.,
Sato
A.,
Yoshida
K.,
Morita
T.,
Tanaka
H.,
Inoue
S.,
Mitochondria-related male infertility. Proceedings of the National Academy of Sciences of the United States of America.
2006;
103
(41)
:
15148-53
.
View Article PubMed Google Scholar -
Maurya
S.,
Kesari
K.K.,
Roychoudhury
S.,
Kolleboyina
J.,
Jha
N.K.,
Jha
S.K.,
Metabolic Dysregulation and Sperm Motility in Male Infertility. Advances in Experimental Medicine and Biology.
2022;
1358
:
257-73
.
View Article PubMed Google Scholar -
Tímermans
A.,
Otero
F.,
Garrido
M.,
Gosálvez
J.,
Johnston
S.,
Fernández
J.L.,
The relationship between sperm nuclear DNA fragmentation, mitochondrial DNA fragmentation, and copy number in normal and abnormal human ejaculates. Andrology.
2024;
12
(4)
:
870-80
.
View Article PubMed Google Scholar -
Amaral
A.,
Ramalho-Santos
J.,
St John
J.C.,
The expression of polymerase gamma and mitochondrial transcription factor A and the regulation of mitochondrial DNA content in mature human sperm. Human Reproduction (Oxford, England).
2007;
22
(6)
:
1585-96
.
View Article PubMed Google Scholar -
Attia
H.,
Mitochondrial and oxidant profile in male infertility: tfam and gpx-1 gene expression in human sperm. Doctoral thesis, Faculty of Medicine, Polythecnic University of Marche . 2023
.
-
Fernández-Silva
P.,
Enriquez
J.A.,
Montoya
J.,
Replication and transcription of mammalian mitochondrial DNA. Experimental Physiology.
2003;
88
(1)
:
41-56
.
View Article PubMed Google Scholar -
Karimian
M.,
Babaei
F.,
Large-scale mtDNA deletions as genetic biomarkers for susceptibility to male infertility: A systematic review and meta-analysis. International Journal of Biological Macromolecules.
2020;
158
:
85-93
.
View Article PubMed Google Scholar -
Gholinezhad
M.,
Yousefnia-Pasha
Y.,
Hosseinzadeh Colagar
A.,
Mohammadoo-Khorasani
M.,
Bidmeshkipour
A.,
Comparison of large-scale deletions of the sperm mitochondrial DNA in normozoospermic and asthenoteratozoospermic men. Journal of Cellular Biochemistry.
2019;
120
(2)
:
1958-68
.
View Article PubMed Google Scholar -
Arck
P.C.,
Overall
R.,
Spatz
K.,
Liezman
C.,
Handjiski
B.,
Klapp
B.F.,
Towards a free radical theory of graying\textquotedblright: melanocyte apoptosis in the aging human hair follicle is an indicator of oxidative stress induced tissue damage. The FASEB Journal.
2006;
20
(9)
:
1567-9
.
View Article PubMed Google Scholar -
Bahrehmand Namaghi
I.,
Vaziri
H.,
Sperm mitochondrial DNA deletion in Iranian infertiles with asthenozoospermia. Andrologia.
2017;
49
(3)
.
View Article PubMed Google Scholar -
Kao
S.H.,
Chao
H.T.,
Wei
Y.H.,
Multiple deletions of mitochondrial DNA are associated with the decline of motility and fertility of human spermatozoa. Molecular Human Reproduction.
1998;
4
(7)
:
657-66
.
View Article PubMed Google Scholar -
Lee
C.M.,
Characterization of age-associated mitochondrial dysfunction in mammalian skeletal muscleThe University of Wisconsin-Madison 1996.
Google Scholar -
Mundy
A.J.,
Ryder
T.A.,
Edmonds
D.K.,
Asthenozoospermia and the human sperm mid-piece. Human Reproduction (Oxford, England).
1995;
10
(1)
:
116-9
.
View Article PubMed Google Scholar -
Pelliccione
F.,
Micillo
A.,
Cordeschi
G.,
D'Angeli
A.,
Necozione
S.,
Gandini
L.,
Altered ultrastructure of mitochondrial membranes is strongly associated with unexplained asthenozoospermia. Fertility and Sterility.
2011;
95
(2)
:
641-6
.
View Article PubMed Google Scholar -
Ferramosca
A.,
Provenzano
S.P.,
Coppola
L.,
Zara
V.,
Mitochondrial respiratory efficiency is positively correlated with human sperm motility. Urology.
2012;
79
(4)
:
809-14
.
View Article PubMed Google Scholar -
Pucci
B.,
Kasten
M.,
Giordano
A.,
Cell cycle and apoptosis. Neoplasia (New York, N.Y.).
2000;
2
(4)
:
291-9
.
View Article PubMed Google Scholar -
Fadeel
B.,
Orrenius
S.,
Apoptosis: a basic biological phenomenon with wide-ranging implications in human disease. Journal of Internal Medicine.
2005;
258
(6)
:
479-517
.
View Article PubMed Google Scholar -
Snoeckx
L.H.,
Cornelussen
R.N.,
Van Nieuwenhoven
F.A.,
Reneman
R.S.,
Van Der Vusse
G.J.,
Heat shock proteins and cardiovascular pathophysiology. Physiological Reviews.
2001;
81
(4)
:
1461-97
.
View Article PubMed Google Scholar -
Koppers
A.J.,
Mitchell
L.A.,
Wang
P.,
Lin
M.,
Aitken
R.J.,
Phosphoinositide 3-kinase signalling pathway involvement in a truncated apoptotic cascade associated with motility loss and oxidative DNA damage in human spermatozoa. The Biochemical Journal.
2011;
436
(3)
:
687-98
.
View Article PubMed Google Scholar -
Davidson
S.M.,
Adameová
A.,
Barile
L.,
Cabrera-Fuentes
H.A.,
Lazou
A.,
Pagliaro
P.,
Action (CA16225)
EU-CARDIOPROTECTION COST,
Mitochondrial and mitochondrial-independent pathways of myocardial cell death during ischaemia and reperfusion injury. Journal of Cellular and Molecular Medicine.
2020;
24
(7)
:
3795-806
.
View Article PubMed Google Scholar -
Baliga
B.,
Kumar
S.,
Apaf-1/cytochrome c apoptosome: an essential initiator of caspase activation or just a sideshow?. Cell Death and Differentiation.
2003;
10
(1)
:
16-8
.
View Article PubMed Google Scholar -
Amaral
A.,
Lourenço
B.,
Marques
M.,
Ramalho-Santos
J.,
Mitochondria functionality and sperm quality. Reproduction (Cambridge, England).
2013;
146
(5)
:
163-74
.
View Article PubMed Google Scholar -
Giorgio
V.,
von Stockum
S.,
Antoniel
M.,
Fabbro
A.,
Fogolari
F.,
Forte
M.,
Dimers of mitochondrial ATP synthase form the permeability transition pore. Proceedings of the National Academy of Sciences of the United States of America.
2013;
110
(15)
:
5887-92
.
View Article PubMed Google Scholar -
Weng
S.L.,
Taylor
S.L.,
Morshedi
M.,
Schuffner
A.,
Duran
E.H.,
Beebe
S.,
Caspase activity and apoptotic markers in ejaculated human sperm. Molecular Human Reproduction.
2002;
8
(11)
:
984-91
.
View Article PubMed Google Scholar -
Paasch
U.,
Grunewald
S.,
Agarwal
A.,
Glandera
H.J.,
Activation pattern of caspases in human spermatozoa. Fertility and Sterility.
2004;
81
:
802-9
.
View Article PubMed Google Scholar -
Barbonetti
A.,
Castellini
C.,
Di Giammarco
N.,
Santilli
G.,
Francavilla
S.,
Francavilla
F.,
In vitro exposure of human spermatozoa to bisphenol A induces pro-oxidative/apoptotic mitochondrial dysfunction. Reproductive Toxicology (Elmsford, N.Y.).
2016;
66
:
61-7
.
View Article PubMed Google Scholar -
Said
T.M.,
Agarwal
A.,
Zborowski
M.,
Grunewald
S.,
Glander
H.J.,
Paasch
U.,
Utility of magnetic cell separation as a molecular sperm preparation technique. Journal of Andrology.
2008;
29
(2)
:
134-42
.
View Article PubMed Google Scholar -
Sakkas
D.,
Ramalingam
M.,
Garrido
N.,
Barratt
C.L.,
Sperm selection in natural conception: what can we learn from Mother Nature to improve assisted reproduction outcomes?. Human Reproduction Update.
2015;
21
(6)
:
711-26
.
View Article PubMed Google Scholar -
Jiang
B.H.,
Liu
L.Z.,
PI3K/PTEN signaling in tumorigenesis and angiogenesis. Biochimica et Biophysica Acta.
2008;
1784
(1)
:
150-8
.
View Article PubMed Google Scholar -
Szymonowicz
K.,
Oeck
S.,
Malewicz
N.M.,
Jendrossek
V.,
New Insights into Protein Kinase B/Akt Signaling: Role of Localized Akt Activation and Compartment-Specific Target Proteins for the Cellular Radiation Response. Cancers (Basel).
2018;
10
(3)
:
78
.
View Article PubMed Google Scholar -
Olivo
E.,
La Chimia
M.,
Ceramella
J.,
Catalano
A.,
Chiaradonna
F.,
Sinicropi
M.S.,
Moving beyond the Tip of the Iceberg: DJ-1 Implications in Cancer Metabolism. Cells.
