Abstract
Introduction: Oxidative stress is a significant issue arising from the excessive production of oxidants by superoxide oxides and nitric oxides in our body, which leads to inflammation and tissue damage. In patients with inflammatory bowel disease (IBD), the immune system mistakenly identifies food as antigens, releasing various cytokines to combat this perceived threat and causing IBD symptoms. The loss of intestinal barrier integrity is directly linked to the severity of IBD. It results in a leaky gut, bacterial infiltration, and an increase in inflammatory cytokines. The immune system typically combats infections through the generation of various T- and B-lymphocytes, leading to an adaptive immune response.
Method: In this study, we evaluated the prophylactic effect of a novel combinatorial probiotic formulation, "ABT," in an IBD model in male BALB/c mice. 106 CFU of ABT was administered orally to the mice. Subsequently, 3% DSS was administered orally to induce colitis. Body weight loss was monitored, as it is one of the critical clinical symptoms of colitis. After sacrifice, various parameters were analyzed to validate the efficacy of the probiotic formulation.
Results: The formulation prevented the symptoms of colitis, oxidative stress, maintained colon length, and achieved a balance in the expression patterns of pro-inflammatory cytokines (iNOS, IFNg) with the junctional proteins mRNA expression (Claudin-1, ZO-1). Thus, our 3-strain novel formulation can prophylactically block the Th1 mediated pathway.
Conclusion: Our study concludes that the probiotic 3-strain "ABT," when administered prophylactically, prevented the Th1 mediated immune response and can be considered for use as a wellness health drink.
Introduction
Inflammatory bowel disease (IBD) is the umbrella term for Crohn's Disease (CD) and Ulcerative Colitis (UC), the two major forms of the disease. CD involves patchy inflammation throughout the intestinal layer and can occur in all layers of the intestine. In contrast, UC exclusively affects the innermost lining of the colon and creates continuous inflammation throughout the colon's lining1. IBD is associated with damaged barrier function in the intestinal epithelium. In healthy individuals, the intestinal epithelium maintains an intact and effective barrier function against pathogens. In IBD, compromised barrier function allows bacterial products to cross the mucosal barrier, alongside infiltration of pathogenic bacteria, igniting uncontrollable inflammatory signal cascades and leading to a classic adaptive immune response2.
The incidence and prevalence of inflammatory bowel disease are highest in Western countries, including the United States of America and the United Kingdom. Over the last decade, IBD has rapidly emerged in Eastern countries as well, narrowing the gap between the two regions with an increasing number of IBD cases in Eastern countries. Asia, notably China, has the highest incidence of IBD.
Various chemical agents play a critical role in mimicking human IBD. DSS (dextran sulfate sodium), TNBS (2, 4, 6-trinitrobenzene sulfonic acid), or Oxazolone act directly on the colonic epithelium, damaging the barrier integrity by infiltrating immune cells, thus accelerating and perpetuating ongoing inflammation. Tight junctions are crucial in regulating the barrier integrity throughout the intestinal epithelium. The mucous layer serves as a major defense system in the gut's intestinal epithelium, crucial for pathogen clearance and inhibition of pathogenic infection and inflammation. The mucous layer's thickness, evidenced by the presence of the highly glycosylated polymeric protein mucin, is decreased in DSS-induced disrupted intestinal epithelium. Inflammatory bowel disease alters the expression of certain tight junctional proteins like Claudin-1 and ZO-1, activating various inflammatory cascades and leading to colitis development3, 4. Activated phagocytic immune cells infiltrate the gut mucosal tissue, generating reactive oxygen species (ROS) and reactive nitrogen species (RNS)5, 6.
Recent evidence suggests the pivotal role of oxidative stress in the pathogenesis and tissue damage associated with IBD. Under normal physiological conditions, the healthy cells of the intestinal tissue layer tolerate ROS levels7. ROS acts as a secondary messenger, regulating cellular physiological processes, including maintaining endogenous homeostasis and biological functions such as redox signal transduction, gene expression, and receptor activation. These processes are beneficial for tissue turnover and cell proliferation7. Excessive generation of ROS enhances membrane permeability and lipid peroxidation in the plasma membrane of intestinal tissue, activating the intestinal immune system. This leads to damage to the intestinal mucosal barrier by reducing mucous secretion and damaging tight junctions, causing an imbalance of pro-oxidant and antioxidant entities, thus triggering inflammation. Oxidative stress leads to increased levels of inflammatory cytokines like TNF-α, IL-1β, and IFN-γ, enhancing the Th1 cell response8. This forms a vicious cycle of oxidative stress-ROS-inflammation-ROS-oxidative stress, presenting a potential target for treating DSS-induced colitis, alleviating oxidative stress, immune markers, and improving intestinal mucosal barrier9, 10.
Dextran sulfate sodium (DSS) is a toxic, water-soluble, negatively charged polysaccharide reagent with molecular weights ranging from 5 to 1400 kDa. Administering DSS to mice induces inflammation and degeneration in the intestinal tract, disrupting the intestinal epithelial monolayer and allowing luminal bacteria and their associated antigens into the mucosa, leading to the generation of pro-inflammatory cytokines. Despite the unclear pathophysiology of DSS-induced intestinal inflammation in IBD, its multi-mechanistic pathological cascade is linked to the disruption of the monolayer lining of the colon, recruiting inflammatory cells, and accelerating the excessive release of pro-inflammatory cytokines such as tumor necrosis factor-α (TNFα) and interleukin1-β (IL1β), leading to necrosis. Thus, the DSS-induced IBD model in research is relevant due to its simplicity and reproducibility in animal studies11.
As a cure, various antibiotics are used in treating IBD. Nitroimidazoles and fluoroquinolones, the two types of antibiotics with the highest IBD risk in the study, are commonly prescribed for gastrointestinal infections. For nitroimidazoles, the increased risk steadily climbs with age for those older than 60 years. With fluoroquinolones, the increased risk is primarily concentrated among people aged 40 to 60 years. Antibiotic exposure is associated with an increased risk of IBD recurrence after 2-3 months of use, mainly in individuals aged 40 years and older12. Moreover, recent work has shown that continual use of antibiotics to treat IBD can increase the effect of enterocolitis. The efficacy of current treatments is temporary, relieving symptomatic complications after a certain time. Excessive use of antibiotics damages the normal luminal microorganisms, allowing pathogenic bacteria to enter the gut, adhere, and grow on the intestinal layer13. Thus, there is a need for an alternative approach to treating colitis14.
