Abstract
Introduction: The growth of endometrial tissue beyond the uterine cavity characterizes endometriosis, a chronic inflammatory disease. IL-17A, along with other immune mediators and a myriad of immune cells including Th17 lymphocytes and macrophages, are more prevalent in the peritoneal fluid of patients with endometriosis. IL–17A facilitates the implantation of endometrial tissue in ectopic locations by inducing growth factors and inflammatory cytokines from other immune cells. This work focuses on the levels of IL-17A gene expression in tissue samples from patient and control groups, as prior research has not extensively covered the gene expression of inflammatory mediators in tissue samples.
Methods: This study is both experimental and case-control in nature. Tissue samples were collected from 32 women undergoing laparoscopy at Yazd Shahid Sadoughi Hospital. The samples were divided into two groups: 16 patients with endometriosis and 16 healthy individuals. Within the patient group, there were two types of samples: eutopic and ectopic. The expression of IL-17A was assessed with RT-qPCR.
Results: RT-qPCR results showed a significant difference in IL-17A gene expression between ectopic lesions and eutopic samples from endometriosis patients (P value < 0.0001). Additionally, there was a significant increase in gene expression in eutopic samples from endometriosis patients compared to control samples (P value = 0.0176).
Conclusion: This study confirms the crucial involvement of inflammatory mediators like IL-17A in endometriosis through molecular findings in tissue samples, reflecting the elevated IL-17A content in the eutopic endometrium and ectopic lesions of endometriosis patients.
Introduction
Endometriosis is a gynecological condition affecting roughly 1 in 10 women of reproductive age, involving the abnormal growth of endometrial tissue outside the uterus. This condition can cause significant problems, including chronic pelvic pain, infertility, and impaired quality of life. While the exact cause of endometriosis is still under investigation, inflammation is emerging as a key factor1, 2, 3, 4. The prevailing theory is that retrograde menstruation, where endometrial fragments are deposited outside the uterus due to menstrual reflux, could trigger the disease. However, research suggests that there are likely other contributing factors. Studies have found elevated levels of inflammatory mediators such as interleukin (IL)-1beta, IL-6, and tumor necrosis factor (TNF)-alpha in women with endometriosis. This increased inflammatory response disrupts normal tissue function and can even promote the malignant transformation of ovarian endometriosis5, 6, 7, 8. Understanding the intricate relationship between endometriosis and inflammation is crucial for developing more effective diagnostic tools and treatment strategies, which could significantly improve the lives of women struggling with this complex disease.
Gene expression studies show promise in identifying specific molecules that influence disease variability. These molecules could serve as biomarkers for diagnosis and lead to the development of new drugs. Endometriosis likely results from a combination of factors. While hormonal influences, genetics, and environmental triggers are all suspected contributors, a growing body of evidence suggests the immune system might play a central role. Women with endometriosis frequently exhibit elevated levels of pro-inflammatory molecules called cytokines, including interleukin-17 (IL-17), in their blood, abdominal cavity fluid (peritoneal fluid), and misplaced endometrial tissues (ectopic lesions). This pro-inflammatory state creates a favorable environment for the displaced endometrial tissue, allowing it to evade the body's immune defenses and even promote its growth. However, the exact mechanisms through which immune cells, such as Th17 cells, contribute to this process are not yet clear9, 10, 11, 12. Given that IL-17A and inflammation are believed to play a crucial role in the development of endometriosis, we will measure the levels of IL-17A in both normal endometrial tissue (eutopic) and misplaced endometrial tissue (ectopic) of women with endometriosis. We will then compare these levels to those of a healthy control group.
Material and Methods
Tissue Specimen Collection
The samples were obtained from patients at Shahid Sadoughi Hospital who were candidates for laparoscopy and were divided into three sample groups: eutopic samples (from the patient's uterus) (n = 16), ectopic samples (from lesions) (n = 16), and control group (from the uterus of healthy women) (n = 16). For the control group, we included women undergoing laparoscopy who had not received any recent hormonal treatments.
