Abstract
Stem cell therapy is promising for treatment of degenerative diseases. In Vietnam, stem cell applications have been performed since the 1990s. In addition to hematopoietic stem cell transplantation for malignant hematologic diseases and disorders, mesenchymal stem cells have also been clinically approved for treatment of diseases such as knee osteoarthritis, chronic obstructive pulmonary disease, autism, cerebral palsy and more in recent years. Unlike countries that only permit use of non-expanded stem cells, the Vietnamese government has permitted use of both non-expanded and expanded stem cells for both local and systemic transfusion in some diseases. After 20 years of stem cell development, the market has finally established stem cell banks and some stem cell clinical services. Although some regulations or guidelines regarding stem cell applications have yet to be published by the government, present breakthroughs in stem cell transplantation may facilitate Vietnam’s recognition as a key player in stem cell application in Asia and, in the near future, the world.
Stem cells and stem cell therapy
Stem cells are unspecific cells endowed with two important properties: self-renewal and differentiation. The self-renewal process allows cells to proliferate for an indefinite period of time, while differentiation permits cells to become specialized. With great potential to treat numerous diseases, stem cells have rapidly become an essential pillar of health science.
Although several different kinds of stem cells have been discovered, almost all can be characterized into two groups: embryonic stem cells (ESCs), which are pluripotent; and adult stem cells (ASCs), which are multipotent. ESCs can differentiate into all types of cells found in the adult human body; therefore, they are a promising source of cells for therapeutics. However, these cells are obtained from blastocysts and this fact raises ethical issues that may diminish clinical applications of ESCs. More recently, ESCs are frequently being replaced by induced pluripotent stem cells (iPSCs), which are adult-derived stem cells possessing pluripotency similar to ESCs. First reported by Yamanaka et al. in 2006, iPSCs were initially produced by reprogramming fibroblasts using four defined factors: Oct-3/4, Klf4, C-myc and Sox-2 Takahashi and Yamanaka, 2006. This technology has opened a new age for stem cell and regenerative medicine research. Recently, iPSCs have been used in a clinical setting for the first time.
Unlike ESCs, ASCs and iPSCs are not confounded by ethical issues, which can delay or prohibit clinical applications. If we consider transplantation of hematopoietic stem cells (HSCs) as the first application of ASCs, therapeutic use of ASCs has a history extending back more than half a century. Indeed, the first bone marrow transfusion took place in 1939 for a patient with aplastic anemia Osgood et al., 1939 and the first allogenic hematopoietic stem cell transplant (HSCT) was performed in 1957 for patients treated with radiation and chemotherapy Thomas et al., 1957. To date, ASCs have been clinically applied in the form of hematopoetic stem cells (HSCs), mesenchymal stem cells (MSCs), limbal stem cells (LSCs), epidermal stem cells and more.
Since 2000, remarkable developments in stem cell research and therapy have facilitated significant control over isolation, proliferation and differentiation of stem cells. Establishment of guidelines for Good Manufacturing Practice (GMP)-compliant stem cell culture and clinical applications in 2010 spurred the interest of hundreds of stem cell laboratories all over the world. Several products previously approved as medical devices have been applied to isolate, proliferate and differentiate stem cells for clinical applications. In addition, various defined culture media, serum-free media and autologous serumsupplemented media have also been invented to fulfill clinical regulations. Such products and technologies have significantly contributed to clinical applications of stem cells.
Recent approval of a few stem cell-based drugs has dramatically increased research into new clinical applications for stem cells. In many countries, both HSCs and MSCs are routinely used to treat certain hematological diseases. New stem cell therapies will not be limited to hematological disorders and, instead, can be used to mitigate autoimmune disease, repair degenerative cartilage, regenerate bone, replace damaged or lost neural tissue, and even treat solid cancers.
History of stem cell transplantation in vietnam
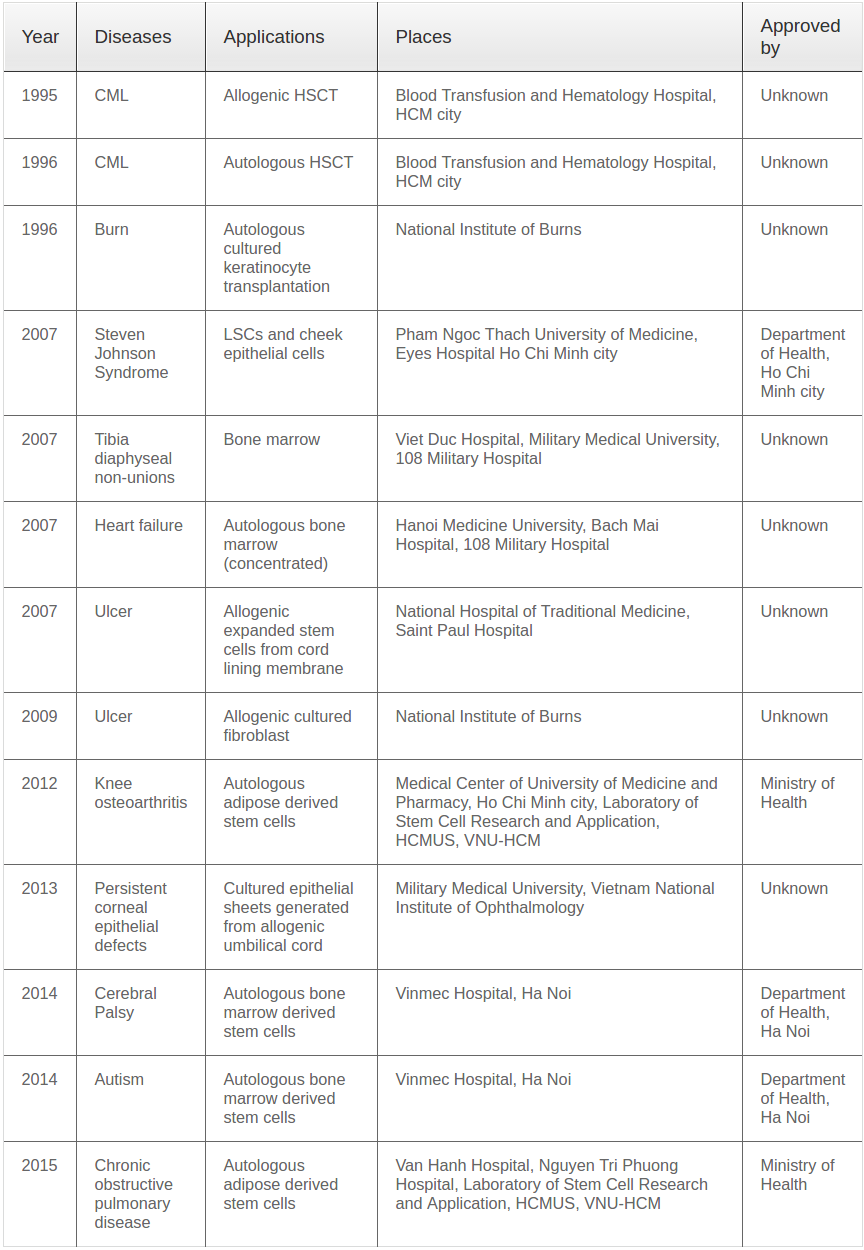