A safer therapeutic alternative is offered by probiotics in treating IBD15. Probiotics are "live microorganisms that beneficially affect the host by improving the indigenous microflora's properties." Probiotics improve immune responses, regulate the gut microbiome, leading to better digestion, and prevent pathogenic infections. Commonly used probiotics include Lactobacillus sp., Bifidobacterium sp., Saccharomyces sp., and Streptococcus sp., effectively used alone or in combination as therapeutic agents in various chemically induced IBD disease models16, 17.
Recent advances in genetic engineering have led to the development of genetically engineered probiotic strains to act as "intestinal biosensors" (to detect inflammatory markers) or "intestinal biotherapeutics" (to improve drug delivery at the mucosal surface and directly release therapeutic substances into the intestinal lumen). Engineered probiotic strains are developed using plasmids as vectors, with exogenous DNA fragments containing genes for immunoregulatory cytokines and anti-inflammatory mediators inserted into the plasmids by restriction enzymes. This enables the recombinant probiotic strain to express these regulatory proteins18.
However, these advancements face challenges due to the limited knowledge of relevant biomarkers specific to gut inflammation. Most published studies have focused on developing engineered probiotics capable of expressing therapeutic molecules (biotherapeutic probiotics). These biotherapeutic probiotics are live bacteria designed to produce anti-inflammatory molecules in situ, offering the main advantage of releasing therapeutics at the inflammation sites. This direct in situ release maximizes therapeutic concentrations in the target tissue using relatively smaller doses of the therapeutic compound, thus limiting systemic side effects. Nonetheless, this approach has limitations, including the need for significant energy for the constitutive expression of these substances, increasing the risk of overproducing the therapeutic substance at unwanted sites and potentially impacting both effectiveness and safety18.
Several bioengineered techniques using Lactococcus lactis are being employed in IBD treatment. Trefoil factors (TFF) and anti-tumor necrosis factor-α (TNF-α) nanobodies (single-domain antibody fragments) have been constitutively expressed in L. lactis and tested for therapeutic effects in DSS-induced colitis in mice19. One study explored the use of L. lactis with the Microbial Anti-inflammatory Molecule (MAM)-encoding plasmid20. MAM, a peptide produced by Faecalibacterium prausnitzii, downregulates NF-κB expression in vitro. Another study used Bifidobacterium longum genetically modified to express the α-melanocyte-stimulating hormone (α-MSH)21. α-MSH is a tridecapeptide derived from pro-opiomelanocortin that exhibits potent anti-inflammatory properties by downregulating the release of proinflammatory cytokines and mediators, such as ILs, TNF-α, and NO, and upregulating the anti-inflammatory cytokine IL-1022. Although bioengineered probiotics have shown beneficial effects in preclinical mouse models, limitations persist in human applications. One main reason for these limitations is the difference in microbiota between mice and humans, potentially reducing the growth rate of genetically engineered bacteria due to differing environmental conditions18. Genetically engineered probiotics could struggle to achieve colonization due to the complexity of establishing a niche to survive in the gut microbiome.
Beyond bioengineering prospects, several studies have demonstrated the effectiveness of probiotics, such as fermented milk products, in reducing numerous infectious and inflammatory diseases23. Probiotic L. paracasei fermented milk reduced infections in the respiratory and gastrointestinal tracts of young children24. Lactobacillus strains like L. jensenii, L. reuteri, and L. casei have been shown to generate anti-inflammatory effects in mice by downregulating the activity of TNF-α, IFN-γ, and maintaining intestinal barrier integrity25. Various therapeutic studies have employed Lactobacillus vulgaricus and Streptococcus thermophilus, showing that both strains synergistically modulated IL-6, IFN-γ, and TNF-α secretion, while enhancing IL-2 and IL-4 expression, thereby regulating the Th1 immune response26. Generally, the three bacteria used in this study, labeled "A", "B", and "T", either separately or together, compete with pathogens and colonize the epithelium. They signal the repair of the leaky barrier by stabilizing the junctional proteins, upregulating mucus production, and downregulating inflammatory genes in the mucosa. This leads to reduced inflammatory mediators and ultimately tissue repair in a therapeutic condition. Probiotics exhibit diverse mechanisms of action, one of which includes a cytoprotective effect on gastric mucosa integrity by strengthening epithelial junctions and preserving mucosal barrier function27.
A literature review revealed that Bifidobacterium bifidum and Lactobacillus acidophilus, as singular strains, are widely studied and are the best probiotics to heal our gut. The prophylactic effect of either a single strain among "A", "B", and "T" or two strains among these ("A", "B", and "T") has been studied and found beneficial, with different species names used in those studies. The specific species used in this study to validate its prophylactic activity have not been studied earlier.
Thus, this study utilizes a novel probiotic formulation to prevent the development of inflammatory bowel disease. The novelty of our study lies in the synergistic use of three bacterial strains and their formulation in a proprietary blend. Our formulation, containing three probiotic bacteria named "A", "B", and "T" mixed in proprietary ratios, has efficiently proven to possess antimicrobial activity against various gut pathogenic bacteria through in vitro assays performed in our lab. We have now advanced this work to a pre-clinical mouse model to study the prophylactic activities of this novel combinatorial formulation against DSS-induced IBD15, 28.
Methods
Preparation of Whey Water
Through in vitro viability assays, it was observed that, among the three-strain probiotic formulation, the "T" bacteria initiates growth, followed by "B" and "A". Through various in vitro biochemical assays, a specific combination, maintained in a proprietary ratio, has shown to have a more beneficial effect. Thus, an in vivo-based prophylactic study has been undertaken to study their efficacy. For culturing the probiotics in milk, a powdered culture containing "A", "B", and "T" together was added to pasteurized double-toned milk that had been boiled and cooled to room temperature. This mixture was then incubated overnight at 42°C. The whey water (ww) formed above the fermented milk (curd) was strained through a nylon mesh and collected in tubes (Figure 1). The shelf life of the probiotics in whey water was determined to be 120 hours as observed through an in vitro growth curve study. The active period of these 3-strain probiotics was maintained for better viability and consistency throughout the study.