RNA Extraction, cDNA Synthesis, RT-qPCR
Total RNA was extracted from the tissue samples using the Biofact kit for total RNA, following the manufacturer's instructions, and the quality of the products was verified with Nanodrop (260/280 absorbance ratio = 1.9-2.0). The total RNA samples were then converted into complementary DNA (cDNA) using a Biofact reverse transcriptase kit, in accordance with the manufacturer's guidelines. The purity of the resulting cDNA was assessed using a Nanodrop instrument, with an ideal ratio of absorbance values at 260 nm to 280 nm falling between 1.7 and 2.0 for pure cDNA. Next, IL-17A and a control gene (GAPDH) were amplified using RT-qPCR. Primers for these gene regions were designed and synthesized specifically for this study (Table 1). The PCR cycling conditions involved three steps, repeated for 40 cycles: Denaturation, briefly heating the samples to separate the DNA strands (95°C for 15 seconds), Annealing, lowering the temperature to allow primers to attach to specific sequences on the target DNA (58°C for 15 seconds), and Extension, raising the temperature again to allow DNA polymerase to synthesize new DNA strands complementary to the target sequence (72°C for 30 seconds).
Finally, the relative expression levels of the target genes were calculated using the 2-ΔΔCt method, which compares the threshold cycle (Ct) values of the target genes to the Ct value of the control gene (GAPDH). The Ct value indicates the cycle at which enough amplified DNA is present to be detected.
Statistical Analysis
This study employed a statistical analysis tool (software called SPSS 16.0) to compare the levels of IL-17A between different groups. A common method called one-way ANOVA was used to determine if the differences were statistically significant. In our analysis, we considered a result to be significant if the p-value was less than 0.05.
| Sequence (5-3) | Genes |
|---|---|
| F: CAGATTACTACAACCGATCCACC R: ACTTTGCCTCCCAGATCACA | IL-17A |
| F: CAAGAGCACAAGAGGAAGAGAGAG R: TCTACATGGCAACTGTGAGGAG | GAPDH |
Results
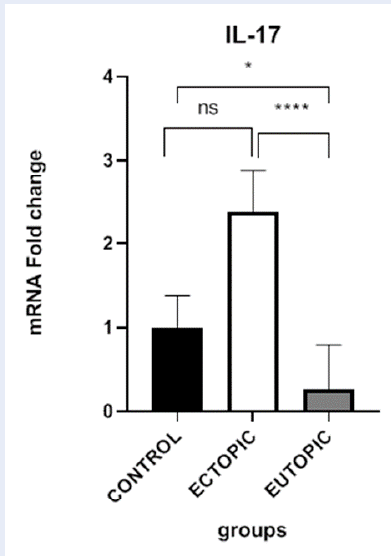
The average expression of IL-17A was significantly higher in ectopic samples than in eutopic samples (P-value < 0.0001). The expression of this gene was also significantly higher in eutopic samples from endometriosis patients than in control samples (P-value = 0.0176). However, there was no significant difference in the expression level of IL-17A between control and ectopic samples (P-value = 0.1531). The expression level of IL-17A in all tissues was normalized to GAPDH and compared between the control, eutopic, and ectopic groups (Figure 1).
Discussion
This study investigated the expression of IL-17A in the context of endometriosis. We found significantly higher levels of IL-17A in endometrial lesions (ectopic tissue) compared to healthy endometrial tissue (eutopic) from women with early-stage endometriosis. Interestingly, eutopic tissue from these patients also showed elevated IL-17A levels compared to healthy controls. However, there was no significant difference in IL-17A expression between healthy controls and the endometrial lesions (ectopic tissue).
The cause of endometriosis continues to puzzle scientists. While a definitive explanation for the development and progression of the disease remains elusive, several contributing factors are emerging. Misdirected tissue can lead to inflammation and the formation of blood vessels (angiogenesis), both of which contribute to the symptoms of the disease. Recent research has focused on the role of a particular molecule called interleukin-17A (IL-17A) in this process. Studies have shown that endometriotic lesions can produce varying amounts of IL-17A, with levels increasing with the severity of the disease13, 14, 15. Interestingly, these levels decrease significantly after the surgical removal of the lesions16, 17, 18, 19. In addition, IL-17A appears to stimulate the production of other molecules from endometrial cells that promote inflammation, cell growth, and the recruitment of immune cells9, 14, 20. This suggests that IL-17A may play a crucial role in both the development and progression of endometriosis. Since IL-17A is involved in several aspects of endometriosis, it represents a potential target for new treatment strategies. Researchers are looking for ways to block IL-17A production or its downstream effects. This could include regulating estrogen levels, inhibiting molecules that trigger IL-17A production, or promoting factors that counteract the influence of IL-17A. In addition, monitoring IL-17A levels could be a valuable tool for evaluating treatment efficacy21, 22, 23. More evidence is required to definitively say whether targeting IL-17A is an effective treatment for endometriosis, although early research shows promise.