In 1995, the first HSCT was performed in Vietnam at the Blood Transfusion and Hematology Hospital (BTH) in Ho Chi Minh City. The procedure incorporated transfusion of HSCs into a 26-year-old male patient with chronic myelogenous leukemia; this patient is still alive and well van Be et al., 2008. In 1996, studies performed in Northern Vietnam used cultured keratinocytes to treat burn patients. Although these studies did not use pluripotent stem cells, they significantly contributed to the promotion of stem cell culture and research in Northern Vietnam, which hosts the first Vietnamese unit to use cultured cells for clinical applications. That same year, the first case of HSCT using autologous HSCs was performed at BTH, which was also the first facility to use HSCs from umbilical cord blood (UCB) to treat hematological disease in 2002.
From 2002 to 2007, translational studies investigating culture, isolation and proliferation of stem cells were performed in Vietnamese laboratories, especially within the first established stem cell laboratory, named the Laboratory of Stem Cell Research and Application (SCL), located at the University of Science at Vietnam National University in Ho Chi Minh City. In 2007, this laboratory collaborated with a group at Pham Ngoc Thach University to examine cultured cheek epithelial cell and LSC transplantation for Stevens–Johnson syndrome at Eyes Hospital, also in Ho Chi Minh City. Meanwhile, in Hanoi, stem cell scientists at Hanoi Medicine University, Bach Mai Hospital and 108 Military Hospital were using autologous concentrated bone marrow to treat heart failure and some orthopedic disorders.
The first clinical application of expanded allogenic stem cells in Vietnam was reported in a clinical study for wound healing by Mai et al. in 2007 Mai Manh Tuan, 2007. The authors used allogenic transplantation of cultured MSCs isolated from the membrane lining of an umbilical cord to treat chronic ulcers in a group of diabetic patients ( Table 1 ).
In recent years, interest has grown in adipose tissuederived stem cell (ADSC) transplantation. In 2012, the first clinical trial using non-expanded ADSCs was approved and performed in collaboration with SCL at the Medical Center at the University of Medicine and Pharmacy in Ho Chi Minh City. The following year, the Ministry of Health approved the first study using ex vivo-cultured ADSC transplantation, a novel treatment for spinal cord injury. This clinical trial was also the first to examine transfusion of ADSCs directly into veins; in 2015, this transfusion method was approved by the Ministry of Health for treatment of chronic obstructive pulmonary disease (COPD). In 2014, two clinical trials at Vinmec Hospital examined the potential of autologous concentrated bone marrow to treat pediatric diseases including cerebral palsy and autism
Hematologic malignancies and disorders
Stem cell transplantation for malignant hematologic diseases and disorders have been performed in Vietnam since 1995 van Be et al., 2008. Transplantable HSCs from a variety of sources are available for patients. After 20 years of HSCT for hematologic disorders, eight HSCT centers have opened in Vietnam (each during the indicated year) including BTH (1995), Hue Central Hospital (2003), 108 Military Hospital (2004), Pediatric Institute (2006), Cho Ray Hospital (2013), Bach Mai Hospital (2013) and Nghe An Oncology Hospital (2013). In 2015, around 500 patients were treated with HSCT for diseases such as acute myeloid leukemia Tran Quoc Tuan, 2011a, bvan Be et al., 2008, acute lymphocytic leukemia van Be et al., 2008, thalassemia Duong Ba Truc, 2013, chronic myeloid leukemia van Be et al., 2008, chronic myelomonocytic leukemia van Be et al., 2008, multiple myeloma Huynh Duc-Vinh Phu, 2013Nguyen Thi-Thu Ha, 2010, myelodysplastic syndromes Huynh Duc Vinh Phu, 2013, lymphoma Nguyen Thi-Thu Ha, 2010 and aplastic anemia Bach Quoc Khanh, 2012.
Although clinics used HSCs from peripheral blood, UCB and bone marrow, nearly 90% of all cases involved transplantation of peripheral blood-derived HSCs. To date, no statistical examination of treatment efficacy between different centers in Vietnam has been reported. However, according to an institutional report published by BTH, after autologous HSC transplantation about 54.50% of patients relapsed and 45.45%remained alive. Overall survival of patients was 5.74 ± 0.82 years, while disease-free survival time was 5.48 ± 0.92 years. Importantly, statistically significant differences in overall and disease-free survival were not observed between allogenic and autologous HSCT procedures (P > 0.05) van Be et al., 2008.