2022;
11
(9)
:
1432
.
View Article PubMed Google Scholar -
Aitken
R.J.,
Baker
M.A.,
Causes and consequences of apoptosis in spermatozoa; contributions to infertility and impacts on development. The International Journal of Developmental Biology.
2013;
57
(2-4)
:
265-72
.
View Article PubMed Google Scholar -
Barroso
G.,
Valdespin
C.,
Vega
E.,
Kershenovich
R.,
Avila
R.,
Avendaño
C.,
Developmental sperm contributions: fertilization and beyond. Fertility and Sterility.
2009;
92
(3)
:
835-48
.
View Article PubMed Google Scholar -
Jourdain
A.,
Martinou
J.C.,
Mitochondrial outer-membrane permeabilization and remodelling in apoptosis. The International Journal of Biochemistry & Cell Biology.
2009;
41
(10)
:
1884-9
.
View Article PubMed Google Scholar -
Yu
R.,
Lendahl
U.,
Nistér
M.,
Zhao
J.,
Regulation of Mammalian Mitochondrial Dynamics: opportunities and Challenges. Frontiers in Endocrinology (Lausanne).
2020;
11
:
374
.
View Article PubMed Google Scholar -
Bock
F.J.,
Tait
S.W.,
Mitochondria as multifaceted regulators of cell death. Nature Reviews. Molecular Cell Biology.
2020;
21
(2)
:
85-100
.
View Article PubMed Google Scholar -
Wang
R.,
Huang
Y.,
Yu
L.,
Li
S.,
Li
J.,
Han
B.,
The role of mitochondrial dynamics imbalance in hexavalent chromium-induced apoptosis and autophagy in rat testis. Chemico-Biological Interactions.
2023;
374
:
110424
.
View Article PubMed Google Scholar -
Rincheval
V.,
Bergeaud
M.,
Mathieu
L.,
Leroy
J.,
Guillaume
A.,
Mignotte
B.,
Differential effects of Bcl-2 and caspases on mitochondrial permeabilization during endogenous or exogenous reactive oxygen species-induced cell death. Cell Biology and Toxicology.
2012;
28
(4)
:
239-53
.
View Article PubMed Google Scholar -
Ferramosca
A.,
Zara
V.,
Bioenergetics of mammalian sperm capacitation. BioMed Research International.
2014;
2014
:
902953
.
View Article PubMed Google Scholar -
Shamsi
M.B.,
Kumar
R.,
Bhatt
A.,
Bamezai
R.N.,
Kumar
R.,
Gupta
N.P.,
Mitochondrial DNA Mutations in etiopathogenesis of male infertility. Indian Journal of Urology.
2008;
24
(2)
:
150-4
.
View Article PubMed Google Scholar -
Ko
E.Y.,
Sabanegh
E.S.,
Agarwal
A.,
Male infertility testing: reactive oxygen species and antioxidant capacity. Fertility and Sterility.
2014;
102
(6)
:
1518-27
.
View Article PubMed Google Scholar -
Agarwal
A.,
Sharma
R.K.,
Desai
N.R.,
Prabakaran
S.,
Tavares
A.,
Sabanegh
E.,
Role of oxidative stress in pathogenesis of varicocele and infertility. Urology.
2009;
73
(3)
:
461-9
.
View Article PubMed Google Scholar -
Zini
A.,
San Gabriel
M.,
Baazeem
A.,
Antioxidants and sperm DNA damage: a clinical perspective. Journal of Assisted Reproduction and Genetics.
2009;
26
(8)
:
427-32
.
View Article PubMed Google Scholar -
Barbagallo
F.,
La Vignera
S.,
Cannarella
R.,
Aversa
A.,
Calogero
A.E.,
Condorelli
R.A.,
Evaluation of Sperm Mitochondrial Function: A Key Organelle for Sperm Motility. Journal of Clinical Medicine.
2020;
9
(2)
:
363
.
View Article PubMed Google Scholar -
Peña
F.J.,
Ortiz-Rodríguez
J.M.,
Gaitskell-Phillips
G.L.,
Gil
M.C.,
Ortega-Ferrusola
C.,
Martín-Cano
F.E.,
An integrated overview on the regulation of sperm metabolism (glycolysis-Krebs cycle-oxidative phosphorylation). Animal Reproduction Science.
2022;
246
:
106805
.
View Article PubMed Google Scholar -
Munro
D.,
Treberg
J.R.,
A radical shift in perspective: mitochondria as regulators of reactive oxygen species. The Journal of Experimental Biology.
2017;
220
(Pt 7)
:
1170-80
.
View Article PubMed Google Scholar -
Chernyak
B.V.,
Izyumov
D.S.,
Lyamzaev
K.G.,
Pashkovskaya
A.A.,
Pletjushkina
O.Y.,
Antonenko
Y.N.,
Production of reactive oxygen species in mitochondria of HeLa cells under oxidative stress. Biochimica et Biophysica Acta.
2006;
1757
(5-6)
:
525-34
.
View Article PubMed Google Scholar -
Aitken
R.J.,
Wingate
J.K.,
De Iuliis
G.N.,
McLaughlin
E.A.,
Analysis of lipid peroxidation in human spermatozoa using BODIPY C11. Molecular Human Reproduction.
2007;
13
(4)
:
203-11
.
View Article PubMed Google Scholar -
Koppers
A.J.,
De Iuliis
G.N.,
Finnie
J.M.,
McLaughlin
E.A.,
Aitken
R.J.,
Significance of mitochondrial reactive oxygen species in the generation of oxidative stress in spermatozoa. The Journal of Clinical Endocrinology and Metabolism.
2008;
93
(8)
:
3199-207
.
View Article PubMed Google Scholar -
Condorelli
R.A.,
La Vignera
S.,
Bellanca
S.,
Vicari
E.,
Calogero
A.E.,
Myoinositol: does it improve sperm mitochondrial function and sperm motility?. Urology.
2012;
79
(6)
:
1290-5
.
View Article PubMed Google Scholar -
Agnihotri
S.K.,
Agrawal
A.K.,
Hakim
B.A.,
Vishwakarma
A.L.,
Narender
T.,
Sachan
R.,
Mitochondrial membrane potential (MMP) regulates sperm motility. In Vitro Cellular & Developmental Biology. Animal.
2016;
52
(9)
:
953-60
.
View Article PubMed Google Scholar -
Wang
X.,
Sharma
R.K.,
Gupta
A.,
George
V.,
Thomas
A.J.,
Falcone
T.,
Alterations in mitochondria membrane potential and oxidative stress in infertile men: a prospective observational study. Fertility and Sterility.
2003;
80
:
844-50
.
View Article PubMed Google Scholar -
Zhao
R.Z.,
Jiang
S.,
Zhang
L.,
Yu
Z.B.,
Mitochondrial electron transport chain, ROS generation and uncoupling (Review). International Journal of Molecular Medicine.
2019;
44
(1)
:
3-15
.
View Article PubMed Google Scholar -
Oluwayiose
O.A.,
Houle
E.,
Whitcomb
B.W.,
Suvorov
A.,
Rahil
T.,
Sites
C.K.,
Altered non-coding RNA profiles of seminal plasma extracellular vesicles of men with poor semen quality undergoing in vitro fertilization treatment. Andrology.
2023;
11
(4)
:
677-86
.
View Article PubMed Google Scholar -
Amaral
A.,
Energy metabolism in mammalian sperm motility. WIREs Mechanisms of Disease.
2022;
14
(5)
:
e1569
.
View Article PubMed Google Scholar -
Castellini
C.,
D'Andrea
S.,
Cordeschi
G.,
Totaro
M.,
Parisi
A.,
Di Emidio
G.,
Pathophysiology of Mitochondrial Dysfunction in Human Spermatozoa: Focus on Energetic Metabolism, Oxidative Stress and Apoptosis. Antioxidants.
2021;
10
(5)
:
695
.
View Article PubMed Google Scholar -
Nascimento
J.M.,
Shi
L.Z.,
Tam
J.,
Chandsawangbhuwana
C.,
Durrant
B.,
Botvinick
E.L.,
Comparison of glycolysis and oxidative phosphorylation as energy sources for mammalian sperm motility, using the combination of fluorescence imaging, laser tweezers, and real-time automated tracking and trapping. Journal of Cellular Physiology.
2008;
217
(3)
:
745-51
.
View Article PubMed Google Scholar -
Hereng
T.H.,
Elgst∅en
K.B.,
Cederkvist
F.H.,
Eide
L.,
Jahnsen
T.,
Sk\aalhegg
B.S.,
Exogenous pyruvate accelerates glycolysis and promotes capacitation in human spermatozoa. Human Reproduction (Oxford, England).
2011;
26
(12)
:
3249-63
.
View Article PubMed Google Scholar -
Boguenet
M.,
Bouet
P.E.,
Spiers
A.,
Reynier
P.,
May-Panloup
P.,
Mitochondria: their role in spermatozoa and in male infertility. Human Reproduction Update.
2021;
27
(4)
:
697-719
.
View Article PubMed Google Scholar -
Davila
M. Plaza,
Muñoz
P. Martin,
Tapia
J.A.,
Ferrusola
C. Ortega,
C
C. Balao da Silva,
Peña
F.J.,
Inhibition of Mitochondrial Complex I Leads to Decreased Motility and Membrane Integrity Related to Increased Hydrogen Peroxide and Reduced ATP Production, while the Inhibition of Glycolysis Has Less Impact on Sperm Motility. PLoS One.