There are different opinions about the amount of cytokines in different samples of affected women, but most confirm that the inflammatory microenvironment plays a vital role in endometriosis occurrence. Recent studies have shown that some inflammatory cytokines, such as IL-1, IL-6, IL-8, and TNF-α, increase in the peritoneal fluid of women with endometriosis24. A study of 51 women with endometriosis and 15 healthy women showed higher levels of IL-1, IL-6, and IL-17 in the affected women25. IL-17 is known as a pro-inflammatory Th17 cytokine, which is not only secreted by these lymphocytes but also by neutrophils and mast cells26, 27. IL-17A plays an important role in inflammation due to the induction of other immune cells, such as neutrophils28, 29. IL-17 can fulfill various immunological functions. In addition to protecting the immune system, IL-17 is considered an immunopathological factor in autoimmunity, cancer, and chronic inflammation30. Zhang and his colleagues were the first, and later other teams, to prove that the IL-17 concentration in PF from patients with early-stage endometriosis is higher than in patients with late-stage endometriosis and in healthy women31, 32. These results may be due to the fact that immune responses are lower in the late stage than in the early stage, as Salmeri et al. suggested in their study33. In Ahn's study, too, immunohistochemistry showed that IL-17-positive cells were localized in eutopic and ectopic areas of the affected women. This study also showed that the removal of ectopic lesions leads to a reduction in IL-17 levels. This work reported that IL-17A promotes the implantation and growth of ectopic lesions and is of greater importance in the early stages of endometriosis34.
Another role reported for IL-17A is increasing the secretion of IL-1β by macrophages. IL-1β can stimulate the production of IL-8 and vascular endothelial growth factor (VEGF)35, 36. Thus, higher levels of IL-17A in patients with endometriosis may imply proliferation, hypervascularization and likely facilitation of implantation37. Some reports have also shown that the level of inflammatory cytokines and the ratio between inflammatory and anti-inflammatory cytokines in the serum and peritoneal fluid of affected women is higher than in healthy women38. A study of 30 affected and 30 healthy women showed that the IL-17/IL-23 ratio in the serum sample, the TNF-α/IL-10 ratio, and the IL-17/IL-10 ratio in the peritoneal fluid of the patients were higher than in the control group39.
IL-17A, an important pro-inflammatory cytokine produced by various immune cells, has been extensively studied for its role in chronic inflammatory diseases, such as rheumatoid arthritis and psoriasis40, 41. In early research, IL-17A was found to stimulate inflammatory factors and cell proliferation in endometrial stromal cells14, 42. Interestingly, IL-17A levels in the peritoneal fluid of women with endometriosis correlate with disease severity and infertility, suggesting a possible link to the pathogenesis of endometriosis. This link is further supported by the presence of IL-17A in the fluid of endometrial cysts and the association with more severe cases of endometriosis. While recent studies highlight the role of IL-17A in triggering pro-inflammatory factors and blood vessel growth in endometriosis, further research is needed to fully understand its effects on cell survival and immune cell interactions in this disease.
This study highlights some limitations inherent in endometriosis research in general. These limitations may hinder the accurate interpretation and comparison of data from different studies. Ideally, tissue samples from patients and control groups would closely match in several aspects: menstrual phase (the phase of a woman's menstrual cycle may influence the results); disease stage (different stages of endometriosis (early vs. advanced) are likely to have different IL-17A expression levels); age (age could also play a role in IL-17A production associated with the disease); medical history and treatment (previous medications and medical conditions, especially those that affect hormone levels (estrogen and progesterone), could influence IL-17A levels. The inherent complexity of endometriosis presents a further difficulty. The disease occurs in different stages and phenotypes, making it difficult to create a single, representative model for study. In addition, spontaneous endometriosis is mainly observed in humans and other old-world primates, limiting the usefulness of conventional animal models for studying the disease. The reliance on surgery for both diagnosis and staging of endometriosis presents an obstacle to studying the disease in its early stages and to identifying true control subjects without the disease. This reliance on surgery makes it difficult to obtain tissue samples from healthy individuals for comparative purposes. Because of these limitations, our understanding of endometriosis remains relatively basic, and current classifications likely don't fully capture the biological diversity of the disease. To gain a deeper understanding of the specific role(s) of IL-17A in the pathogenesis of endometriosis, future research must address these limitations. This could include more precise sampling techniques; techniques for obtaining tissue samples from patients at specific stages of their menstrual cycle and disease progression; consideration of individual factors: careful consideration of a patient's medical history and ongoing treatments when analyzing data; alternative models: exploring alternative research models beyond traditional animals, potentially including cell cultures or induced pluripotent stem cells (iPSCs). By overcoming these challenges, researchers can develop a more nuanced understanding of how IL-17A contributes to endometriosenosis and potentially pave the way for targeted treatment strategies.