| Temperature | Time | No. of cycles | Purpose |
| 42˚C | 30 mins | 1 | cDNA synthesis |
| 95˚C | 2 mins | Inactivation of enzymes |
| Temperature | Time | No. of cycle |
|---|---|---|
| 95ºC | 5 min | 1 cycle |
| 95ºC | 45 sec | 30 cycles |
| TºC | 30 sec | |
| 72ºC | 45 sec | |
| 72ºC | 10 min | 1 cycle |
| 4ºC | Infinite |
| Gene | Primer sequences | Tm (˚C) | Product size |
|---|---|---|---|
| GAPDH | F 5’- GAGGGGCCATCCACAGTCTTC 3’ R 5’- CATCACCATCTTCCAGGAGCG 3’ | 62.75 | 357bp |
| Claudin 1 | F 5’ AGGTCTGGCGACATTAGTGG 3’ R 5’ CGTGGTGTTGGGTAAGAGGT 3’ | 59.35 | 204 bp |
| ZO-1 | F 5’ ACTCCCACTTCCCCAAAAAC 3’ R 5’ CCACAGCTGAAGGACTCACA 3’ | 58.32 | 166 bp |
| IFN γ | F 5’AGCGGCTGAACTCAGATTGTAG 3’ R 5’GTCACGTTTTCAGCTGTATAGGG 3’ | 62.9 | 247 bp |
| iNOS | F 5’ AATGGCAACATCAGGTCGGCCATCACT 3’ R 5’ GCTGTGTGTCACAGAAGTCTCGAACTC 3’ | 66.48 | 454 bp |
| TGF β | F 5’ ACCGCAACAACGCCATCTAT 3’ R 5’ GTAACGCCAGGAATTGTTGC 3’ | 51.8 | 200 bp |
In Vivo Validation
Ethical Approval
This in vivo experiment was performed according to the guidelines published by the institutional and departmental animal ethics committee. Mice were housed under proper infection-free conditions at the departmental animal house. The ethical approval code is ERB/ZOO/2023/I, dated August 2nd, 2023.
Study Design
Six-week-old male BALB/c mice (20-25 gm) were divided into four groups (n = 4):
Mice were sacrificed on day 15, and desired tissues of interest were collected for performing various assays.
Change in Body Weight in Mice
The body weights of mice from all 4 groups were recorded throughout the experiment model on specific days 0, 7, and 14.
Visual Observation of Colon
Colon tissues from different groups were immediately isolated after sacrifice. The colon was excised between the ileocecal junction and the proximal rectum and placed on a non-absorbent surface; its length was measured with a ruler without stretching or damaging the colon.
Collection of Tissues
For subsequent anti-oxidative assays, colon and intestine tissues were collected in 1X PBS after flushing fecal matters from the colon and intestine with chilled 1X PBS. These tissues were homogenized in RIPA buffer and mixed thoroughly using a rotary mixer (TARSON) for 2 hours at 4°C, followed by centrifugation at 10,000 g at 4°C for 20 mins. The supernatant was collected and analyzed for oxidative stress and anti-oxidative markers15.
For gene expression studies by RT-PCR, intestine tissues were collected in RNA Later (Ambion, Inc) and stored at -20˚C until further use. Colon tissues were collected in 4% paraformaldehyde (PFA) (TCI, P0018) for the preparation of cryo-blocks for necessary histological studies.
Morphological Observation of Colon Tissue
Colon tissues were processed with 15% and 30% sucrose overnight. After the sucrose was fully imbibed in the tissue, it was embedded in Tissue Freezing media (Leica) and cut into 5 µm sections using a cryocut instrument (Leica CM1860). The sections were stained with hematoxylin (Merck- DB1DF71034) and eosin (Nice- E30971) to assess cellular and overall morphological architecture, and alcian blue (HIMEDIA-RM471) to identify mucus-containing goblet cells of the colon. The stained sections were observed under a light microscope at 4X and 10X magnifications (Olympus BX41)15.
Determination of Oxidative Stress Markers (NO and MDA) and Anti-Oxidative Markers (SOD and Catalase)
Nitric oxide content (NO) in colon and intestine tissues was determined using Griess reagents, which comprise 1% Sulphanilamide (SRL: 1949107), 0.1% NED (HIMEDIA-RM1073), 5% ortho-phosphoric acid (MERCK-AG8A580394) added to the sample (supernatant of colon and intestine), incubated for 10 minutes, and Optical Density (OD) was measured at 540 nm. A standard curve was prepared using 0.1M NaNO2 (MERCK-17543).
The concentration of malondialdehyde (MDA) in both colon and intestine tissues was determined using Trichloro-Acetic acid (TCA, SRL-90544), Thio-Barbituric acid (TBA, SIGMA-T5500), n-butanol (SRL-227139) added to the sample, and OD was measured at 532 nm29.
Similarly, Superoxide dismutase (SOD) activity in these tissues was determined using 50 mM Na2CO3 (MERCK-17844), 0.1 mM EDTA (Promega-000014215), 5 mM NBT (Nitroblue Tetrazolium) (SRL-48898), and 1 mM Hydroxylamine hydrochloride (HAH) (SRL-84784) along with the sample, incubated for 5 minutes, and OD was measured at 560 nm30. A standard curve was prepared using different concentrations (20 units/ml, 5 units/ml, and 1.5 units/ml) of Superoxide dismutase (SOD) tablet (NUTRACEUTICAL: HGS AESTHETICS: ALC-023).
Catalase activity was measured using 10mM H2O2 reagent (MERCK-CJ9C90644) dissolved in 50 mM Phosphate buffer along with the sample, and OD was measured at 240 nm at an interval of 10 mins. The OD for all the above-mentioned assays was measured using a spectrophotometer (Varioskan LUX)29.
Determination of Intestinal Alkaline Phosphatase Activity
The brush border enzyme, intestinal alkaline phosphatase (IAP), plays an important role in maintaining barrier integrity and preventing bacterial invasion through the gut mucosal barrier. The intestinal supernatant was incubated with 2-Amino-2-Methyl-1-Propanol (AMP) buffer (SRL:50407) and p-nitrophenyl phosphate (PNPP) substrate (SRL-88485) at 37°C for 15 minutes. Both reagents were freshly prepared. Following the addition of 6N HCl and 1N NaOH (BDH: 89021-500G), the OD was measured at 405 nm15. The standard curve was prepared using various concentrations (200 µM, 100 µM, 80 µM, 60 µM, 40 µM, and 20 µM) of PNP (p-nitrophenol) (Himedia: GRM1182-500G).