2015;
10
(9)
:
e0138777
.
View Article PubMed Google Scholar -
Nowicka-Bauer
K.,
Nixon
B.,
Molecular Changes Induced by Oxidative Stress that Impair Human Sperm Motility. Antioxidants.
2020;
9
(2)
:
134
.
View Article PubMed Google Scholar -
Serafini
S.,
O'Flaherty
C.,
Redox Regulation to Modulate Phosphorylation Events in Human Spermatozoa. Antioxidants & Redox Signaling.
2022;
37
(7-9)
:
437-50
.
View Article PubMed Google Scholar -
Amorini
A.M.,
Listorti
I.,
Bilotta
G.,
Pallisco
R.,
Saab
M.W.,
Mangione
R.,
Antioxidant-Based Therapies in Male Infertility: Do We Have Sufficient Evidence Supporting Their Effectiveness?. Antioxidants.
2021;
10
(2)
:
220
.
View Article PubMed Google Scholar -
Nitti
M.,
Marengo
B.,
Furfaro
A.L.,
Pronzato
M.A.,
Marinari
U.M.,
Domenicotti
C.,
Hormesis and Oxidative Distress: Pathophysiology of Reactive Oxygen Species and the Open Question of Antioxidant Modulation and Supplementation. Antioxidants.
2022;
11
(8)
:
1613
.
View Article PubMed Google Scholar -
Sies
H.,
Belousov
V.V.,
Chandel
N.S.,
Davies
M.J.,
Jones
D.P.,
Mann
G.E.,
Defining roles of specific reactive oxygen species (ROS) in cell biology and physiology. Nature Reviews. Molecular Cell Biology.
2022;
23
(7)
:
499-515
.
View Article PubMed Google Scholar -
Miller
A.F.,
Superoxide dismutases: ancient enzymes and new insights. FEBS Letters.
2012;
586
(5)
:
585-95
.
View Article PubMed Google Scholar -
Fatima
S. Kaneez,
Kanti
D. Tushar,
Introductory Chapter: Role of Fenton and Haber-Weiss Reaction in Epilepsy, in Epilepsy, S. Kaneez Fatima, Editor. 2023, IntechOpen: Rijeka. p. Ch. 1.. 2023
.
-
Haber–Weiss reaction' in IUPAC Compendium of Chemical Terminology, 3rd ed. International Union of Pure and Applied Chemistry; 2006. Online version 3.0.1, 2019 . 2019
.
View Article Google Scholar -
Latham
K.E.,
Endoplasmic reticulum stress signaling in mammalian oocytes and embryos: life in balance. International Review of Cell and Molecular Biology.
2015;
316
:
227-65
.
View Article PubMed Google Scholar -
Dikalov
S.,
Cross talk between mitochondria and NADPH oxidases. Free Radical Biology & Medicine.
2011;
51
(7)
:
1289-301
.
View Article PubMed Google Scholar -
Sies
H.,
Oxidative eustress and oxidative distress: Introductory remarks. InOxidative stress 2020 Jan 1 (pp. 3-12). Academic Press .
View Article Google Scholar -
Soto
I.,
Couvillion
M.,
Hansen
K.G.,
McShane
E.,
Moran
J.C.,
Barrientos
A.,
Balanced mitochondrial and cytosolic translatomes underlie the biogenesis of human respiratory complexes. Genome Biology.
2022;
23
(1)
:
170-170
.
View Article PubMed Google Scholar -
Aitken
R.J.,
The Amoroso Lecture. The human spermatozoon - a cell in crisis?. Journal of Reproduction and Fertility.
1999;
115
(1)
:
1-7
.
View Article PubMed Google Scholar -
Wyck
S.,
Herrera
C.,
Requena
C.E.,
Bittner
L.,
Hajkova
P.,
Bollwein
H.,
Oxidative stress in sperm affects the epigenetic reprogramming in early embryonic development. Epigenetics & Chromatin.
2018;
11
(1)
:
60
.
View Article PubMed Google Scholar -
McWilliams
T.G.,
Suomalainen
A.,
Mitochondrial DNA can be inherited from fathers, not just mothers. Nature.
2019;
565
(7739)
:
296-7
.
View Article PubMed Google Scholar -
Fernandez-Vizarra
E.,
Zeviani
M.,
Mitochondrial disorders of the OXPHOS system. FEBS Letters.
2021;
595
(8)
:
1062-106
.
View Article PubMed Google Scholar -
Bui
A.D.,
Sharma
R.,
Henkel
R.,
Agarwal
A.,
Reactive oxygen species impact on sperm DNA and its role in male infertility. Andrologia.
2018;
50
(8)
:
e13012
.
View Article PubMed Google Scholar -
Dutta
S.,
Physiological role of ROS in sperm function. Male infertility: Contemporary clinical approaches, Andrology, ART and antioxidants.
2020;
:
337-45
.
View Article Google Scholar -
Aitken
R.J.,
Wingate
J.K.,
De Iuliis
G.N.,
Koppers
A.J.,
McLaughlin
E.A.,
Cis-unsaturated fatty acids stimulate reactive oxygen species generation and lipid peroxidation in human spermatozoa. The Journal of Clinical Endocrinology and Metabolism.
2006;
91
(10)
:
4154-63
.
View Article PubMed Google Scholar -
Garrido
C.,
Galluzzi
L.,
Brunet
M.,
Puig
P.E.,
Didelot
C.,
Kroemer
G.,
Mechanisms of cytochrome c release from mitochondria. Cell Death and Differentiation.
2006;
13
(9)
:
1423-33
.
View Article PubMed Google Scholar -
Esteves
S.C.,
Santi
D.,
Simoni
M.,
An update on clinical and surgical interventions to reduce sperm DNA fragmentation in infertile men. Andrology.
2020;
8
(1)
:
53-81
.
View Article PubMed Google Scholar -
Cummins
J.M.,
Mitochondria: potential roles in embryogenesis and nucleocytoplasmic transfer. Human Reproduction Update.
2001;
7
(2)
:
217-28
.
View Article PubMed Google Scholar -
Passos
J.F.,
Saretzki
G.,
Ahmed
S.,
Nelson
G.,
Richter
T.,
Peters
H.,
Mitochondrial dysfunction accounts for the stochastic heterogeneity in telomere-dependent senescence. PLoS Biology.
2007;
5
(5)
:
e110
.
View Article PubMed Google Scholar -
Sun
N.,
Youle
R.J.,
Finkel
T.,
The Mitochondrial Basis of Aging. Molecular Cell.
2016;
61
(5)
:
654-66
.
View Article PubMed Google Scholar -
Chiang
J.L.,
Shukla
P.,
Pagidas
K.,
Ahmed
N.S.,
Karri
S.,
Gunn
D.D.,
Mitochondria in Ovarian Aging and Reproductive Longevity. Ageing Research Reviews.
2020;
63
:
101168
.
View Article PubMed Google Scholar -
Yang
L.,
Chen
Y.,
Liu
Y.,
Xing
Y.,
Miao
C.,
Zhao
Y.,
The Role of Oxidative Stress and Natural Antioxidants in Ovarian Aging. Frontiers in Pharmacology.
2021;
11
:
617843
.
View Article PubMed Google Scholar -
Tsirka
G.,
Zikopoulos
A.,
Papageorgiou
K.,
Kostoulas
C.,
Tsigkas
I.,
Moustakli
E.,
The Ratio of cf-mtDNA vs. cf-nDNA in the Follicular Fluid of Women Undergoing IVF Is Positively Correlated with Age. Genes.
2023;
14
(7)
:
1504
.
View Article PubMed Google Scholar -
Hance
N.,
Ekstrand
M.I.,
Trifunovic
A.,
Mitochondrial DNA polymerase gamma is essential for mammalian embryogenesis. Human Molecular Genetics.
2005;
14
(13)
:
1775-83
.
View Article PubMed Google Scholar -
Tang
S.,
Wang
J.,
Lee
N.C.,
Milone
M.,
Halberg
M.C.,
Schmitt
E.S.,
Mitochondrial DNA polymerase γ mutations: an ever expanding molecular and clinical spectrum. Journal of Medical Genetics.
2011;
48
(10)
:
669-81
.
View Article PubMed Google Scholar -
Gurgel-Giannetti
J.,
Camargos
S.T.,
Cardoso
F.,
Hirano
M.,
DiMauro
S.,
POLG1 Arg953Cys mutation: expanded phenotype and recessive inheritance in a Brazilian family. Muscle {&}amp; Nerve.
2012;
45
(3)
:
453-4
.
View Article PubMed Google Scholar -
Luoma
P.,
Melberg
A.,
Rinne
J.O.,
Kaukonen
J.A.,
Nupponen
N.N.,
Chalmers
R.M.,
Parkinsonism, premature menopause, and mitochondrial DNA polymerase gamma mutations: clinical and molecular genetic study. Lancet.
2004;
364
(9437)
:
875-82
.
View Article PubMed Google Scholar -
Pagnamenta
A.T.,
Taanman
J.W.,
Wilson
C.J.,
Anderson
N.E.,
Marotta
R.,
Duncan
A.J.,
Dominant inheritance of premature ovarian failure associated with mutant mitochondrial DNA polymerase gamma. Human Reproduction (Oxford, England).