It was found that the expression of the inflammatory cytokine IL-17A was significantly higher at the ectopic site than in eutopic tissue. The concentration of this cytokine was also higher at the eutopic site than in control samples, but there was no significant difference between ectopic lesions and control samples. Like previous studies, these results also showed the importance of inflammatory factors in the pathogenesis of endometriosis. The current study confirms the central role of mononuclear immune cells and inflammatory mediators in endometriosis by molecular findings in tissue sections.
Conclusion
In summary, it can be said that endometriosis is a complex disease with a multi-layered etiology. Understanding the intricate interplay between hormones, the immune system, and other factors is crucial for the development of effective diagnostic tools and treatments. The increase in inflammatory cells and mediators in the peritoneal fluid and serum samples from endometriosis patients has been demonstrated in previous studies. However, most of these studies did not focus on tissue samples and molecular findings. Based on the findings, the expression of IL-17A is significantly higher in ectopic lesions than in eutopic tissues and higher in the eutopic endometrium of endometriosis patients than in the control group. With regard to our results, we can confirm the important role of mononuclear immune cells and inflammatory mediators in endometriosis by molecular findings in tissue samples.
Abbreviations
ANOVA - Analysis of Variancec, DNA - Complementary DNA, Ct - Threshold Cycle, GAPDH - Glyceraldehyde 3-phosphate dehydrogenase, IL - Interleukin, IL-17A - Interleukin 17A, iPSCs - Induced Pluripotent Stem Cells, mRNA - messenger RNA, PCR - Polymerase Chain Reaction, RT-qPCR - Reverse Transcription quantitative Polymerase Chain Reaction, SPSS - Statistical Package for the Social Sciences, Th17 - T helper 17, TNF-alpha - Tumor Necrosis Factor-alpha, VEGF - Vascular Endothelial Growth Factor
Acknowledgments
The authors would like to acknowledge the central lab of the international campus of Shahid Sadoughi University of medical sciences.
Author’s contributions
All authors significantly contributed to this work, read and approved the final manuscript.
Funding
None.
Availability of data and materials
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
This study recruited 32 premenopausal women undergoing laparoscopy for gynecological reasons at Shahid Sadoughi Hospital. All participants provided written informed consent before being enrolled. The study received approval from the Medical Ethics Committee of Yazd University of Medical Sciences (approval number: IR. SSU. MEDICINE. REC. 1398. 117).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
-
Lamceva
J.,
Uljanovs
R.,
Strumfa
I.,
The main theories on the pathogenesis of endometriosis. International Journal of Molecular Sciences.
2023;
24
(5)
:
4254
.
View Article PubMed Google Scholar -
Allaire
C.,
Bedaiwy
M.A.,
Yong
P.J.,
Diagnosis and management of endometriosis. Canadian Medical Association Journal.
2023;
195
(10)
:
363-71
.
View Article PubMed Google Scholar -
Rahmioglu
N.,
Mortlock
S.,
Ghiasi
M.,
M∅ller
P.L.,
Stefansdottir
L.,
Galarneau
G.,
Genomic Consortium
DBDS,
FinnGen Study
FinnGen Endometriosis Taskforce
Celmatix Research Team
23andMe Research Team
The genetic basis of endometriosis and comorbidity with other pain and inflammatory conditions. Nature Genetics.
2023;
55
(3)
:
423-36
.
View Article PubMed Google Scholar -
Nazari
M.,
Babakhanzadeh
E.,
Mohsen Aghaei Zarch
S.,
Talebi
M.,
Narimani
N.,
Dargahi
M.,
Upregulation of the RNF8 gene can predict the presence of sperm in azoospermic individuals. Clinical and Experimental Reproductive Medicine.
2020;
47
(1)
:
61-7
.