Determination of Changes in Gene Expression by Reverse Transcriptase PCR (RT-PCR)
Intestine tissue was washed in chilled 1X PBS, chopped into pieces, and homogenized in Trizol reagent (Life Technologies, USA). Chloroform was added, and the tubes were shaken vigorously before being centrifuged at 12000g for 10 minutes at 4°C. The upper aqueous layer containing RNA was extracted, mixed with 100% chilled alcohol, and incubated overnight at -20°C. The next day, the RNA was centrifuged again at 12000g for 10 minutes at 4°C, the supernatant was discarded, and the pellet was washed in 75% ethanol and centrifuged at 7500g for 5 minutes at 4°C until it dried into a gel-like pellet. The concentration of RNA was measured by resuspending the pellet in nuclease-free water using a Nanodrop (Jenway, UK). RT-PCR was performed from the extracted RNA to produce cDNA using the Verso cDNA synthesis kit (Life Technologies, USA). cDNA was prepared by following the below-mentioned cycle using 2 µg RNA (Table 1)15, 31.
Given that the DSS-induced IBD model primarily follows TH1-mediated immune responses, it involves the roles of IFNγ (interferon-gamma), iNOS (inducible nitric oxide synthase), TGFβ (transforming growth factor-beta), Claudin1, and ZO1 (Zonula occludens) in the degeneration of intestinal epithelium by disrupting barrier function integrity. Thus, the expression of these genes was assessed by PCR (Table 2) using specific primers (Table 3), with GAPDH as the housekeeping gene and a 100 bp marker (100-2000 bp; Proxy B, SRL 84628). PCR products were run on a 1.2% agarose gel (G-Biosciences, USA). Bands were observed under UV light in a gel-doc system (Biorad Chemidoc Imaging System), and band intensities were determined using ImageJ software.
Statistical Analysis
All statistical analyses were performed using the t-test. A value of P < 0.05 was considered significant.
Results
Administration of whey water prevented the symptoms of colitis in mice
Weight loss is one of the most prominent clinical symptoms of IBD. Prophylactic administration of whey water increased body weight compared to disease groups throughout the disease model. Whey water (W1D) significantly restored body weight by 1.13-fold (p < 0.05) compared to the DSS group on day 14. Meanwhile, in the W2D groups, body weight was restored with respect to DSS by 1.05 and 1.19-fold on day 7 and 14, respectively. Administration of DSS caused gradual weight loss over 7 days of treatment with respect to the control (Figure 3 A). Thus, it may be inferred that mice groups, where whey water was continually administered along with DSS (W2D), showed an increased body weight as compared to group where whey water was administered only on days before DSS administration (W1D).
Administration of whey water maintained the colon length of mice
The length of the colon was maintained in prophylactic groups as compared to the disease group. Colon length in DSS-induced colitis mice showed a significant reduction by 1.48-fold with respect to control, whereas the prophylactic groups (W1D) increased their colon length by 1.30-fold each with respect to the diseased groups, and W2D groups also increased the colon length by 1.25-fold with respect to the DSS groups (Figure 3 B). Thus, from the colon length study, it may be concluded that W1D groups, where whey water was administered only on days before DSS administration, showed better colon length maintenance compared to W2D groups.
Prophylactic whey water maintained the morphological structure of the colon
DSS-induced inflammation and histological damage in the colon are demonstrated by increased crypt distortion and inflammatory cellular infiltration that leads to mucosal destruction. The epithelial architecture of the colon was damaged in DSS groups, which was observed by hematoxylin and eosin staining, whereas, the prophylactic groups prevented the architectural damage caused by DSS. In the morphological structure, it may be inferred that W1D better restored the original epithelial architecture compared to DSS.
The mucus layer along the GI tract, formed by mucin secretion from goblet cells, is most important for mucosal defense. In a healthy condition, mucin granules are present throughout the colon. When stained with alcian blue, it stains only the mucin granules in the goblet cells, with little to no staining of the peri-nuclear cytoplasm, whereas, in IBD, the degeneration of colonic mucosa resulted in the depletion of the mucus layer and reduction in the number of goblet cells. In our histological staining, goblet cells were depicted by alcian blue staining. The DSS-induced mice group showed depleted goblet cells, whereas the presence of goblet cells in prophylactic groups was similar to control. If both groups are compared, the W2D group mice showed better prophylactic activity by maintaining their histological morphology and optimum mucus level after DSS administration (Figure 4 and Figure 5).
Prophylactic whey water prevented the development of oxidative stress in the colon and intestine
In IBD, oxidative stress is an important factor that leads to a degenerated colonic layer, which is already seen through histology. There occurs a huge imbalance between oxidants and antioxidants. Oxidative stress results in a significant increase in NO levels in colitis by 2.34-fold with respect to control in the colon, whereas, the prophylactic groups (W1D and W2D) exhibited a significant reduction in NO level by 1.79 and 4.89-fold with respect to DSS. W1D groups showed better inhibition of NO, a potent oxidative stress marker (Figure 6 A). Lipid peroxidation is another oxidative stress-induced reaction which is prominent in DSS-induced colitis by 1.16-fold with respect to control. Lipid peroxidation results in the generation of Malonaldehyde (MDA). MDA content in the colon of the prophylactic group (W1D & W2D) was reduced compared to the DSS group by 5.80 and 1.03-fold (significant) respectively, showing the preventive effect of whey water (Figure 6 B). W1D groups showed better preventive activity by lowering the oxidative stress byproduct, Malonaldehyde (MDA).
Antioxidative enzymes (SOD and catalase) work in a simultaneous fashion (Figure 6 C, D). Besides nitric oxide, superoxide radicals are also released due to oxidative stress in IBD. SOD plays an important role in scavenging superoxides to produce hydrogen peroxides, and in turn, catalase acts on hydrogen peroxides to form H2O. Activity of SOD and catalase in the colon of prophylactic mouse groups (W1D & W2D) was enhanced with respect to DSS. SOD activity in the colon of W1D & W2D groups was enhanced by 1.16 and 1.53 folds respectively. DSS-induced colitis results in decreased catalase and SOD activity as compared to control by 3.33 and 1.67 folds respectively. Catalase activity in the colon of W1D & W2D was significantly increased by 1.08 and 1.44 folds respectively with respect to DSS. Both prophylactic groups (W1D and W2D) showed reduced oxidative stress and an increased antioxidative activity compared to DSS. Likewise, in DSS-induced intestine of mice, oxidative stress was also evident by the generation of nitric oxide (NO) and Malonaldehyde (MDA) by-product. Both NO and MDA content got significantly increased by 1.15 fold and 1.21 fold respectively as compared to control and in the case of prophylactic groups (W1D and W2D), showed a reduced NO content by 4.9 and 4.5 fold, and reduced MDA content by 1.63 and 1.33 fold (Figure 7 A and B), whereas antioxidative enzymes (SOD and CAT) decreased significantly in the intestine of DSS groups by 4.79 and 0.12 fold, whereas, prophylactic groups (W1D and W2D) were found to have enhanced activity with respect to DSS. SOD level was enhanced by 1.16 and 1.15 folds respectively and CAT activity was significantly enhanced by 3.00 and 2.71 folds (Figure 7 C and D).