2006;
21
(10)
:
2467-73
.
View Article PubMed Google Scholar -
Bekheirnia
M.R.,
Zhang
W.,
Eble
T.,
Willis
A.,
Shaibani
A.,
Wong
L.J.,
POLG mutation in a patient with cataracts, early-onset distal muscle weakness and atrophy, ovarian dysgenesis and 3-methylglutaconic aciduria. Gene.
2012;
499
(1)
:
209-12
.
View Article PubMed Google Scholar -
Yang
L.,
Lin
X.,
Tang
H.,
Fan
Y.,
Zeng
S.,
Jia
L.,
Mitochondrial DNA mutation exacerbates female reproductive aging via impairment of the NADH/NAD+ redox. Aging Cell.
2020;
19
(9)
:
e13206
.
View Article PubMed Google Scholar -
Babayev
E.,
Seli
E.,
Oocyte mitochondrial function and reproduction. Current Opinion in Obstetrics & Gynecology.
2015;
27
(3)
:
175-81
.
View Article PubMed Google Scholar -
Rodríguez-Nuevo
A.,
Torres-Sanchez
A.,
Duran
J.M.,
De Guirior
C.,
Martínez-Zamora
M.A.,
Böke
E.,
Oocytes maintain ROS-free mitochondrial metabolism by suppressing complex I. Nature.
2022;
607
(7920)
:
756-61
.
View Article PubMed Google Scholar -
Liu
L.,
Blasco
M.A.,
Keefe
D.L.,
Requirement of functional telomeres for metaphase chromosome alignments and integrity of meiotic spindles. EMBO Reports.
2002;
3
(3)
:
230-4
.
View Article PubMed Google Scholar -
Liu
L.,
Franco
S.,
Spyropoulos
B.,
Moens
P.B.,
Blasco
M.A.,
Keefe
D.L.,
Irregular telomeres impair meiotic synapsis and recombination in mice. Proceedings of the National Academy of Sciences of the United States of America.
2004;
101
(17)
:
6496-501
.
View Article PubMed Google Scholar -
Liu
L.,
Trimarchi
J.R.,
Smith
P.J.,
Keefe
D.L.,
Mitochondrial dysfunction leads to telomere attrition and genomic instability. Aging Cell.
2002;
1
(1)
:
40-6
.
View Article PubMed Google Scholar -
Zhang
L.Y.,
Lin
M.,
Qingrui
Z.,
Zichuan
W.,
Junjin
L.,
Kexiong
L.,
Mitochondrial Calcium uniporters are essential for meiotic progression in mouse oocytes by controlling Ca2+ entry. Cell Proliferation.
2021;
54
(11)
:
e13127
.
View Article PubMed Google Scholar -
Zhang
C.,
Gao
L.,
Wu
D.,
Wang
G.,
Lan
H.,
Li
L.,
IP3R1 regulates calcium balance in porcine oocyte maturation and early embryonic development. Theriogenology.
2023;
209
:
151-61
.
View Article PubMed Google Scholar -
Wang
F.,
Li
A.,
Meng
T.G.,
Wang
L.Y.,
Wang
L.J.,
Hou
Y.,
Regulation of [Ca2+]i oscillations and mitochondrial activity by various calcium transporters in mouse oocytes. Reproductive Biology and Endocrinology.
2020;
18
(1)
:
87
.
View Article PubMed Google Scholar -
Kala
M.,
Shaikh
M.V.,
Nivsarkar
M.,
Equilibrium between anti-oxidants and reactive oxygen species: a requisite for oocyte development and maturation. Reproductive Medicine and Biology.
2016;
16
(1)
:
28-35
.
View Article PubMed Google Scholar -
Zhang
M.,
Lu
Y.,
Chen
Y.,
Zhang
Y.,
Xiong
B.,
Insufficiency of melatonin in follicular fluid is a reversible cause for advanced maternal age-related aneuploidy in oocytes. Redox Biology.
2020;
28
:
101327
.
View Article PubMed Google Scholar -
Lim
J.,
Luderer
U.,
Oxidative damage increases and antioxidant gene expression decreases with aging in the mouse ovary. Biology of Reproduction.
2011;
84
(4)
:
775-82
.
View Article PubMed Google Scholar -
Jiao
L.,
Hu
C.X.,
Zhang
Y.,
Zhang
Y.X.,
Cai
W.W.,
Pan
W.L.,
SIRT3 Regulates Levels of Deacetylated SOD2 to Prevent Oxidative Stress and Mitochondrial Dysfunction During Oocyte Maturation in Pigs. Microscopy and Microanalysis.
2023;
29
(6)
:
2149-60
.
View Article PubMed Google Scholar -
Qi
X.Y.,
Yuan
J.D.,
Liu
Z.Y.,
Jiang
X.Q.,
Zhang
Q.,
Zhang
S.L.,
Sirtuin 3-mediated deacetylation of superoxide dismutase 2 ameliorates sodium fluoride-induced mitochondrial dysfunction in porcine oocytes. The Science of the Total Environment.
2024;
908
:
168306
.
View Article PubMed Google Scholar -
Harasimov
K.,
Gorry
R.L.,
Welp
L.M.,
Penir
S.M.,
Horokhovskyi
Y.,
Cheng
S.,
The maintenance of oocytes in the mammalian ovary involves extreme protein longevity. Nature Cell Biology.
2024;
26
(7)
:
1124-38
.
View Article PubMed Google Scholar -
Gonzalez
B.,
Tare
A.,
Ryu
S.,
Johnson
S.C.,
Atzmon
G.,
Barzilai
N.,
High-throughput sequencing analysis of nuclear-encoded mitochondrial genes reveals a genetic signature of human longevity. GeroScience.
2023;
45
(1)
:
311-30
.
View Article PubMed Google Scholar -
Vögtle
F.N.,
Open questions on the mitochondrial unfolded protein response. The FEBS Journal.
2021;
288
(9)
:
2856-69
.
View Article PubMed Google Scholar -
Mecha
M.F.,
Hutchinson
R.B.,
Lee
J.H.,
Cavagnero
S.,
Protein folding in vitro and in the cell: from a solitary journey to a team effort. Biophysical Chemistry.
2022;
287
:
106821
.
View Article PubMed Google Scholar -
Arnould
T.,
Michel
S.,
Renard
P.,
Mitochondria Retrograde Signaling and the UPR mt: Where Are We in Mammals?. International Journal of Molecular Sciences.
2015;
16
(8)
:
18224-51
.
View Article PubMed Google Scholar -
Gregersen
N.,
Bross
P.,
Protein misfolding and cellular stress: an overview. Methods in Molecular Biology (Clifton, N.J.).
2010;
648
:
3-23
.
View Article PubMed Google Scholar -
Kriegenburg
F.,
Ellgaard
L.,
Hartmann-Petersen
R.,
Molecular chaperones in targeting misfolded proteins for ubiquitin-dependent degradation. The FEBS Journal.
2012;
279
(4)
:
532-42
.
View Article PubMed Google Scholar -
Baker
M.J.,
Tatsuta
T.,
Langer
T.,
Quality control of mitochondrial proteostasis. Cold Spring Harbor Perspectives in Biology.
2011;
3
(7)
:
a007559
.
View Article PubMed Google Scholar -
Hamon
M.P.,
Bulteau
A.L.,
Friguet
B.,
Mitochondrial proteases and protein quality control in ageing and longevity. Ageing research reviews.
2015;
23
:
56-66
.
View Article Google Scholar -
Li
G.,
Gu
J.,
Zhou
X.,
Wu
T.,
Li
X.,
Hua
R.,
Mitochondrial stress response gene Clpp deficiency impairs oocyte competence and deteriorate cyclophosphamide-induced ovarian damage in young mice. Frontiers in Endocrinology (Lausanne).
2023;
14
:
1122012
.
View Article PubMed Google Scholar -
Sanchez
T.,
Wang
T.,
Pedro
M.V.,
Zhang
M.,
Esencan
E.,
Sakkas
D.,
Metabolic imaging with the use of fluorescence lifetime imaging microscopy (FLIM) accurately detects mitochondrial dysfunction in mouse oocytes. Fertility and Sterility.
2018;
110
(7)
:
1387-97
.
View Article PubMed Google Scholar -
Cozzolino
M.,
Disruption of Mitochondrial Unfolded Protein Response Results in Telomere Shortening in Mouse Oocytes and Somatic Cells. Aging (Albany NY).
2023;
16
(3)
:
2047
.
View Article Google Scholar -
Hennig
M.,
Fiedler
S.,
Jux
C.,
Thierfelder
L.,
Drenckhahn
J.D.,
Prenatal Mechanistic Target of Rapamycin Complex 1 (m TORC1) Inhibition by Rapamycin Treatment of Pregnant Mice Causes Intrauterine Growth Restriction and Alters Postnatal Cardiac Growth, Morphology, and Function. Journal of the American Heart Association.
2017;
6
(8)
:
e005506
.
View Article PubMed Google Scholar -
Dumollard
R.,
Duchen
M.,
Carroll
J.,
The role of mitochondrial function in the oocyte and embryo. Current Topics in Developmental Biology.
2007;
77
:
21-49
.
View Article PubMed Google Scholar -
Harvey
A.J.,
Mitochondria in early development: linking the microenvironment, metabolism and the epigenome. Reproduction (Cambridge, England).