Orthopedic disorders
Orthopedic disorders represent a large group of conditions and diseases related to bone or cartilage. With an aging population in many countries, the number of patients with these disorders is significantly increasing. Common diagnoses include osteoarthritis, degenerative cartilage and femoral head necrosis; in Vietnam, there has been a strong increase in these disorders during recent years. Thus, in addition to routine treatment techniques, application of stem cells have been investigated for treatment of common orthopedic diseases and disorders since 2007.
The first orthopedic stem cell transplant in Vietnam was recorded in 2007 in a study investigating bone marrow to treat tibia diaphyseal non-unions Nguyen Manh Khanh, 2007. Since this time, orthopedic applications of stem cells have expanded significantly; yet, almost all clinical applications make use of autologous bone marrow including treatments for femoral head necrosis Mai Dac Viet, 2010Nguyen Thi-Thu Ha, 2013, bone defect and short limb Nguyen Van Luong, 2010, tibia non-unions Nguyen Manh Khanh, 2010, long bone non-union Le Van Doan, 2011Nguyen Thi-Thu Ha, 2013Thua et al., 2015a, long bone diaphyseal non-unions Ngo Ba Toan, 2012, 2014, joint diseases Duong Dinh Toan, 2013aLy Tuan Khai, 2013Nguyen Thanh Binh, 2012, limb lengthening Nguyen Thi-Thu Ha, 2013, knee osteoarthritis Duong Dinh Toan, 2013bDuong Dinh Toan, 2014 and delayed or non-union bone Thua et al., 2015b. The common property of these studies is use of non-expanded autologous stem cells; each used concentrated autologous bone marrow generated using a centrifuge-based method. Almost all studies showed that bone marrow-derived stem cells (BMSCs) provide many benefits, making them capable of improving a range of orthopedic disorders. Notably, all of these results relied upon a manual procedure to enrich stem cells from bone marrow.
More recently, clinical studies have begun to another source of stem cells, ADSCs, for treatment some diseases of bone and cartilage. In 2013, the first application of ADSCs (in the form of non-expanded ADSCs, the so-called stromal vascular fraction – SVF) was performed by Bui et al. in collaboration with SCL at the Medical Center at the University of Medicine and Pharmacy in Ho Chi Minh City Pham et al., 2014. In this study, mixtures of SVF and platelet rich plasma (PRP) were directly injected into patients with grade II and III knee osteoarthritis after microfracture. Initial results indicated this method could improve both pain levels and life quality of patients after 6 months Pham et al., 2014. At this point, other hospitals in Hanoi Pham Hoai Thu, 2014 and Ho Chi Minh City (https://clinicaltrials.gov/ct2/show/NCT02142842?term=NCT02142842&rank=1) also began using this technique to treat osteoarthritis of the knee. Collaboration between three organizations (115 Hospital, Van Hanh General Hospital and SCL) resulted in the first study comparing efficacy of SVF transplantation to treat osteoarthritis with or without arthroscopic microfracture (results in press). After 18 months of monitoring, results of this study showed SVF injections combined with arthroscopic microfracture significantly improved grade II and III knee osteoarthritis compared with arthroscopic microfracture alone. In a small pilot study, SVF and PRP were also used to treat femoral head necrosis; however, these results have not been published.
Wounds and ulcers
Application of cell therapy to treatment of wounds and ulcers began almost two decades ago. In 1998, Le et al. carried out the first study, which examined the treatment of burns with grafts of cultured keratinocytes Le The Trung, 1998. Soon after, cultured fibroblasts were used in similar studies, including autologous cultured fibroblasts for radiation wound treatment from 2005 to 2010 Dinh Van Han, 2006Luong, 2010 and allogenic cultured fibroblast grafts for partial thickness burn wound treatment in 2007 Nguyen Hai An, 2007.
The first application of stem cells for wound treatment in Vietnam was reported by Mai et al. in 2007 Mai Manh Tuan, 2007. In this study, performed in collaboration with Dr. Phan from the National University of Singapore, patients with skin wounds were treated with an allogenic transplantation of cultured MSCs isolated from the membrane lining of an umbilical cord Mai Manh Tuan, 2007. This was also the first clinical study to use expanded allogenic stem cells in Vietnam.
Recently, diabetic foot ulcers have emerged as a potential indication for stem cell therapy. The first clinical study, reported by Tran et al. in 2014, used non-expanded autologous ADSCs in combination with PRP to treat diabetic foot ulcers Tran et al., 2014. The promising results of this study have significantly increased clinical application of this technique in hospitals in Ho Chi Minh City and Hanoi.
Ophthalmological diseases
While Penetrating Keratoplasty, replacement of the host cornea with a donor cornea, has been used to treat corneal diseases in Vietnam since the 1950s, this technique has only been widely applied since the millennium Pham Ngoc Dong, 2007, 2006. From 2002 to 2011, 1,016 patients received corneal transplants at the Vietnam National Institute of Ophthalmology. Corneal transplantation was used in 53.4% of patients with corneal ulcers, 18.3% with corneal scar, 9.4% with corneal dystrophy, 6.5% with failure graft, 4.1% with pseudophakic bullous keratopathy, and 4% with corneal burn Pham Ngoc Dong, 2013.
Though successful, this technique has recently been replaced by amniotic membrane transplantation because of low availability of donated cornea. The first report describing long term results of trabeculectomy with amniotic membrane transplantation for treatment of recurrent glaucoma was published in 2011 Thuy,2011. Although amniotic membrane is a rich source, it does not contain viable epithelial cells or LSCs; therefore, the efficacy of this treatment can be low in cases requiring corneal regeneration.