According to aforementioned immune modulations, it can be inferred that lipid oxidation by-product, Malonaldehyde (MDA) was significantly enhanced in the intestine as compared with the colon due to the exposure to DSS. SOD activity in both colon and intestine of DSS-induced mice was reduced, whereas catalase activity in the colon was much reduced as compared to the intestine. Overall, in both colon and intestine, W1D groups (where whey water was administered only on days before DSS administration) significantly possess better preventive activity in inhibiting lipid peroxidation, and W2D groups have enhanced catalase activity and efficiently reduce the NO content as compared to W1D, as assessed with respect to the ability to generate oxidative stress and antioxidative enzymes.
Whey water administered prophylactically has maintained an intestinal alkaline phosphatase level
Intestinal alkaline phosphatase (IAP) is an important inflammatory marker. It is secreted by the brush border epithelial cells of the proximal small intestine and is found amongst the brush border of the murine intestine and plays an important role in maintaining intestinal barrier function. DSS causes inflammation and degeneration in the intestinal gut layer that result in a reduced level of IAP by 1.48-fold with respect to control, which in turn causes reduced expression of tight junction mRNA. Prophylactic groups (W1D and W2D) succeeded in reducing the intestinal alkaline phosphatase level by 2.16 and 1.11-fold with respect to DSS in the intestinal brush border region (Figure 8). From this assay, it may be inferred that W1D groups showed an increased alkaline phosphatase level as compared to W2D groups.
Prophylactic whey water maintained a balance of pro-inflammatory cytokines and junctional proteins in the intestine
Inflammation that occurs in IBD results in the up-regulation of pro-inflammatory markers (iNOS, IFNγ) and results in the degradation of the barrier function integrity of the intestine. We have concluded that there is a significant down-regulation of junctional proteins like ZO-1 and Claudin-1 in DSS groups by 1.35 and 3.29 fold respectively with respect to DSS (Figure 9). On the contrary, disruption was prevented by up-regulating junctional proteins ZO-1 (by 1.12 and 1.63 fold respectively with respect to DSS) and Claudin-1 (by 1.15 and 1.06 respectively with respect to DSS), as evident in W1D & W2D groups (Figure 9). Moreover, about inflammatory markers, we found an upregulation of cytokine markers (iNOS, IFNγ), which was significantly increased by 1.37 and 5.58 fold with respect to control. On the other hand, whey water (W1D and W2D) groups decreased the increase in the pro-inflammatory cytokine IFN-γ (by 1.85 and 1.25 fold with respect to DSS), and the inflammatory mediator iNOS (by 3.27 and 3.77 fold with respect to DSS). Anti-inflammatory cytokines (TGFβ) showed a decreased expression pattern in DSS colitis by 4.92 fold and enhanced its expression in prophylactic groups (W1D and W2D) by 3.04 and 4.17 fold respectively as compared to DSS (Figure 10).
Discussion
Inflammatory bowel disease (IBD) is a chronic inflammatory illness characterized by an abnormal immune response in the gastrointestinal (GI) tract. Chemically induced colitis (DSS) has been extensively used to investigate the molecular mechanisms of colitis. Thus, we have established that 3% DSS-induced colitis and our novel probiotic formulation was applied as prophylaxis to study the activity against DSS-induced colitis. DSS leads to acute colitis that morphologically results in ulceration, moderate to severe submucosal edema, and lesions. These are accompanied by histopathological modifications that include granulocyte infiltration, symptoms of which are eventually expressed as bloody diarrhea. This considerably increases the output of all proinflammatory cytokines in the intestine that increases the degeneration of the intestinal and colonic mucosa32, 33.
Administration of whey water to the prophylactic (W1D and W2D) groups has maintained their body weights and prevented colitis compared to DSS. But compared to W1D, W2D groups have significantly enhanced their body weight throughout the disease model compared to DSS. Thus, prophylactic administration of probiotic formulation prevented the clinical symptoms (like body weight) in DSS-induced colitis. Moreover, experimental results have demonstrated that the colon length of the control mice group was the longest while being the shortest in the colitis mice group, whereas, the colon length of prophylactic groups was maintained with respect to DSS. W1D groups had better preventive activity as they could restore the colon length compared to W2D. The prophylactic groups prevented the degeneration in the colon even after the administration of DSS. From the above observation, it may be concluded that prophylactic administration of probiotics had significantly prevented the degeneration throughout the colonic epithelium. The degeneration of the colon was histologically validated by hematoxylin and eosin staining. DSS degenerated the morphology of the epithelial lining of the colon. The architecture of the colon tissue was preserved from DSS-induced tissue damage by both prophylactic (W1D and W2D) groups. In homeostatic conditions, the mucus layer plays an important role in the murine epithelial defense mechanism. Alcian blue stains the goblet cells, which are responsible for mucin secretion. Goblet cells are located in the colonic epithelium and secrete mucus to prevent pathogenic invasion and protect the colonic barrier function. Histological staining by alcian blue depicted a reduction of goblet cells in DSS-induced colitis, whereas, prophylactic groups had an intact mucous layer compared to DSS. Our study has shown that higher levels of antioxidants and lower levels of oxidative stress markers in the prophylactic groups were evident compared to DSS. Oxidative stress results in lipid peroxidation of colon tissue that leads to the production of Malonaldehyde (MDA) as a by-product. The administration of DSS has led to an increase in the level of pro-inflammatory mediators (iNOS) that results in the formation of nitric oxide (NO), a potent oxidative stress marker. From the above assay, it may be inferred that 3% DSS led to a significant increase in oxidative stress by generating nitric oxide. NO, in turn, leads to the epithelial lipid bilayer peroxidation, which generates MDA as a byproduct. This may result an increased degeneration of the colonic epithelium. This oxidative stress-mediated degeneration activity was significantly prevented by our novel probiotic formulation (W1D and W2D) after administering prophylactically. In the context with antioxidative enzyme activity, in homeostatic conditions, superoxide dismutase (SOD) enzymes convert superoxides generated in our body into hydrogen peroxides. In turn, catalase scavenges this hydrogen peroxide into water molecules. Antioxidative enzyme activity got significantly hindered in the diseased condition. In our disease model, prophylactically treated mice groups had maintained a similar activity as compared to control groups. After analyzing both the antioxidative and oxidative activity, it may be concluded that W1D mice groups had shown a better preventive activity than W2D groups in both the colon and intestine in the context of antioxidative and oxidative stress markers. The interconnected antioxidative enzyme activities got disrupted in DSS-induced colitis, portraying that the oxidative stress was initiated by the administration of DSS in mice.