2019;
157
(5)
:
159-79
.
View Article PubMed Google Scholar -
Xu
W.,
Zhu
H.,
Gu
M.,
Luo
Q.,
Ding
J.,
Yao
Y.,
DHTKD1 is essential for mitochondrial biogenesis and function maintenance. FEBS Letters.
2013;
587
(21)
:
3587-92
.
View Article PubMed Google Scholar -
Del Bianco
D.,
Gentile
R.,
Sallicandro
L.,
Biagini
A.,
Quellari
P.T.,
Gliozheni
E.,
Electro-Metabolic Coupling of Cumulus-Oocyte Complex. International Journal of Molecular Sciences.
2024;
25
(10)
:
5349
.
View Article PubMed Google Scholar -
Dalton
C.M.,
Szabadkai
G.,
Carroll
J.,
Measurement of ATP in single oocytes: impact of maturation and cumulus cells on levels and consumption. Journal of Cellular Physiology.
2014;
229
(3)
:
353-61
.
View Article PubMed Google Scholar -
Ma
K.,
Chen
G.,
Li
W.,
Kepp
O.,
Zhu
Y.,
Chen
Q.,
Mitophagy, Mitochondrial Homeostasis, and Cell Fate. Frontiers in Cell and Developmental Biology.
2020;
8
:
467
.
View Article PubMed Google Scholar -
Gottlieb
R.A.,
Bernstein
D.,
Mitochondrial remodeling: Rearranging, recycling, and reprogramming. Cell Calcium.
2016;
60
(2)
:
88-101
.
View Article PubMed Google Scholar -
Hao
X.,
Zhao
J.,
Rodriguez-Wallberg
K.A.,
Comprehensive atlas of mitochondrial distribution and dynamics during oocyte maturation in mouse models. Biomarker Research.
2024;
12
(1)
:
125
.
View Article PubMed Google Scholar -
Yu
Y.,
Dumollard
R.,
Rossbach
A.,
Lai
F.A.,
Swann
K.,
Redistribution of mitochondria leads to bursts of ATP production during spontaneous mouse oocyte maturation. Journal of Cellular Physiology.
2010;
224
(3)
:
672-80
.
View Article PubMed Google Scholar -
Zhang
X.,
Wu
X.Q.,
Lu
S.,
Guo
Y.L.,
Ma
X.,
Deficit of mitochondria-derived ATP during oxidative stress impairs mouse MII oocyte spindles. Cell Research.
2006;
16
(10)
:
841-50
.
View Article PubMed Google Scholar -
Kankanam Gamage
U.S.,
Hashimoto
S.,
Miyamoto
Y.,
Nakano
T.,
Yamanaka
M.,
Koike
A.,
Mitochondria Transfer from Adipose Stem Cells Improves the Developmental Potential of Cryopreserved Oocytes. Biomolecules.
2022;
12
(7)
:
1008
.
View Article PubMed Google Scholar -
Seo
A.Y.,
Joseph
A.M.,
Dutta
D.,
Hwang
J.C.,
Aris
J.P.,
Leeuwenburgh
C.,
New insights into the role of mitochondria in aging: mitochondrial dynamics and more. Journal of Cell Science.
2010;
123
(Pt 15)
:
2533-42
.
View Article PubMed Google Scholar -
Tilokani
L.,
Nagashima
S.,
Paupe
V.,
Prudent
J.,
Mitochondrial dynamics: overview of molecular mechanisms. Essays in Biochemistry.
2018;
62
(3)
:
341-60
.
View Article PubMed Google Scholar -
Ni
H.M.,
Williams
J.A.,
Ding
W.X.,
Mitochondrial dynamics and mitochondrial quality control. Redox Biology.
2015;
4
:
6-13
.
View Article PubMed Google Scholar -
Kornfeld
O.S.,
Qvit
N.,
Haileselassie
B.,
Shamloo
M.,
Bernardi
P.,
Mochly-Rosen
D.,
Interaction of mitochondrial fission factor with dynamin related protein 1 governs physiological mitochondrial function in vivo. Scientific Reports.
2018;
8
(1)
:
14034
.
View Article PubMed Google Scholar -
Osellame
L.D.,
Blacker
T.S.,
Duchen
M.R.,
Cellular and molecular mechanisms of mitochondrial function. Best Practice & Research. Clinical Endocrinology & Metabolism.
2012;
26
(6)
:
711-23
.
View Article PubMed Google Scholar -
Zou
W.,
Ji
D.,
Zhang
Z.,
Yang
L.,
Cao
Y.,
Players in Mitochondrial Dynamics and Female Reproduction. Frontiers in Molecular Biosciences.
2021;
8
:
717328
.
View Article PubMed Google Scholar -
Udagawa
O.,
Ishihara
T.,
Maeda
M.,
Matsunaga
Y.,
Tsukamoto
S.,
Kawano
N.,
Mitochondrial fission factor Drp1 maintains oocyte quality via dynamic rearrangement of multiple organelles. Current Biology.
2014;
24
(20)
:
2451-8
.
View Article PubMed Google Scholar -
Westermann
B.,
Mitochondrial fusion and fission in cell life and death. Nature Reviews. Molecular Cell Biology.
2010;
11
(12)
:
872-84
.
View Article PubMed Google Scholar -
Zhang
M.,
Bener
M.B.,
Jiang
Z.,
Wang
T.,
Esencan
E.,
Iii
R. Scott,
Mitofusin 1 is required for female fertility and to maintain ovarian follicular reserve. Cell Death & Disease.
2019;
10
(8)
:
560
.
View Article PubMed Google Scholar -
Zhang
M.,
Bener
M.B.,
Jiang
Z.,
Wang
T.,
Esencan
E.,
Scott
R.,
Mitofusin 2 plays a role in oocyte and follicle development, and is required to maintain ovarian follicular reserve during reproductive aging. Aging (Albany NY).
2019;
11
(12)
:
3919-38
.
View Article PubMed Google Scholar -
Toupance
S.,
Fattet
A.J.,
Thornton
S.N.,
Benetos
A.,
Guéant
J.L.,
Koscinski
I.,
Ovarian Telomerase and Female Fertility. Biomedicines.
2021;
9
(7)
:
842
.
View Article PubMed Google Scholar -
Ankel-Simons
F.,
Cummins
J.M.,
Misconceptions about mitochondria and mammalian fertilization: implications for theories on human evolution. Proceedings of the National Academy of Sciences of the United States of America.
1996;
93
(24)
:
13859-63
.
View Article PubMed Google Scholar -
Cummins
J.M.,
Wakayama
T.,
Yanagimachi
R.,
Fate of microinjected spermatid mitochondria in the mouse oocyte and embryo. Zygote (Cambridge, England).
1998;
6
(3)
:
213-22
.
View Article PubMed Google Scholar -
Molina
P.,
Lim
Y.,
Boyd
L.,
Ubiquitination is required for the initial removal of paternal organelles in C. elegans. Developmental Biology.
2019;
453
(2)
:
168-79
.
View Article PubMed Google Scholar -
Luo
S.,
Valencia
C.A.,
Zhang
J.,
Lee
N.C.,
Slone
J.,
Gui
B.,
Biparental Inheritance of Mitochondrial DNA in Humans. Proceedings of the National Academy of Sciences of the United States of America.
2018;
115
(51)
:
13039-44
.
View Article PubMed Google Scholar -
St John
J.C.,
Okada
T.,
Andreas
E.,
Penn
A.,
The role of mtDNA in oocyte quality and embryo development. Molecular Reproduction and Development.
2023;
90
(7)
:
621-33
.
View Article PubMed Google Scholar -
Bradley
J.,
Swann
K.,
Mitochondrial functionality in reproduction: from gonads and gametes to embryos and embryonic stem cells. Human reproduction update.
2009;
15
(5)
:
553-72
.
View Article Google Scholar -
Ramalho-Santos
J.,
Varum
S.,
Amaral
S.,
Mota
P.C.,
Sousa
A.P.,
Amaral
A.,
Mitochondrial functionality in reproduction: from gonads and gametes to embryos and embryonic stem cells. Human Reproduction Update.
2009;
15
(5)
:
553-72
.
View Article PubMed Google Scholar -
Bailey
P.S.,
Nathan
J.A.,
Metabolic Regulation of Hypoxia-Inducible Transcription Factors: The Role of Small Molecule Metabolites and Iron. Biomedicines.
2018;
6
(2)
:
60
.
View Article PubMed Google Scholar -
Hashimoto
S.,
Morimoto
Y.,
Mitochondrial function of human embryo: decline in their quality with maternal aging. Reproductive Medicine and Biology.
2022;
21
(1)
:
e12491
.
View Article PubMed Google Scholar -
Zhang
W.,
Wu
F.,
Effects of adverse fertility-related factors on mitochondrial DNA in the oocyte: a comprehensive review. Reproductive Biology and Endocrinology.
2023;
21
(1)
:
27
.
View Article PubMed Google Scholar -
Dumollard
R.,
Duchen
M.,
Sardet
C.,
Calcium signals and mitochondria at fertilisation. Seminars in Cell & Developmental Biology.
2006;
17
(2)
:
314-23
.
View Article PubMed Google Scholar -
Liu
L.,
Hammar
K.,
Smith
P.J.,
Inoue
S.,
Keefe
D.L.,
Mitochondrial modulation of calcium signaling at the initiation of development. Cell Calcium.