View Article PubMed Google Scholar -
Appazova
L.,
Akhmaltdinova
L.,
Turdybekova
Y.,
Kopobayeva
I.,
Amirbekova
Z.,
Determination of cytokines for assessment of inflammatory status at genital endometriosis. Bangladesh Journal of Medical Science.
2024;
23
(1)
:
121-9
.
View Article Google Scholar -
Sutrisno
S.,
Destikatari
L.,
Genistein Effect on Estrogen Receptor especially Interleukin-6 and Tumor Necrosis Factor-α in Mice Model of Endometriosis: a Systematic Review and Meta-analysis. Asian Journal of Health Research.
2023;
2
(1)
:
96-104
.
View Article Google Scholar -
Werdel
R.,
Mabie
A.,
Evans
T.L.,
Coté
R.D.,
Schlundt
A.,
Doehrman
P.,
Serum Levels of Interleukins in Endometriosis Patients: A Systematic Review and Meta-analysis. Journal of Minimally Invasive Gynecology.
2024;
31
(5)
:
387-396.e11
.
View Article PubMed Google Scholar -
Khodadadian
A.,
Varghaiyan
Y.,
Babakhanzadeh
E.,
Alipourfard
I.,
Haghi-Daredeh
S.,
Ghobadi
A.,
Fertility preservation in women with ovarian cancer: Finding new pathways: A case-control study. International Journal of Reproductive Biomedicine (Yazd, Iran).
2021;
19
(2)
:
157-66
.
PubMed Google Scholar -
Tanaka
S.,
Sawachika
M.,
Yoshida
N.,
Futani
K.,
Murata
H.,
Okada
H.,
IL17A Suppresses IGFBP1 in Human Endometrial Stromal Cells. Reproductive Medicine (Basel, Switzerland).
2024;
5
(2)
:
43-56
.
View Article Google Scholar -
Prastiwi
A.P.,
L.R. Ariana,
G.A. Salsabila,
S. Sutrisno,
N. Nurdiana,
N. Setijowati,
I.W. Indrawan,
Potential Of Mahkota Dewa Fruit (Phaleria macrocarpa) Flavonoids Extract and Dienogest to Reduce IL-17A Levels in Mice Endometriosis Model. Medical Laboratory Technology Journal.
2023;
9
(2)
:
184-192
.
View Article Google Scholar -
Salsabila
G.A.,
A.P. Prastiwi,
L.R. Ariana,
S. Sutrisno,
N.S. Nurdiana,
I.W. Indrawan,
D.S. Wardani,
The Effect of Flavonoid from Extract of Phaleria Macrocarpa on Levels Il-2, Il-17a and Il-33 in Endometriosis Mice. Journal of Advanced Zoology.
2023;
44
(3)
:
841-845
.
-
Babakhanzadeh
E.,
Danaei
H.,
Abedinzadeh
M.,
Ashrafzadeh
H.R.,
Ghasemi
N.,
Association of miR-146a and miR196a2 genotype with susceptibility to idiopathic recurrent pregnancy loss in Iranian women: A case-control study. International Journal of Reproductive Biomedicine (Yazd, Iran).
2021;
19
(8)
:
725-32
.
View Article PubMed Google Scholar -
Cheng
R.,
Xue
X.,
Liu
X.,
Expression of IL17A in endometrial carcinoma and effects of IL17A on biological behaviour in Ishikawa cells. The Journal of International Medical Research.
2020;
48
(9)
:
300060520950563
.
View Article PubMed Google Scholar -
Crosby
D.A.,
Glover
L.E.,
Brennan
E.P.,
Kelly
P.,
Cormican
P.,
Moran
B.,
Dysregulation of the interleukin-17A pathway in endometrial tissue from women with unexplained infertility affects pregnancy outcome following assisted reproductive treatment. Human Reproduction (Oxford, England).
2020;
35
(8)
:
1875-88
.
View Article PubMed Google Scholar -
Sisnett
D.J.,
Zutautas
K.B.,
Miller
J.E.,
Lingegowda
H.,
Ahn
S.H.,
McCallion
A.,
The dysregulated IL-23/TH17 axis in endometriosis pathophysiology. The Journal of Immunology : Official Journal of the American Association of Immunologists.
2024;
212
(9)
:
1428-41
.
View Article PubMed Google Scholar -
International working group of AAGL, ESGE, ESHRE and WES, Tomassetti C, Johnson NP, Petrozza J, Abrao MS, Einarsson JI, Horne AW, Lee TT, Missmer S, Vermeulen N, Zondervan KT. An international terminology for endometriosis, 2021. Human Reproduction Open.