Generally, junctional proteins (Claudin 1, ZO1) have a role in maintaining intestinal barrier integrity in homeostatic condition. Inflammatory bowel diseases, encompassing Crohn’s disease and ulcerative colitis, are characterized by chronic inflammation of the intestinal mucosa, coincident with high levels of the pro-inflammatory cytokines IFNγ, and mediators (iNOS), whereas reduced expression of TGFβ, Claudin 1, ZO1 level was found in DSS-induced inflammation. In this study, DSS induced oxidative stress which resulted in the gradual increase of pro-inflammatory cytokines (iNOS and IFNγ) and a significant decrease in the anti-inflammatory marker (TGFβ) in diseased mice, that eventually enhanced the disruption of barrier function integrity (Claudin 1, ZO1). Prophylactic groups (W1D and W2D) had significantly decreased the Th1 immune response and enhanced regulatory cytokines (TGFβ) compared to DSS. If both prophylactic groups are compared, W1D imposed a better significant preventive action by regulating the hyperactive immune response.
Our probiotic formulation has significantly prevented the occurrence of Th1 mediated inflammation. After analyzing the overall parameters, it may be inferred that when whey water is orally administered only on days before DSS administration (W1D), it works by inhibiting the lipid peroxidation and preventing degeneration in the intestine, whereas, W2D functions by enhancing both the antioxidative enzymes (SOD and CAT) as well as it inhibits the formation of reactive oxygen species by inhibiting the pro-inflammatory mediators (iNOS) and the lipid peroxidation reaction and better functions in the murine colon.
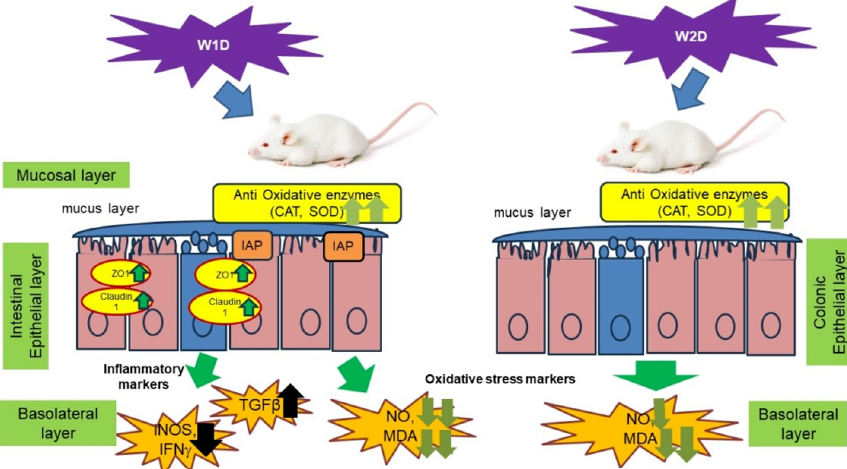
Thus, a hypothetical pathway may be inferred from this study. Administration of 3% DSS in mice has developed a degenerated colonic epithelium with reduced levels of intestinal alkaline phosphatase (IAP). This leads to the formation of the lipid peroxidation by-product, MDA (malonaldehyde), and a reduced antioxidative enzyme activity. This results in a decrease in intestinal barrier integrity by lessening the gene expression levels of Claudin 1 and ZO-1 and also increases the pro-inflammatory cytokine markers (IFNγ) and (mediator iNOS). Consequently, this generates the formation of reactive oxygen species (NO) as a potent oxidative stress marker. Antioxidative enzymes (SOD and catalase) worked together to maintain intact intestinal barrier integrity. Disbalance in these enzymes led to degenerated colonic epithelium, which subsequently enhanced Th1 mediated inflammation in the colon. Our novel combinatorial probiotics prevented inflammation by regulating these parameters. This gradual intestinal degeneration is prevented in prophylactically administered whey water groups (Figure 11, Figure 12).
Conclusions
It can be concluded from the study that novel combinatorial probiotics in the form of whey water possess significant prophylactic activities by maintaining an intact barrier function. Thus, DSS-mediated colon and intestine tissue degeneration may be overcome through prophylactic administration of whey water as per the titrated dosage regimen.
The possible prophylactic mechanism of action of whey water is by downregulating the pro-inflammatory cytokines and helping to maintain an intact barrier function. In the case of acute colitis, a reduction in tight junction gene expression patterns led to an increased expression of iNOS, which consequently increased the production of NO, a potent oxidative stress marker. The prophylactic administration of whey water has upregulated the level of intestinal brush border enzyme, specifically intestinal alkaline phosphatase (IAP), which in turn has enhanced the tight junction gene expression (Claudin 1, ZO-1), as well as the level of anti-oxidative enzyme markers. Whey water administration has maintained the level of mucus secretion from the goblet cells of colonic tissue. Moreover, it has prevented the increase in the expression of pro-inflammatory cytokines (iNOS, IFNγ, TGFβ). Eventually, the initiation of oxidative stress by the generation of reactive oxygen species (NO) is inhibited.
In conclusion, the prophylactic groups (W1D and W2D) have downregulated the Th1 mediated immune response. This study is suitable for developing a refreshing health drink for managing oxidative stress and further helping to detoxify gut toxins. This whey water, suitably evaluated through a clinical trial, may be developed as a nutraceutical for target consumer groups. Prior to the clinical trial, this combinatorial probiotic formulation may be applied to potentially upregulate the level of short-chain fatty acids, positioning it as an effective gut-detoxifying agent.