The first efforts to culture and expand ex vivo LSCs and cheek epithelial cells to treat corneal epithelium disease were reported by a collaborative group (Pham Ngoc Thach University of Medicine, University of Science, Vietnam National University and Eyes Hospital) in September 2007. This group also examined use of these cultured cells in patients with Stevens–Johnson syndrome or conjunctivitis. Initial results showed transplantation of both cheek epithelial cells and LSCs improved patients’ vision Tran Cong Toai, 2011. Similar techniques were also performed to produce corneal epithelial sheets from LSCs to treat unilateral LSC deficiency syndrome Do Thuy Huong, 2009Vu Thi-Tue Khanh, 2009Vu Thi- Tue Khanh, 2012.
In addition to cultured LSCs, cultured epithelial sheets generated from allogenic umbilical cord have also been applied to persistent corneal epithelial defects in a collaborative study with the National University of Singapore (NUS) Nguyen Dinh Ngan, 2013. Cultured epithelial sheets from umbilical cord were prepared at NUS and transferred to Vietnam. Thirtyone patients took part in this research from November 2011 to November 2012. Results showed that transplanted eyes suffered from corneal ulcers for an average time of 10.33 ± 5.4 weeks, with an average time of persistent corneal epithelial defect of 5.2 ± 2.8 weeks. Notably, 29/31 eyes were healed within 2 weeks of cell transplantation Nguyen Dinh Ngan, 2013.
Other indications
Stem cell transplantation has also been applied to treat a range of other diseases such as COPD (https://clinicaltrials.gov/ct2/show/NCT02645305?term=NCT02645305&rank=1), cerebral palsy (https://clinicaltrials.gov/ct2/show/NCT02569775?term=NCT02569775&rank=1), autism (https://clinicaltrials.gov/ct2/show/NCT02627131?term=NCT02627131&rank=1), heart failure Nguyen Lan Viet, 2014, recessive dystrophic epidermolysis bullosa Tran Minh Dien, 2014 and spinal cord injury (https://clinicaltrials.gov/ct2/show/NCT02034669?term=NCT02034669&rank=1). Non-expanded, BMSCs have been used to treat acute myocardial infarction since 2011. All participants were transfused via intracoronary with BMSCs after the acute phase of acute myocardial infarction. A recent report showed that the 100 participants in this study were divided equally into two groups: a control group (50 patients) treated as usual and a stem cell group (50 patients) treated with stem cells. After 12 months, results indicated the incidence of prespecified, combined clinical endpoints including death, recurrence of myocardial infarction, coronary revascularization, and rehospitalization for heart failure were significantly lower in the stem cell-treated group (12%) compared with the control group (26%). Improvements in New York Heart Association classification and pro-brain natriuretic peptide levels were statistically significant in both groups. Absolute improvements in left ventricular ejection fraction were significantly greater in the stem cell group compared with the control group Nguyen Lan Viet,2014.
The first clinical trial investigating a stem cell therapy for spinal cord injury was performed at Viet Duc Hospital in 2013. Approved by the Vietnamese Ministry of Health, this trial is using both local injections and systemic transfusion of non-expanded autologous SVF for initial treatment, and then expanded ADSCs for the second and third treatments. This trial marks the first approval of a procedure incorporating systemic transfusion of ex vivoexpanded ADSCs. While this study has just completed phase I/II in a limited number of patients, initial results indicate this procedure may provide some benefit to patients.
In 2015, the Ministry of Health approved the first clinical trial investigating transplantation of ADSCs for COPD. A search of clinical trial databases suggests this is one of only a few clinical trials in the world using a stem cell therapy for COPD (https://clinicaltrials.gov/ct2/show/NCT02645305?term=NCT02645305&rank=1). All patients had adipose tissue collected in order to isolate SVF. SVF was mixed with autologous PRP before being diluted with saline for transfusion. Transfused patients were monitored for 12 months to evaluate both safety and efficacy. Preliminary reports suggest this therapy can significantly reduce COPD symptoms, especially in a few cases showing dramatic improvement after 6 months.
A few instances of other disorders, including cerebral palsy (https://clinicaltrials.gov/ct2/show/NCT02569775?term=NCT02569775&rank=1), autism (https://clinicaltrials.gov/ct2/show/NCT02627131?term=NCT02627131&rank=1) and recessive dystrophic epidermolysis bullosa Tran Minh Dien, 2014, have also been treated with BMSCs. Almost all of these studies used autologous, non-expanded BMSCs. In a similar manner, patients with solid cancers, such as ovarian or breast cancers, have been transplanted with HSCs to recover bone marrow function after treatment with high-dose chemotherapies. Although some improvements have been reported in patients with recessive dystrophic epidermolysis bullosa Tran Minh Dien, 2014 and in a few other diseases, larger numbers of patients need to be treated and monitored to confirm results. Moreover, translational studies should also be performed to understand underlying therapeutic mechanisms.
Cosmetics
Similar to other Asian countries, stem cell transplantation for cosmetic surgical purposes began in Vietnam around the year 2000. Almost all of these activities directly relate to business; therefore, few studies or projects have been published. The most popular applications of stem cells use ADSCs, SVF and PRP during breast augmentation, as well as in wrinkle treatment. These techniques are often performed at beauty institutes that license adipose tissue transfer techniques. Indeed, in the 2000s, fat transfer techniques imported from Korea and Japan began to be used for breast augmentation, and then to fill wrinkles in facial skin. This technique rapidly developed from 2005 to 2010; however, because of its limitations, a modified SVF transplantation came to fruition. SVF is extracted by a commercial kit and then combined with PRP and fat before being transferred into breast or other target tissues. Although no scientific publications describe this procedure, SVF transplantations are performed every day at cosmetic hospitals or “Institutes of Cosmetics”. SVF transplantation for breast regeneration after cancerrelated surgery has also been reported in some cases.