2001;
30
(6)
:
423-33
.
View Article PubMed Google Scholar -
Eichenlaub-Ritter
U.,
Vogt
E.,
Yin
H.,
Gosden
R.,
Spindles, mitochondria and redox potential in ageing oocytes. Reproductive Biomedicine Online.
2004;
8
(1)
:
45-58
.
View Article PubMed Google Scholar -
Allio
R.,
Donega
S.,
Galtier
N.,
Nabholz
B.,
Large Variation in the Ratio of Mitochondrial to Nuclear Mutation Rate across Animals: Implications for Genetic Diversity and the Use of Mitochondrial DNA as a Molecular Marker. Molecular Biology and Evolution.
2017;
34
(11)
:
2762-72
.
View Article PubMed Google Scholar -
Nie
X.,
Dong
X.,
Hu
Y.,
Xu
F.,
Hu
C.,
Shu
C.,
Coenzyme Q10 Stimulate Reproductive Vatality. Drug Design, Development and Therapy.
2023;
17
:
2623-37
.
View Article PubMed Google Scholar -
Van Blerkom
J.,
Davis
P.,
Alexander
S.,
Differential mitochondrial distribution in human pronuclear embryos leads to disproportionate inheritance between blastomeres: relationship to microtubular organization, ATP content and competence. Human Reproduction (Oxford, England).
2000;
15
(12)
:
2621-33
.
View Article PubMed Google Scholar -
Chatzovoulou
K.,
Mayeur
A.,
Gigarel
N.,
Jabot-Hanin
F.,
Hesters
L.,
Munnich
A.,
Mitochondrial DNA mutations do not impact early human embryonic development. Mitochondrion.
2021;
58
:
59-63
.
View Article PubMed Google Scholar -
Miyamoto
K.,
Sato
E.F.,
Kasahara
E.,
Jikumaru
M.,
Hiramoto
K.,
Tabata
H.,
Effect of oxidative stress during repeated ovulation on the structure and functions of the ovary, oocytes, and their mitochondria. Free Radical Biology & Medicine.
2010;
49
(4)
:
674-81
.
View Article PubMed Google Scholar -
Gibson
T.C.,
Kubisch
H.M.,
Brenner
C.A.,
Mitochondrial DNA deletions in rhesus macaque oocytes and embryos. Molecular Human Reproduction.
2005;
11
(11)
:
785-9
.
View Article PubMed Google Scholar -
Lee
S.T.,
Oh
S.J.,
Lee
E.J.,
Han
H.J.,
Lim
J.M.,
Adenosine triphosphate synthesis, mitochondrial number and activity, and pyruvate uptake in oocytes after gonadotropin injections. Fertility and Sterility.
2006;
86
(4)
:
1164-9
.
View Article PubMed Google Scholar -
Lopes
A.S.,
Lane
M.,
Thompson
J.G.,
Oxygen consumption and ROS production are increased at the time of fertilization and cell cleavage in bovine zygotes. Human Reproduction (Oxford, England).
2010;
25
(11)
:
2762-73
.
View Article PubMed Google Scholar -
Vandaele
L.,
Thys
M.,
Bijttebier
J.,
Van Langendonckt
A.,
Donnay
I.,
Maes
D.,
Short-term exposure to hydrogen peroxide during oocyte maturation improves bovine embryo development. Reproduction (Cambridge, England).
2010;
139
(3)
:
505-11
.
View Article PubMed Google Scholar -
Dumollard
R.,
Campbell
K.,
Halet
G.,
Carroll
J.,
Swann
K.,
Regulation of cytosolic and mitochondrial ATP levels in mouse eggs and zygotes. Developmental Biology.
2008;
316
(2)
:
431-40
.
View Article PubMed Google Scholar -
Cirilli
I.,
Damiani
E.,
Dludla
P.V.,
Hargreaves
I.,
Marcheggiani
F.,
Millichap
L.E.,
Role of Coenzyme Q10 in Health and Disease: An Update on the Last 10 Years (2010-2020). Antioxidants.
2021;
10
(8)
:
1325
.
View Article PubMed Google Scholar -
Salvio
G.,
Cutini
M.,
Ciarloni
A.,
Giovannini
L.,
Perrone
M.,
Balercia
G.,
Coenzyme Q10 and Male Infertility: A Systematic Review. Antioxidants.
2021;
10
(6)
:
874
.
View Article PubMed Google Scholar -
Florou
P.,
Anagnostis
P.,
Theocharis
P.,
Chourdakis
M.,
Goulis
D.G.,
Does coenzyme Q10 supplementation improve fertility outcomes in women undergoing assisted reproductive technology procedures? A systematic review and meta-analysis of randomized-controlled trials. Journal of Assisted Reproduction and Genetics.
2020;
37
(10)
:
2377-87
.
View Article PubMed Google Scholar -
Ben-Meir
A.,
Burstein
E.,
Borrego-Alvarez
A.,
Chong
J.,
Wong
E.,
Yavorska
T.,
Coenzyme Q10 restores oocyte mitochondrial function and fertility during reproductive aging. Aging Cell.
2015;
14
(5)
:
887-95
.
View Article PubMed Google Scholar -
Marei
W.F.,
Mohey-Elsaeed
O.,
Pintelon
I.,
Leroy
J.L.,
Risks of using mitoquinone during in vitro maturation and its potential protective effects against lipotoxicity-induced oocyte mitochondrial stress. Journal of Assisted Reproduction and Genetics.
2024;
41
(2)
:
371-83
.
View Article PubMed Google Scholar -
Zare
Z.,
Masteri Farahani
R.,
Salehi
M.,
Piryaei
A.,
Ghaffari Novin
M.,
Fadaei Fathabadi
F.,
Effect of L-carnitine supplementation on maturation and early embryo development of immature mouse oocytes selected by brilliant cresyle blue staining. Journal of Assisted Reproduction and Genetics.
2015;
32
(4)
:
635-43
.
View Article PubMed Google Scholar -
Agarwal
A.,
Sengupta
P.,
Durairajanayagam
D.,
Role of L-carnitine in female infertility. Reproductive Biology and Endocrinology.
2018;
16
(1)
:
5
.
View Article PubMed Google Scholar -
Jegal
H.G.,
Park
H.J.,
Kim
J.W.,
Yang
S.G.,
Kim
M.J.,
Koo
D.B.,
Ruthenium red improves blastocyst developmental competence by regulating mitochondrial Ca2+ and mitochondrial functions in fertilized porcine oocytes in vitro. The Journal of Reproduction and Development.
2020;
66
(4)
:
377-86
.
View Article PubMed Google Scholar -
Parikh
S.,
Goldstein
A.,
Koenig
M.K.,
Scaglia
F.,
Enns
G.M.,
Saneto
R.,
Diagnosis and management of mitochondrial disease: a consensus statement from the Mitochondrial Medicine Society. Genetics in Medicine.
2015;
17
(9)
:
689-701
.
View Article PubMed Google Scholar -
Sue
C.M.,
Balasubramaniam
S.,
Bratkovic
D.,
Bonifant
C.,
Christodoulou
J.,
Coman
D.,
Patient care standards for primary mitochondrial disease in Australia: an Australian adaptation of the Mitochondrial Medicine Society recommendations. Internal Medicine Journal.
2022;
52
(1)
:
110-20
.
View Article PubMed Google Scholar -
McCormick
E.M.,
Muraresku
C.C.,
Falk
M.J.,
Mitochondrial Genomics: A complex field now coming of age. Current Genetic Medicine Reports.
2018;
6
(2)
:
52-61
.
View Article PubMed Google Scholar -
Kivisild
T.,
Maternal ancestry and population history from whole mitochondrial genomes. Investigative Genetics.
2015;
6
(1)
:
3
.
View Article PubMed Google Scholar -
Farnezi
H.C.,
Goulart
A.C.,
Santos
A.D.,
Ramos
M.G.,
Penna
M.L.,
Three-parent babies: mitochondrial replacement therapies. JBRA Assisted Reproduction.
2020;
24
(2)
:
189-96
.
PubMed Google Scholar -
Tachibana
M.,
Kuno
T.,
Yaegashi
N.,
Mitochondrial replacement therapy and assisted reproductive technology: A paradigm shift toward treatment of genetic diseases in gametes or in early embryos. Reproductive Medicine and Biology.
2018;
17
(4)
:
421-33
.
View Article PubMed Google Scholar -
Klopstock
T.,
Klopstock
B.,
Prokisch
H.,
Mitochondrial replacement approaches: challenges for clinical implementation. Genome Medicine.
2016;
8
(1)
:
126
.
View Article PubMed Google Scholar -
Appleby
J.B.,
The ethical challenges of the clinical introduction of mitochondrial replacement techniques. Medicine, Health Care, and Philosophy.
2015;
18
(4)
:
501-14
.
View Article PubMed Google Scholar -
Cohen
I.G.,
Adashi
E.Y.,
Gerke
S.,
Palacios-González
C.,
Ravitsky
V.,
The Regulation of Mitochondrial Replacement Techniques Around the World. Annual Review of Genomics and Human Genetics.
2020;
21
(1)
:
565-86
.
View Article PubMed Google Scholar -
Knoppers
B.M.,
Leader
A.,
Hume
S.,
Shoubridge
E.A.,
Isasi
R.,
Noohi
F.,
Mitochondrial Replacement Therapy: The Road to the Clinic in Canada. Journal of Obstetrics and Gynaecology Canada.