Abbreviations
α-MSH - α-Melanocyte-Stimulating Hormone, AMP - 2-Amino-2-Methyl-1-Propanol, BALB/c - A strain of laboratory mice, CD - Crohn's Disease, CFU - Colony Forming Units, Claudin-1 - Claudin-1, DSS - Dextran Sulfate Sodium, ERB - Ethics Review Board, GAPDH - Glyceraldehyde 3-Phosphate Dehydrogenase, HAH - Hydroxylamine Hydrochloride, IAP - Intestinal Alkaline Phosphatase, IBD - Inflammatory Bowel Disease, IFNγ - Interferon Gamma, IL-1β - Interleukin 1 Beta, iNOS - Inducible Nitric Oxide Synthase, MAM - Microbial Anti-Inflammatory Molecule, MDA - Malondialdehyde, mRNA - Messenger RNA, NBT - Nitroblue Tetrazolium, NO - Nitric Oxide, NP - Not Applicable / Not Provided, OD - Optical Density, PBS - Phosphate Buffered Saline, PFA - Paraformaldehyde, PNP - p-Nitrophenol, PNPP - p-Nitrophenyl Phosphate, RNA - Ribonucleic Acid, ROS - Reactive Oxygen Species, RNS - Reactive Nitrogen Species, RT-PCR - Reverse Transcriptase Polymerase Chain Reaction, SOD - Superoxide Dismutase, TBA - Thiobarbituric Acid, TCA - Trichloroacetic Acid, TFF - Trefoil Factors, TGFβ - Transforming Growth Factor Beta, TNBS - 2, 4, 6-Trinitrobenzene Sulfonic Acid, TNF-α - Tumor Necrosis Factor Alpha, UC - Ulcerative Colitis, UK - United Kingdom, USA - United States of America, ZO-1 - Zonula Occludens-1
Acknowledgments
We would like to acknowledge the Council for Scientific Research (CSIR), New Delhi, for awarding fellowship to Ms. Saheli Biswas. We cordially thank the Department of Zoology and the Dept of Neurosciences for allowing all the authors to use the Chemidoc (Biorad Chemidoc imaging system) and spectrophotometer (Varioskan LUX) instrument respectively, in the central instrument facility, University of Calcutta, Kolkata, India.
Author’s contributions
SB has performed the assays and analyzed the data related to body weight, colon length, different biochemical assays like nitric oxide, MDA, SOD and catalase activity and has written the manuscript. RD has performed and analyzed the gene expression studies. SM has given her contributions towards the writing of the manuscript. ERB has overall conceptualized the experiment plan and gave her inputs in writing the manuscript. All authors have read and approved the final manuscript.
Funding
None.
Availability of data and materials
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval
Ethical approval has been sanctioned to Ms. Saheli Biswas, no. ERB/ZOO/2023/I by the Institutional Animal Ethics Committee of the Department of Zoology, University of Calcutta on 2nd August 2023 to support the work done in this manuscript using BALB/c mouse.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
-
Zundler
S.,
Becker
E.,
Schulze
L.L.,
Neurath
M.F.,
Immune cell trafficking and retention in inflammatory bowel disease: mechanistic insights and therapeutic advances. Gut.
2019;
68
(9)
:
1688-700
.
View Article Google Scholar -
Bhattacharyya
A.,
Chattopadhyay
R.,
Mitra
S.,
Crowe
S.E.,
Oxidative stress: an essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiological Reviews.
2014;
94
(2)
:
329-54
.
View Article Google Scholar -
Mehandru
S.,
Colombel
J.F.,
The intestinal barrier, an arbitrator turned provocateur in IBD. Nature Reviews. Gastroenterology & Hepatology.
2021;
18
(2)
:
83-4
.
View Article Google Scholar -
Acharya
P.,
Beckel
J.,
Ruiz
W.G.,
Wang
E.,
Rojas
R.,
Birder
L.,
Distribution of the tight junction proteins ZO-1, occludin, and claudin-4, -8, and -12 in bladder epithelium. American Journal of Physiology. Renal Physiology.
2004;
287
(2)
:
305-18
.
View Article Google Scholar -
Chassaing
B.,
Aitken
J.D.,
Malleshappa
M.,
Matam
V.K.,
Dextran sulfate sodium (DSS)‐induced colitis in mice. Current protocols in immunology.
2014;
104
(1)
:
15-25
.
View Article Google Scholar -
Hendrickson
B.A.,
Gokhale
R.,
Cho
J.H.,
Clinical aspects and pathophysiology of inflammatory bowel disease. Clinical Microbiology Reviews.
2002;
15
(1)
:
79-94
.
View Article Google Scholar -
Wang
B.,
Wu
C.,
Dietary soy isoflavones alleviate dextran sulfate sodium-induced inflammation and oxidative stress in mice. Experimental and Therapeutic Medicine.
2017;
14
(1)
:
276-82
.
View Article Google Scholar -
Cochran
K.E.,
Lamson
N.G.,
Whitehead
K.A.,
Expanding the utility of the dextran sulfate sodium (DSS) mouse model to induce a clinically relevant loss of intestinal barrier function. PeerJ.
2020;
8
(8)
:
e8681
.
View Article Google Scholar -
Li
L.,
Peng
P.,
Ding
N.,
Jia
W.,
Huang
C.,
Tang
Y.,
Oxidative Stress, Inflammation, Gut Dysbiosis: What Can Polyphenols Do in Inflammatory Bowel Disease?. Antioxidants.
2023;
12
(4)
:
967
.
View Article Google Scholar -
Zhang
J.J.,
Xu
Z.M.,
Zhang
C.M.,
Dai
H.Y.,
Ji
X.Q.,
Wang
X.F.,
Pyrrolidine dithiocarbamate inhibits nuclear factor-κB pathway activation, and regulates adhesion, migration, invasion and apoptosis of endometriotic stromal cells. Molecular Human Reproduction.
2011;
17
(3)
:
175-81
.
View Article Google Scholar -
Guo
Y.,
Wu
X.,
Wu
Q.,
Lu
Y.,
Shi
J.,
Chen
X.,
Dihydrotanshinone I, a natural product, ameliorates DSS-induced experimental ulcerative colitis in mice. Toxicology and Applied Pharmacology.
2018;
344
:
35-45
.
View Article Google Scholar -
Jonkers
D.,
Stockbrügger
R.,
Probiotics and inflammatory bowel disease. Journal of the Royal Society of Medicine.
2003;
96
(4)
:
167-71
.
View Article Google Scholar -
Shimodaira
Y.,
Watanabe
K.,
Iijima
K.,
The risk of antibiotics and enterocolitis for the development of inflammatory bowel disease: a Japanese administrative database analysis. Scientific Reports.
2022;
12
(1)
:
7604
.
View Article Google Scholar -
Park
J.,
Cheon
J.H.,
Incidence and Prevalence of Inflammatory Bowel Disease across Asia. Yonsei Medical Journal.
2021;
62
(2)
:
99-108
.