Stem cell banks
Stem cell transplantation in Vietnam has been stimulated and facilitated by the establishment of a stem cell bank system. Established in 2002 at BTH, the first stem cell bank was a public bank that only cryopreserved UCB. To date, Vietnam has five stem cell banks: two in Ho Chi Minh City (BTH’s public cord blood bank and Mekostem’s private bank established in 2008) and three in Hanoi (the National Institute of Hematology and Blood Transfusion’s public cord blood bank established in 2013, the Pediatric Institute’s public cord blood bank established in 2011, and Vinmec’s private cord blood bank established in 2014). Excepting Mekostem’s bank of both cryopreserved UCB and stem cells from umbilical cord lining membrane, all the remaining banks only store UCB.
Although five banks exist, the total number of UCB samples (both private and public) is limited to no more than 10,000 samples. This is the primary reason only a few transplant cases use this source of cells. In fact, the ratio of human leukocyte antigen (HLA) matching likely ranges from 1:10,000 to 1:100,000 of samples. Hence, UCB banks are useful for HSC transplantation only when the HLA-typing of total samples reaches 100,000 to 1,000,000 samples. Fortunately, some banks have become members of international cord blood societies and associations, such as AsiaCord or NetCord.
Technologies for UCB processing and cryopreservation in these banks can vary greatly; therefore, service fees also vary between banks. While some banks use automated, functionally closed systems to harvest stem cell rich buffy coat from UCB (e.g., the AutoXpress® System developed by Cesca Therapeutics, Rancho Cordova, CA), others use manual procedures to collect mononuclear cells with Ficoll centrifugation. In recent years, many banks have standardized their practices with international guidelines, including performing HLA-typing of UCB samples. These important steps can push UCBderived stem cell transplantation into the clinic.
Stem cell industry
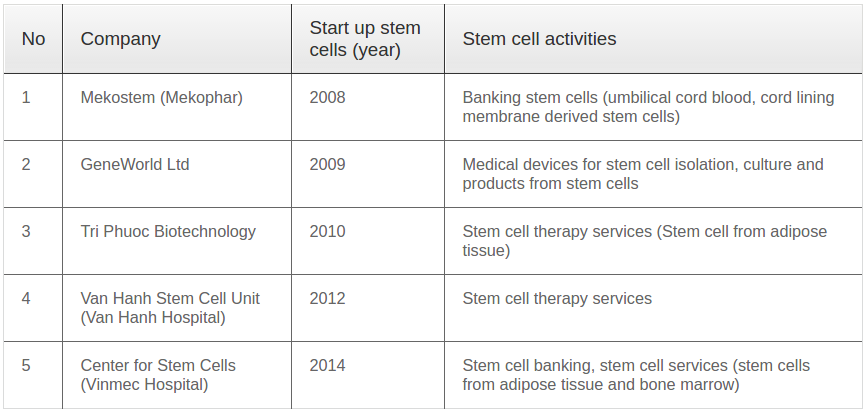
A true stem cell industry only began in Vietnam in 2008 when the first private stem cell company, Mekostem (then Mekophar), established itself in Ho Chi Minh City. This company opened the first stem cell bank in Vietnam; before this time, almost all stem cell transplantation was carried out during clinical studies in government hospitals. Mekostem ushered a new age for stem cell business and there are now four other stem cell companies in Vietnam. In addition to two private stem cell banks, two stem cell service companies have also formed. The main objective of these companies is GMP-compliant isolation and culture of stem cells for clinical applications. In 2009, the first company producing stem cell extraction and culture kits, as well as stem cell-based products was established. These companies actively push stem cell business growth in Vietnam ( Table 2 ).
Recently, the stem cell industry has been stimulated by collaborations between companies and university laboratories. A notable collaboration between SCL and GeneWorld, Ltd. (Saigon Hightech Park) successfully developed and commercialized the first ADSC isolation/extraction kits in Vietnam. Currently, this kit is also exported to several other Asian countries.
The stem cell industry within Vietnam is predicted to rapidly increase beginning in 2020. Indeed, almost all current clinical trials using stem cells will be finalized between 2017 and 2020. If all present clinical trials finish with satisfactory results, personalized medicine based on stem cell transplantation will grow exponentially. At present, there are at least four companies taking part clinical trials for five indications, including Geneworld, Ltd. (knee osteoarthritis and COPD), Van Hanh Hospital (knee osteoarthritis and COPD), Vinmec Hospital (autism and cerebral palsy), and Tri Phuoc Biotechnology Company (spinal cord injury).
Based on the results of a SCL study performed in animal models, other diseases such as type I and II diabetes, autoimmune diseases, heart failure and liver cirrhosis will also be treated in the clinic with stem cell transplantation in the near future. The stem cell industry in Vietnam has also benefitted from contributions from cosmetic stem cell transplantation (in particular, breast augmentation and anti-wrinkle therapies). This field has developed silently, but revenue from these services was estimated at over 10 million USD in 2015. As the fourteenth most populous country on the planet with a population of around 100 million, Vietnam is a large and valuable market for future stem cell therapies.
Challenges and opportunities
With a 20-year history of stem cell transplantation, growth in Vietnam’s stem cell industry has many distinct advantages. The most notable advantage is the awareness of its citizens about stem cells; they believe in the potential efficacy of stem cell therapies. Demand for stem cell transplantation has significantly increased in recent years, especially with sudden increases in many chronic diseases such as COPD, knee osteoarthritis, diabetes and liver cirrhosis. Moreover, Vietnam’s economy has experienced significant growth for the past ten years and its citizens are interested in taking care of themselves.