2017;
39
(10)
:
916-8
.
View Article PubMed Google Scholar -
Herbert
M.,
Turnbull
D.,
Mitochondrial Donation - Clearing the Final Regulatory Hurdle in the United Kingdom. The New England Journal of Medicine.
2017;
376
(2)
:
171-3
.
View Article PubMed Google Scholar -
Gomzikova
M.O.,
James
V.,
Rizvanov
A.A.,
Mitochondria Donation by Mesenchymal Stem Cells: Current Understanding and Mitochondria Transplantation Strategies. Frontiers in Cell and Developmental Biology.
2021;
9
:
653322
.
View Article PubMed Google Scholar -
Wang
Z.H.,
Chen
L.,
Li
W.,
Chen
L.,
Wang
Y.P.,
Mitochondria transfer and transplantation in human health and diseases. Mitochondrion.
2022;
65
:
80-7
.
View Article PubMed Google Scholar -
Liu
D.,
Gao
Y.,
Liu
J.,
Huang
Y.,
Yin
J.,
Feng
Y.,
Intercellular mitochondrial transfer as a means of tissue revitalization. Signal Transduction and Targeted Therapy.
2021;
6
(1)
:
65
.
View Article PubMed Google Scholar -
Wang
Z.B.,
Hao
J.X.,
Meng
T.G.,
Guo
L.,
Dong
M.Z.,
Fan
L.H.,
Transfer of autologous mitochondria from adipose tissue-derived stem cells rescues oocyte quality and infertility in aged mice. Aging (Albany NY).
2017;
9
(12)
:
2480-8
.
View Article PubMed Google Scholar -
Igarashi
H.,
Takahashi
T.,
Abe
H.,
Nakano
H.,
Nakajima
O.,
Nagase
S.,
Poor embryo development in post-ovulatory in vivo-aged mouse oocytes is associated with mitochondrial dysfunction, but mitochondrial transfer from somatic cells is not sufficient for rejuvenation. Human Reproduction (Oxford, England).
2016;
31
(10)
:
2331-8
.
View Article PubMed Google Scholar -
Sheng
X.,
Yang
Y.,
Zhou
J.,
Yan
G.,
Liu
M.,
Xu
L.,
Mitochondrial transfer from aged adipose-derived stem cells does not improve the quality of aged oocytes in C57BL/6 mice. Molecular Reproduction and Development.
2019;
86
(5)
:
516-29
.
View Article PubMed Google Scholar -
Zhang
C.,
Tao
L.,
Yue
Y.,
Ren
L.,
Zhang
Z.,
Wang
X.,
Mitochondrial transfer from induced pluripotent stem cells rescues developmental potential of in vitro fertilized embryos from aging females\textdagger. Biology of Reproduction.
2021;
104
(5)
:
1114-25
.
View Article PubMed Google Scholar -
Pérez-Sánchez
M.,
Díez-Juan
A.,
Beltrán
D.,
Mifsud
A.,
Mercader
A.,
Vidal
C.,
Mitochondrial DNA content decreases during in vitro human embryo development: insights into mitochondrial DNA variation in preimplantation embryos donated for research. F&S Science.
2020;
1
(1)
:
36-45
.
View Article PubMed Google Scholar -
Jiang
Z.,
Shi
C.,
Han
H.,
Fu
M.,
Zhu
H.,
Han
T.,
Autologous non-invasively derived stem cells mitochondria transfer shows therapeutic advantages in human embryo quality rescue. Biological Research.
2023;
56
(1)
:
60
.
View Article PubMed Google Scholar -
Zhang
Q.,
Hao
J.X.,
Liu
B.W.,
Ouyang
Y.C.,
Guo
J.N.,
Dong
M.Z.,
Supplementation of mitochondria from endometrial mesenchymal stem cells improves oocyte quality in aged mice. Cell Proliferation.
2023;
56
(3)
:
e13372
.
View Article PubMed Google Scholar -
Woods
D.C.,
Tilly
J.L.,
Autologous Germline Mitochondrial Energy Transfer (AUGMENT) in Human Assisted Reproduction. Seminars in Reproductive Medicine.
2015;
33
(6)
:
410-21
.
View Article PubMed Google Scholar -
Fakih
M.H.,
Oogonial precursor cell-derived autologous mitochondria injection to improve outcomes in women with multiple IVF failures due to low oocyte quality: a clinical translation. Reproductive Sciences.
2015;
22
(12)
:
1612-7
.
View Article Google Scholar -
Oktay
K.,
Baltaci
V.,
Sonmezer
M.,
Turan
V.,
Unsal
E.,
Baltaci
A.,
Oogonial Precursor Cell-Derived Autologous Mitochondria Injection to Improve Outcomes in Women With Multiple IVF Failures Due to Low Oocyte Quality: A Clinical Translation. Reproductive Sciences (Thousand Oaks, Calif.).
2015;
22
(12)
:
1612-7
.
View Article PubMed Google Scholar -
Labarta
E.,
de Los Santos
M.J.,
Herraiz
S.,
Escribá
M.J.,
Marzal
A.,
Buigues
A.,
Autologous mitochondrial transfer as a complementary technique to intracytoplasmic sperm injection to improve embryo quality in patients undergoing in vitro fertilization-a randomized pilot study. Fertility and Sterility.
2019;
111
(1)
:
86-96
.
View Article PubMed Google Scholar -
Tilly
J.L.,
Woods
D.C.,
The obligate need for accuracy in reporting preclinical studies relevant to clinical trials: autologous germline mitochondrial supplementation for assisted human reproduction as a case study. Therapeutic Advances in Reproductive Health.
2020;
14
:
2633494120917350
.
View Article PubMed Google Scholar -
Cabrera
F.,
Castañeda
V.,
Morales
E.,
Velarde
F.,
Ortega
M.,
Leon-Sosa
A.,
Early evidence of the artificial transfer/transplant of mitochondria to oocytes and zygotes by MitoCeption. Mitochondrion.
2022;
65
:
102-12
.
View Article PubMed Google Scholar -
Tang
S.,
Yang
N.,
Yu
M.,
Wang
S.,
Hu
X.,
Ni
H.,
Noninvasive autologous mitochondria transport improves the quality and developmental potential of oocytes from aged mice. F&S Science.
2022;
3
(4)
:
310-21
.
View Article PubMed Google Scholar -
Tilly
J.L.,
Woods
D.C.,
Compositions and Methods for Autologous Germline Mitochondrial Energy Transfer. United States Patent Number. 8,642,329. 2014
.
-
Santos
T.A.,
El Shourbagy
S.,
St John
J.C.,
Mitochondrial content reflects oocyte variability and fertilization outcome. Fertility and Sterility.
2006;
85
(3)
:
584-91
.
View Article PubMed Google Scholar -
Diez-Juan
A.,
Rubio
C.,
Marin
C.,
Martinez
S.,
Al-Asmar
N.,
Riboldi
M.,
Mitochondrial DNA content as a viability score in human euploid embryos: less is better. Fertility and Sterility.
2015;
104
(3)
:
534-41.e1
.
View Article PubMed Google Scholar -
Fragouli
E.,
Spath
K.,
Alfarawati
S.,
Kaper
F.,
Craig
A.,
Michel
C.E.,
Altered levels of mitochondrial DNA are associated with female age, aneuploidy, and provide an independent measure of embryonic implantation potential. PLOS Genetics.
2015;
11
(6)
:
e1005241
.
View Article PubMed Google Scholar -
Castelluccio
N.,
Reliability of blastomere versus trophectoderm biopsy in preimplantation genetic testing for mitochondrial DNA disorders. Human Reproduction.
2023;
38
(Supplement_1)
:
dead093-586
.
View Article Google Scholar -
Wang
J.,
Diao
Z.,
Zhu
L.,
Zhu
J.,
Lin
F.,
Jiang
W.,
Trophectoderm Mitochondrial DNA Content Associated with Embryo Quality and Day-5 Euploid Blastocyst Transfer Outcomes. DNA and Cell Biology.
2021;
40
(5)
:
643-51
.
View Article PubMed Google Scholar -
Arnanz
A.,
De Munck
N.,
Bayram
A.,
El-Damen
A.,
Abdalla
A.,
ElKhatib
I.,
Blastocyst mitochondrial DNA (mtDNA) is not affected by oocyte vitrification: a sibling oocyte study. Journal of Assisted Reproduction and Genetics.
2020;
37
(6)
:
1387-97
.
View Article PubMed Google Scholar -
Scott
R.T.,
Sun
L.,
Zhan
Y.,
Marin
D.,
Tao
X.,
Seli
E.,
Mitochondrial DNA content is not predictive of reproductive competence in euploid blastocysts. Reproductive Biomedicine Online.
2020;
41
(2)
:
183-90
.
View Article PubMed Google Scholar -
May-Panloup
P.,
Boguenet
M.,
Hachem
H.E.,
Bouet
P.E.,
Reynier
P.,
Embryo and Its Mitochondria. Antioxidants.
2021;
10
(2)
:
139
.
View Article PubMed Google Scholar -
Richani
D.,
Dunning
K.R.,
Thompson
J.G.,
Gilchrist
R.B.,
Metabolic co-dependence of the oocyte and cumulus cells: essential role in determining oocyte developmental competence. Human Reproduction Update.
2021;
27
(1)
:
27-47
.