2021;
2021
(4)
:
hoab029
.
-
Horne
A.W.,
Missmer
S.A.,
Pathophysiology, diagnosis, and management of endometriosis. BMJ.
2022;
2022
:
379
.
View Article Google Scholar -
Lee
S.Y.,
Koo
Y.J.,
Lee
D.H.,
Classification of endometriosis. Yeungnam University Journal of Medicine.
2021;
38
(1)
:
10-8
.
View Article PubMed Google Scholar -
Nazari
M.,
Khorshidian
A.,
Alizadeh
S.,
Falahati
A.M.,
Haghparast
A.,
Ghasemifar
S.,
Association between peroxisome proliferator activated receptor gamma coactivator 1 gene with overweight and obesity risk: case\textendashcontrol study and meta-analysis. Human Gene.
2022;
34
:
201123
.
View Article Google Scholar -
You
Y.,
Stelzl
P.,
Joseph
D.N.,
Aldo
P.B.,
Maxwell
A.J.,
Dekel
N.,
TNF-α regulated endometrial stroma secretome promotes trophoblast invasion. Frontiers in Immunology.
2021;
12
:
737401
.
View Article PubMed Google Scholar -
Miller
J.E.,
Ahn
S.H.,
Marks
R.M.,
Monsanto
S.P.,
Fazleabas
A.T.,
Koti
M.,
IL-17A modulates peritoneal macrophage recruitment and M2 polarization in endometriosis. Frontiers in Immunology.
2020;
11
:
108
.
View Article PubMed Google Scholar -
Shi
J.L.,
Zheng
Z.M.,
Chen
M.,
Shen
H.H.,
Li
M.Q.,
Shao
J.,
IL-17: an important pathogenic factor in endometriosis. International Journal of Medical Sciences.
2022;
19
(4)
:
769-78
.
View Article PubMed Google Scholar -
Jahangir
M.,
Nazari
M.,
Babakhanzadeh
E.,
Manshadi
S.D.,
Where do obesity and male infertility collide?. BMC Medical Genomics.
2024;
17
(1)
:
128
.
View Article PubMed Google Scholar -
Giudice
L.C.,
Kao
L.C.,
Endometriosis. Lancet.
2004;
364
(9447)
:
1789-99
.
View Article PubMed Google Scholar -
Sikora
J.,
Smycz-Kubańska
M.,
Mielczarek-Palacz
A.,
Bednarek
I.,
Kondera-Anasz
Z.,
The involvement of multifunctional TGF-β and related cytokines in pathogenesis of endometriosis. Immunology Letters.
2018;
201
:
31-7
.
View Article PubMed Google Scholar -
Cua
D.J.,
Tato
C.M.,
Innate IL-17-producing cells: the sentinels of the immune system. Nature Reviews. Immunology.
2010;
10
(7)
:
479-89
.
View Article PubMed Google Scholar -
Gogacz
M.,
Winkler
I.,
Bojarska-Junak
A.,
Tabarkiewicz
J.,
Semczuk
A.,
Rechberger
T.,
Increased percentage of Th17 cells in peritoneal fluid is associated with severity of endometriosis. Journal of Reproductive Immunology.
2016;
117
:
39-44
.
PubMed Google Scholar -
Iwakura
Y.,
Ishigame
H.,
Saijo
S.,
Nakae
S.,
Functional specialization of interleukin-17 family members. Immunity.
2011;
34
(2)
:
149-62
.
PubMed Google Scholar -
Kolls
J.K.,
Lindén
A.,
Interleukin-17 family members and inflammation. Immunity.
2004;
21
(4)
:
467-76
.
PubMed Google Scholar -
Noack
M.,
Miossec
P.,
Th17 and regulatory T cell balance in autoimmune and inflammatory diseases. Autoimmunity Reviews.
2014;
13
(6)
:
668-77
.
PubMed Google Scholar -
Zhang
X.,
Xu
H.,
Lin
J.,
Qian
Y.,
Deng
L.,
Peritoneal fluid concentrations of interleukin-17 correlate with the severity of endometriosis and infertility of this disorder. BJOG.
2005;
112
(8)
:
1153-5
.
PubMed Google Scholar -
Omwandho
C.O.,
Konrad
L.,
Halis
G.,
Oehmke
F.,
Tinneberg
H.R.,
Role of TGF-betas in normal human endometrium and endometriosis. Human Reproduction (Oxford, England).