However, a lot of challenges remain for continued and sustainable growth of the Vietnamese stem cell industry. One of the biggest challenges is a lack of stem cell researchers and scientists, which, to date, only includes about 300 stem cell researchers including basic, translational and clinical studies.
Almost all of these researchers graduated from biotechnology or general medicine programs at local universities, while only a few graduated from foreign universities. Currently, no undergraduate or graduate stem cell programs are being administered at any Vietnamese university; accordingly, stem cell laboratories and banks remain few in number.
To overcome these disadvantages, Vietnam must realize some essential solutions, including: (1) increasing domestic and international collaborations for translational research and clinical studies, (2) establishing undergraduate and graduate programs for stem cell science, (3) building stem cell research and transplantation policy guidelines, and (4) building a stem cell master plan for Vietnam.
Conclusion
Vietnam has a long history of stem cell transplantation, which has rapidly increased from focusing on hematologic malignance and disorders in the early 2000s, to orthopedics, wound healing and ophthalmology. Since 2010, numerous other applications for stem cell transplantation have evolved, including COPD, autism, cerebral palsy and spinal cord injury. Almost all clinical applications used bone marrow or ADSCs to generate transplantable HSCs or MSCs. Although growth of Vietnam’s stem cell industry still faces some difficulties, with generous government support and greater participation by private companies, Vietnam will become a stem cell center for Asia and, in the near future, the world.
Abbreviations
ASC: adult stem cell
BMSC: bone marrow-derived stem cell
BTH: Blood Transfusion and Hematology Hospital
COPD: chronic obstructive pulmonary disease
ESC: embryonic stem cell
GMP: Good Manufacturing Practice
HLA: human leukocyte antigen
HSC: hematopoietic stem cell
HSCT: hematopoietic stem cell transplant
iPSC: induced pluripotent stem cell
LSC: limbal stem cell
MSC: mesenchymal stem cell
NUS: National University of Singapore
PRP: platelet rich plasma
SCL: Laboratory of Stem Cell Research and Application
SVF: stromal vascular fraction
UCB: umbilical cord blood
Ethics Approval and Consent to Participate
Not applicable.
References
-
Khanh
Bach Quoc,
V.T.T.B.,
Tri
Nguyen Anh.
Hematopoietic stem cell transplantation for serve aplastic anemia. Viet Nam Journal of Medicine.
2012;
:
72-75
.
-
Han
Dinh Van,
N.H.A..
Treatment of radiation wounds by cultured fiboblasts. Jourmal of Disaster Medicine and Burn Injuries.
2006;
:
113-155
.
-
Huong
Do Thuy,
.
N.T.B,
Son
Nguyen Khang,
Binh
Trinh,
Tien
Trinh Sinh,
Phuong
Dao Thi Thuy,
Chau
Hoang Thi Minh,
Khanh
Vu Thi Tue,
Oanh
Nguyen Thi Kim,
Lan
Nguyen Thu,
Thien
Nguyen Phu,
Phuong
Phan Thi,
Thanh
Hoang Thi,
Loi
Do Doan.
Researching the culture method to form corneal epithelial sheet from corneoscleral limbal stem cell. Journal of Medicine Research.
2009;
5
:
7-10
.
-
Truc
Duong Ba,
B.V.V.,
Ha
Tran Thi-Hong,
Mai
Nguyen Thi-Thanh,
Dung
Khu Thi-Khanh.
Result of transplantation of hematopoietic stem cells in treatment of beta thalassemia patients. Vietnam Journal of Medicine.
2013;
:
114-117
.
-
Toan
Duong Dinh,
D.X.T.,
Thuy
Nguyen Xuan,
Ha
Nguyen Thi Thu,
Khai
Ly Tuan,
Binh
Nguyen Thanh.
Arthroscopic subchondral drilling and autologous bone marrow stem cell injection. Vietnam Journal of Medicine.
2013a;
:
41-47
.
-
Toan
Duong Dinh,
D.X.T.,
Thuy
Nguyen Xuan,
Ha
Nguyen Thi Thu,
Khai
Ly Tuan,
Binh
Nguyen Thanh.
Treatment of osteoarthritis of the knee by arthrosopic subchondral drilling and autologous bone marrow stem cell injection. Journal of Medicine Research.
2013b;
3
:
122-128
.
-
Toan
Duong Dinh,
L.T.K..
The initial results of treatment of knee osteoarthritis by arthroscopic subchondral drilling and autologous bone marrow stem cell injection. Journal of Military of Pharmaco-Medicine.
2014;
:
108-114
.
-
Phu
Huynh Duc Vinh,
N.H.T.,
Phu Chi Dung
Huynh Van man.
Allogeneic hematopoietic stem cell transplantation in the myelodysplastic syndrome: a case report. Viet Nam Journal of Medicine.
2013;
:
48-55
.
-
Phu
Huynh Duc-Vinh,
D.Q.N.,
Man
Huynh Van,
Dung
Phu Chi,
Binh
Nguyen Tan.
Initial assesment of autologous peripheral blood stem cell transplantation for multiple myeloma at HCMC blood transfusion and hematology hospital. Vietnam Journal of Medicine.
2013;
:
118-215
.
-
Trung
Le The,
T.V.H.,
Dong
Nong Ich.
The first case of treatment of burn injury by cultured keratinocytes. Journal of Disaster Medicine and Burn Injuries.
1998;
:
67-70
.
-
Doan
Le Van,
C.D.N.,
Ha
Nguyen Thi-Thu,
Dung
Do Tien,
Khai
Ly Tuan.