View Article Google Scholar -
Mitra
S.,
Banerjee
E. Ray,
Novel 3-strain Combinatorial Probiotics Alleviates Symptoms of Inflammatory Bowel Disease in Mice. Acta Scientific Microbiology.
2022;
2022
:
2581-3226
.
View Article Google Scholar -
Kanwal
S.,
Joseph
T.P.,
Aliya
S.,
Song
S.,
Saleem
M.Z.,
Nisar
M.A.,
Attenuation of DSS induced colitis by Dictyophora indusiata polysaccharide (DIP) via modulation of gut microbiota and inflammatory related signaling pathways. Journal of Functional Foods.
2020;
64
:
1756-4646
.
View Article Google Scholar -
Prakash
T.,
Janadri
S.,
Anti-inflammatory effect of wedelolactone on DSS induced colitis in rats: IL-6/STAT3 signaling pathway. Journal of Ayurveda and Integrative Medicine.
2023;
14
(2)
:
100544
.
View Article Google Scholar -
Pesce
M.,
Seguella
L.,
Del Re
A.,
Lu
J.,
Palenca
I.,
Corpetti
C.,
Next-Generation Probiotics for Inflammatory Bowel Disease. International Journal of Molecular Sciences.
2022;
23
(10)
:
5466
.
View Article Google Scholar -
Vandenbroucke
K.,
de Haard
H.,
Beirnaert
E.,
Dreier
T.,
Lauwereys
M.,
Huyck
L.,
Orally administered L. lactis secreting an anti-TNF Nanobody demonstrate efficacy in chronic colitis. Mucosal Immunology.
2010;
3
(1)
:
49-56
.
View Article Google Scholar -
Breyner
N.M.,
Michon
C.,
de Sousa
C.S.,
Vilas Boas
P.B.,
Chain
F.,
Azevedo
V.A.,
Microbial Anti-Inflammatory Molecule (MAM) from Faecalibacterium prausnitzii Shows a Protective Effect on DNBS and DSS-Induced Colitis Model in Mice through Inhibition of NF-κB Pathway. Frontiers in Microbiology.
2017;
8
:
114
.
View Article Google Scholar -
Wei
P.,
Yang
Y.,
Ding
Q.,
Li
X.,
Sun
H.,
Liu
Z.,
Oral delivery of Bifidobacterium longum expressing α-melanocyte-stimulating hormone to combat ulcerative colitis. Journal of Medical Microbiology.
2016;
65
(2)
:
160-8
.
View Article Google Scholar -
Brzoska
T.,
Böhm
M.,
Lügering
A.,
Loser
K.,
Luger
T.A.,
Terminal signal: anti-inflammatory effects of α-melanocyte-stimulating hormone related peptides beyond the pharmacophore. Advances in Experimental Medicine and Biology.
2010;
681
:
107-16
.
View Article Google Scholar -
Singh
U.P.,
Singh
N.P.,
Busbee
B.,
Guan
H.,
Singh
B.,
Price
R.L.,
Alternative medicines as emerging therapies for inflammatory bowel diseases. International Reviews of Immunology.
2012;
31
(1)
:
66-84
.
View Article Google Scholar -
Shadnoush
M.,
Hosseini
R.S.,
Khalilnezhad
A.,
Navai
L.,
Goudarzi
H.,
Vaezjalali
M.,
Effects of probiotics on gut microbiota in patients with inflammatory bowel disease: a double-blind, placebo-controlled clinical trial. The Korean Journal of Gastroenterology.
2015;
65
(4)
:
215-21
.
View Article Google Scholar -
Berni Canani
R.,
De Filippis
F.,
Nocerino
R.,
Laiola
M.,
Paparo
L.,
Calignano
A.,
Specific signatures of the gut microbiota and increased levels of butyrate in children treated with fermented cow's milk containing heat-killed Lactobacillus paracasei CBA L74. Applied and Environmental Microbiology.
2017;
83
(19)
:
1206-17
.
View Article Google Scholar -
Li
C.,
Peng
K.,
Xiao
S.,
Long
Y.,
Yu
Q.,
The role of Lactobacillus in inflammatory bowel disease: from actualities to prospects. Cell Death Discovery.
2023;
9
(1)
:
361
.
View Article Google Scholar -
Stavropoulou
E.,
Bezirtzoglou
E.,
Probiotics in medicine a long debate. Frontiers in immunology.
2020;
11
:
2192
.
View Article Google Scholar -
Wasilewska
E.,
Zlotkowska
D.,
Wroblewska
B.,
Yogurt starter cultures of Streptococcus thermophilus and Lactobacillus bulgaricus ameliorate symptoms and modulate the immune response in a mouse model of dextran sulfate sodium-induced colitis. Journal of Dairy Science.
2019;
102
(1)
:
37-53
.
View Article Google Scholar -
Nagababu
E.,
Rifkind
J.M.,
Boindala
S.,
Nakka
L.,
Assessment of antioxidant activity of eugenol in vitro and in vivo. Methods in Molecular Biology (Clifton, N.J.).
2010;
610
:
165-80
.
View Article Google Scholar -
Aminabee
S.,
Rao
A.L.,
Eswaraiah
M.C.,
In Vivo Antioxidant Activity of Different Fractions of Indigofera Barberi Against Paracetamol-induced Toxicity in Rats. Turkish Journal of Pharmaceutical Sciences.
2020;
17
(2)
:
136-40
.
View Article Google Scholar -
Mitra
S.,
Ghosh
N.,
Paul
P.,
Banerjee
E.R.,
Orally Administered Fisetin Reduces the Symptoms of Acute Allergic Asthma in a Preclinical Mouse Model. Biomedical Research and Therapy.
2022;
9
(3)
:
4953-70
.
View Article Google Scholar -
Meers
G.K.,
Bohnenberger
H.,
Reichardt
H.M.,
Lühder
F.,
Reichardt
S.D.,
Impaired resolution of DSS-induced colitis in mice lacking the glucocorticoid receptor in myeloid cells. PLoS One.
2018;
13
(1)
:
e0190846
.
View Article Google Scholar -
Fijan
S.,
Microorganisms with claimed probiotic properties: an overview of recent literature. International Journal of Environmental Research and Public Health.
2014;
11
(5)
:
4745-67
.
View Article Google Scholar
Comments
Article Details
Volume & Issue : Vol 11 No 8 (2024)
Page No.: 6666-6682
Published on: 2024-08-31
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 2139 times
- PDF downloaded - 655 times
- XML downloaded - 64 times
 Biomedpress
Biomedpress