2010;
25
(1)
:
101-9
.
View Article PubMed Google Scholar -
Salmeri
F.M.,
Laganà
A.S.,
Sofo
V.,
Triolo
O.,
Sturlese
E.,
Retto
G.,
Behavior of tumor necrosis factor-α and tumor necrosis factor receptor 1/tumor necrosis factor receptor 2 system in mononuclear cells recovered from peritoneal fluid of women with endometriosis at different stages. Reproductive Sciences (Thousand Oaks, Calif.).
2015;
22
(2)
:
165-72
.
PubMed Google Scholar -
Ahn
S.H.,
Edwards
A.K.,
Singh
S.S.,
Young
S.L.,
Lessey
B.A.,
Tayade
C.,
IL-17A Contributes to the Pathogenesis of Endometriosis by Triggering Proinflammatory Cytokines and Angiogenic Growth Factors. The Journal of Immunology : Official Journal of the American Association of Immunologists.
2015;
195
(6)
:
2591-600
.
View Article PubMed Google Scholar -
Xavier
T.,
Pallikara
S.,
Saji
N.,
Radhakrishnan
N.,
Menon
K.N.,
Pillai
G.S.,
Significance of monitoring vascular endothelial growth factor, monocyte chemoattractant protein-1 and Interleukin-8 in diabetic macular edema towards early identification of nonresponders to ranibizumab therapy. Indian Journal of Ophthalmology.
2021;
69
(6)
:
1475-81
.
PubMed Google Scholar -
Koh
S.M.,
Chan
C.K.,
Teo
S.H.,
Singh
S.,
Merican
A.,
Ng
W.M.,
Elevated plasma and synovial fluid interleukin-8 and interleukin-18 may be associated with the pathogenesis of knee osteoarthritis. The Knee.
2020;
27
(1)
:
26-35
.
View Article PubMed Google Scholar -
Shi
J.L.,
Zheng
Z.M.,
Chen
M.,
Shen
H.H.,
Li
M.Q.,
Shao
J.,
IL-17: an important pathogenic factor in endometriosis. International Journal of Medical Sciences.
2022;
19
(4)
:
769-78
.
View Article PubMed Google Scholar -
Rafi
U.,
Ahmad
S.,
S.S. Bokhari,
M.A. Iqbal,
A. Zia,
M.A. Khan,
N. Roohi,
Association of inflammatory markers/cytokines with cardiovascular risk manifestation in patients with endometriosis. Mediators of Inflammation.
2021;
2021
(1)
:
3425560
.
View Article PubMed Google Scholar -
Tarokh
M.,
Ghaffari Novin
M.,
Poordast
T.,
Tavana
Z.,
Nazarian
H.,
Norouzian
M.,
Serum and Peritoneal Fluid Cytokine Profiles in Infertile Women with Endometriosis. Iranian Journal of Immunology.
2019;
16
(2)
:
151-62
.
PubMed Google Scholar -
Kouri
V.P.,
Olkkonen
J.,
Nurmi
K.,
Peled
N.,
Ainola
M.,
Mandelin
J.,
IL-17A and TNF synergistically drive expression of proinflammatory mediators in synovial fibroblasts via IκBζ-dependent induction of ELF3. Rheumatology (Oxford, England).
2023;
62
(2)
:
872-85
.
View Article PubMed Google Scholar -
Liu
M.,
Wu
K.,
Lin
J.,
Xie
Q.,
Liu
Y.,
Huang
Y.,
Emerging biological functions of IL-17A: a new target in chronic obstructive pulmonary disease?. Frontiers in Pharmacology.
2021;
12
:
695957
.
View Article PubMed Google Scholar -
Salsabila
G.A.,
A.P. Prastiwi,
L.R. Ariana,
S. Sutrisno,
N.S. Nurdiana,
I.W. Indrawan,
D.S. Wardani,
The Effect of Flavonoid from Extract of Phaleria Macrocarpa on Levels Il-2, Il-17a and Il-33 in Endometriosis Mice. Journal of Advanced Zoology.
2023;
44
(3)
:
841-845
.
View Article Google Scholar
Comments
Article Details
Volume & Issue : Vol 11 No 6 (2024)
Page No.: 6482-6487
Published on: 2024-06-30
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 2801 times
- PDF downloaded - 917 times
- XML downloaded - 103 times
 Biomedpress
Biomedpress