View Article PubMed Google Scholar -
Adriaenssens
T.,
Wathlet
S.,
Segers
I.,
Verheyen
G.,
De Vos
A.,
Van der Elst
J.,
Cumulus cell gene expression is associated with oocyte developmental quality and influenced by patient and treatment characteristics. Human Reproduction (Oxford, England).
2010;
25
(5)
:
1259-70
.
View Article PubMed Google Scholar -
Sayutti
N.,
Abu
M.A.,
Ahmad
M.F.,
PCOS and Role of Cumulus Gene Expression in Assessing Oocytes Quality. Frontiers in Endocrinology (Lausanne).
2022;
13
:
843867
.
View Article PubMed Google Scholar -
Gebhardt
K.M.,
Feil
D.K.,
Dunning
K.R.,
Lane
M.,
Russell
D.L.,
Human cumulus cell gene expression as a biomarker of pregnancy outcome after single embryo transfer. Fertility and Sterility.
2011;
96
(1)
:
47-52.e2
.
View Article PubMed Google Scholar -
Adriaenssens
T.,
Van Vaerenbergh
I.,
Coucke
W.,
Segers
I.,
Verheyen
G.,
Anckaert
E.,
Cumulus-corona gene expression analysis combined with morphological embryo scoring in single embryo transfer cycles increases live birth after fresh transfer and decreases time to pregnancy. Journal of Assisted Reproduction and Genetics.
2019;
36
(3)
:
433-43
.
View Article PubMed Google Scholar -
Martínez-Moro
Á.,
Lamas-Toranzo
I.,
González-Brusi
L.,
Pérez-Gómez
A.,
Padilla-Ruiz
E.,
García-Blanco
J.,
mtDNA content in cumulus cells does not predict development to blastocyst or implantation. Human Reproduction Open.
2022;
2022
(3)
:
hoac029
.
View Article PubMed Google Scholar -
Ijuin
A.,
Non-invasive analysis of mitochondrial DNA mutation for embryo evaluation. Human Reproduction.
2023;
38
(Supplement_1)
:
dead093-1028
.
-
Kobayashi
M.,
Kobayashi
J.,
Shirasuna
K.,
Iwata
H.,
Abundance of cell-free mitochondrial DNA in spent culture medium associated with morphokinetics and blastocyst collapse of expanded blastocysts. Reproductive Medicine and Biology.
2020;
19
(4)
:
404-14
.
View Article PubMed Google Scholar -
Stigliani
S.,
Orlando
G.,
Massarotti
C.,
Casciano
I.,
Bovis
F.,
Anserini
P.,
Non-invasive mitochondrial DNA quantification on Day 3 predicts blastocyst development: a prospective, blinded, multi-centric study. Molecular Human Reproduction.
2019;
25
(9)
:
527-37
.
View Article PubMed Google Scholar -
Stigliani
S.,
Anserini
P.,
Venturini
P.L.,
Scaruffi
P.,
Mitochondrial DNA content in embryo culture medium is significantly associated with human embryo fragmentation. Human Reproduction (Oxford, England).
2013;
28
(10)
:
2652-60
.
View Article PubMed Google Scholar -
Wolff
M. von,
Haaf
T.,
In Vitro Fertilization Technology and Child Health. Deutsches Ärzteblatt International.
2020;
117
(3)
:
23-30
.
View Article PubMed Google Scholar -
Giorgione
V.,
Parazzini
F.,
Fesslova
V.,
Cipriani
S.,
Candiani
M.,
Inversetti
A.,
Congenital heart defects in IVF/ICSI pregnancy: systematic review and meta-analysis. Ultrasound in Obstetrics & Gynecology.
2018;
51
(1)
:
33-42
.
View Article PubMed Google Scholar -
Mirzaeian
L.,
Eivazkhani
F.,
Saber
M.,
Moini
A.,
Esfandiari
F.,
Valojerdi
M.R.,
In-vivo oogenesis of oogonial and mesenchymal stem cells seeded in transplanted ovarian extracellular matrix. Journal of Ovarian Research.
2023;
16
(1)
:
56
.
View Article PubMed Google Scholar -
Tiwari
S.,
Dewry
R.K.,
Srivastava
R.,
Nath
S.,
Mohanty
T.K.,
Targeted antioxidant delivery modulates mitochondrial functions, ameliorates oxidative stress and preserve sperm quality during cryopreservation. Theriogenology.
2022;
179
:
22-31
.
View Article PubMed Google Scholar -
Escada-Rebelo
S.,
Cristo
M.I.,
Ramalho-Santos
J.,
Amaral
S.,
Mitochondria-Targeted Compounds to Assess and Improve Human Sperm Function. Antioxidants {&}amp; Redox Signaling.
2022;
37
(7-9)
:
451-80
.
View Article PubMed Google Scholar -
Ismail
H.Y.,
Hussein
S.,
Shaker
N.A.,
Rizk
H.,
Wally
Y.R.,
Stem Cell Treatment Trials for Regeneration of Testicular Tissue in Laboratory Animals
Stem Cell Treatment Trials for Regeneration of Testicular Tissue in Laboratory Animals. Reproductive Sciences (Thousand Oaks, Calif.).
2023;
30
(6)
:
1770-81
.
View Article PubMed Google Scholar -
Baert
Y.,
Rombaut
C.,
Goossens
E.,
Scaffold-Based and Scaffold-Free Testicular Organoids from Primary Human Testicular Cells. Methods in Molecular Biology (Clifton, N.J.).
2019;
1576
:
283-90
.
View Article PubMed Google Scholar
Comments
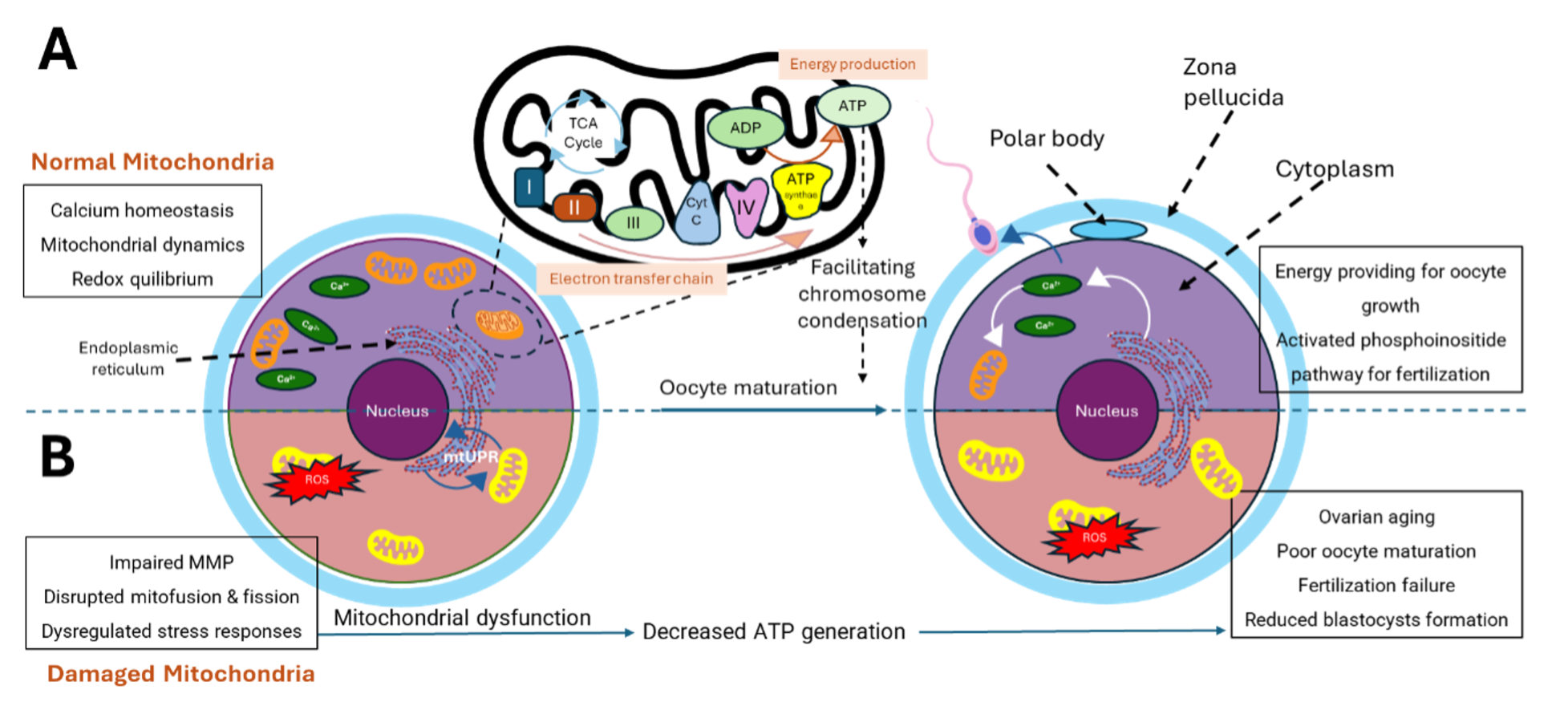
Article Details
Volume & Issue : Vol 12 No 1 (2025)
Page No.: 7054-7081
Published on: 2025-01-31
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 1851 times
- PDF downloaded - 464 times
- XML downloaded - 104 times
 Biomedpress
Biomedpress