Treatment of long bone non-union by autologous bone marrow. Vietnam Journal of Medicine.
2011;
:
138-145
.
-
N.V.
Luong.
Preparation of autologous cultured keratinocytes for full thickness burned and wounded patients. Journal of Disaster Medicine and Burn Injuries.
2010;
:
37-44
.
-
Khai
Ly Tuan,
N.T.B..
The effects of autologous bone marrow stem cells in treatment of some bone and joint diseases. Journal of Military of Pharmaco-Medicine.
2013;
:
175-185
.
-
Viet
Mai Dac,
L.H.H.,
Hai
Luu Hong,
Dung
Nguyen Quoc,
Tuan
Phung Van,
Hanh
Le.
Evaluation of results of treatment of osteonecrosis of the femoral head with forage decompression and implantation of autologous bone marrow cells. Vietnam Journal of Medicine.
2010;
:
348-355
.
-
Tuan
Mai Manh,
C.Q.T.,
Bui Thi Nga
Tran Thanh Loan.
Preliminary outcome on applying stem cells isolated from membrane lining of umbilical cord in the treatment of wound. Journal of Traditional Medicine and Pharmacy Research.
2007;
:
27-33
.
-
Toan
Ngo Ba,
N.M.K.,
Nguyen Dac Vinh
Tran Dinh Chien.
Evaluate the effect of percutanous drilling and autologous bone marrow derived stem cells grafting on bone healing of the long bone diaphyseal non-unions in Vietduc hospital. Journal of Preventive Medicine.
2012;
7
:
99-105
.
-
Toan
Ngo Ba,
N.M.K.,
Chien
Tran Dinh,
Vinh
Nguyen Dac,
Hai
Le Xuan,
Tri
Nguyen Anh.
The effectiveness of procedures: percutanenous drilling and autologous bone marrow derived stem cells grafting for bone diaphyseal nonunions. Journal of Preventive Medicine.
2014;
4
:
85-91
.
-
Ngan
Nguyen Dinh,
H.T.M.C.,
Thang
Phan Toan.
Stuyding on treatment of persistent corneal epithelial defect by cultured epithelium sheet from umbilical cord. Vietnam Journal of Medicine.
2013;
:
93-100
.
-
An
Nguyen Hai,
N.V.V..
Application of allogenic cultured fibroblasts to treat partial thickness burned wounds. Jounral of Disaster Medicine and Burn Injuries.
2007;
:
50-57
.
-
Viet
Nguyen Lan,
D.D.L.,
Yen
Nguyen Thi Bach,
Hung
Pham Manh,
Anh
Nguyen Quoc,
Khai
Ly Tuan,
Quang
Nguyen Ngoc,
Dat
Phan Tuan,
Hieu
Tran Ba.
Evaluating the results of treatment of heart failure after myocardial infarction by transplantation of autologous bone marrow stem cells. Journal of Vietnamese Cardiology.
2014;
:
8
.
-
Khanh
Nguyen Manh,
N.T.-T.H.,
Binh
Nguyen Tien,
Khai
Ly Tuan,
Nguyet
Truong Thi-Minh,
Binh
Nguyen Thanh,
Toan
Ngo Van.
Autologous bone marrow transfusion to treat the tibia diaphyseal nonunions. Journal of Medicine Research.
2007;
4
:
4-8
.
-
Khanh
Nguyen Manh,
N.T.T.H.,
Binh
Nguyen Tien,
Khai
Ly Tuan,
Binh
Nguyen Thanh.
The effect of bone marrow derived stem cell grafting in the tibia nonunions treatment. Journal of Medicine Research.
2010;
5
:
13-19
.
-
Binh
Nguyen Thanh,
N.T.-T.H.,
Khai
Ly Tuan,
Tich
Dao Xuan,
Dung
Do Tien,
Hai
Le Hong,
Khanh
Nguyen Manh.
The effect of autologous bone marrow stem cells in treatment of some bone and joint diseases. Vietnam Journal of Medicine.
2012;
:
551-558
.
-
Ha
Nguyen Thi-Thu,
L.T.K.,
Binh
Nguyen Thanh,
Dung
Do Tien,
Hai
Le Hong,
Khanh
Nguyen Manh.
The effects of autologous bone marrow stem cell in treatment of long bone nonunions, aseptoc femroal head osteonecrosis and limb lengthening. Vietnam Journal of Medicine.
2013;
:
33-40
.
-
Ha
Nguyen Thi-Thu,
L.T.K.,
Binh
Nguyen Thanh,
Chinh
Nguyen Trung,
Truong
La Van,
Hai
Nguyen Duy,
Huong
Pham Thi-Thu,
Loi
Do Doan.
Autologous peripheral blood stem cell carried out at the central military hospital 108 for the patients with non-hodgkin lymphoma and multuple myeloma. Journal of Medicine Research.
2010;
5
:
1-7
.
-
Luong
Nguyen Van,
D.T.D.,
Binh
Nguyen The,
Anh
Nguyen Vu-Tuan,
Hung
Nguyen Viet.
The preliminary result of transplantation of autologous bone marrow stem cells in treatment of bone defect and short limb. Vietnam Journal of Medicine.
2010;
:
257-262
.
-
E.E.
Osgood,
M.C.
Riddle,
T.J.
Mathews.
Aplastic anemia treated with daily transfusions and intravenous marrow: case report. Annals of Internal Medicine.
1939;
13
:
357-367
.
-
Thu
Pham Hoai,
M.T.K.,
Hong
Nguyen Mai,
Lan
Nguyen Thi Ngoc.
Initial evaluation of clinical results of intra-articular injection of autologous adipose tissue derived stem cell therapy in primary knee osteoarthritis treatment. Journal of Military Pharmaco-Medicine.
2014;
:
83-88
.
-
Dong
Pham Ngoc,
H.T.M.C..
Treatment’s result of severe fungal conerneal ulcer by penetrating keratoplasty. Vietnam Journal of Medicine.
2007;
:
34-39
.
-
Dong
Pham Ngoc,
H.T.M.C.,
Van
Pham Thi Khanh.
Treatment of severe corneal ulcer by penetrating keratoplasty. Journal of Medicine Research.
2006;
5
:
51-55
.
-
Dong
Pham Ngoc,
T.N.H.,
Cung
Le Xuan.
Characteristics of corneal transplanted patients in Vietnam National Insitute of Ophthamology over period of 10 years (2002-2011). Journal of Medicine Research.
2013;
5
:
24-29
.
-
P.
Pham,
H.-T.K.
Bui,
D.T.
Duong,
T.N.
Nguyen,
D.T.
Nguyen,
T.V.
Le,
T.V.
Mai,
L.-C.N.
Phan,
M.D.
Le,
K.N.
Ngoc.
Symptomatic knee osteoarthritis treatment using autologous adipose derived stem cells and platelet-rich plasma: a clinical study. Biomedical Research and Therapy.
2014;
1
:
1-7
.
-
K.
Takahashi,
S.
Yamanaka.
Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell.
2006;
126
:
663-676
.
-
E.D.
Thomas,
H.L. Jr.
Lochte,
W.C.
Lu,
J.W.
Ferrebee.
Intravenous infusion of bone marrow in patients receiving radiation and chemotherapy. N Engl J Med.
1957;
257
:
491-496
.
-
L.T.H.
Thua,
P.D.
Bui,
T.D.
Nguyen,
N.D.
Pham,
B.Q.
Le,
H.P.
Nguyen,
V.N.
Tran,
Q.P.
Le,
D.W.
Boeckx,
A.
Mey.
Autologous Bone Marrow Stem Cells combined with Allograft Cancellous Bone in Treatment of Nonunion. Biomedical Research and Therapy.
2015a;
2
:
1-9
.
-
L.T.H.
Thua,
N.D.
Pham,
B.Q.N.
Le,
H.P.
Nguyen,
H.T.T.
Phan,
D.H.
Phan,
Q.P.
Le,
D.W.
Boeckx,
A.
Mey.
Miniinvasive treatment for delayed or nonunion: the use of percutaneous autologous bone marrow injection. Biomedical Research and Therapy.
2015b;
2
:
1-7
.
-
T.T.
Thuy.
Long term results of trabeculectomy wit aminiotic membrane transplantation for the treatment of recurrent glaucoma. Journal of Medicine Research.
2011;
3
:
253-257
.
-
Toai
Tran Cong,
T.T.-T.T.,
Ngoc
Phan Kim,
Thang
Diep Huu.
Culture of autograft limbal stem cells and cheek epithelial cells to treat corneal epithelium disease. HCMC Journal of Medicine.
2011;
15
:
24-30
.
-
D.-X.T.
Tran,
T.-B.P.
Le,
P.
Pham.
Diabetic foot ulcer treatment by activated platelet rich plasma: a clinical study. Biomedical Research and Therapy.
2014;
1
:
1-6
.
-
Dien
Tran Minh,
N.D.N.,
Mai
Nguyen Thi-Thanh,
Thach
Hoang Ngoc,
Dung
Vu Chi,
Liem
Nguyen Thanh,
Huong
Le Thi Minh.
Bone marrow stem cell transplantation for recessive dystrophic epidermolysis bullosa: a case report. Vietnam Journal of Medicine.
2014;
:
51-55
.
-
Tuan
Tran Quoc,
N.T.B..
Research of application of the allogenic peripheral blood stem cell transplantation by cryopreservation at -196 oC in acute myeloid leukemia at the blood transfusion and hematology hospital in HCM city. HCMC Journal of Medicine.
2011a;
4
:
167-174
.
-
Tuan
Tran Quoc,
N.T.B..
Research of application of the autologous peripheral blood stem cell transplantation by cropreservation at -196oC in acute myelogenous leukemia at the blood transfusion and hematology hospital HCMC. HCMC Journal of Medicine.
2011b;
15
:
174-179
.
-
T.
Be,
T.
van Binh,
N.
Binh,
T.
Tuan,
H.
Nghia,
B.
Hien.
Current status of hematopoietic stem cell transplantations in Vietnam. Bone Marrow Transplant 42 Suppl.
2008;
1
:
S146-S148
.
-
Khanh
Vu Thi-Tue,
H.T.-M.C.,
Binh
Nguyen Thi,
Son
Nguyen Khang,
Tien
Trinh Sinh,
Huong
Do Thuy,
Phuong
Dao Thi-Thuy,
Thien
Nguyen Phu,
Oanh
Nguyen Thi-Kim,
Lan
Nguyen Thu,
Phuong
Phan Thi,
Loi
Do Doan.
Primary outcome of auto-cultivated limbal epithelial transplantation in management of limbal stem cell deficiency syndrome. Journal of Medicine Research.
2009;
6
:
1-6
.
-
Khanh
Vu Thi-Tue,
H.T.M.C.,
Son
Nguyen Khang,
Binh
Nguyen Thi.
Auto cultivated limbal epithelial transplantation in management of unlateral limbal stem cell deficiency syndrome following by lime injury. Journal of Medicine Research 3C.
2012;
:
60-67
.
Comments
Downloads
Article Details
Volume & Issue : Vol 3 No 04 (2016)
Page No.: 578-587
Published on: 2016-04-15
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
- HTML viewed - 13024 times
- Download PDF downloaded - 2536 times
- View Article downloaded - 11 times
 Biomedpress
Biomedpress