Abstract
Introduction: The role of Tridax procumbens leaf extract in erectile dysfunction (ED) of chronic variable stress (CVS) etiology is unknown. This study investigates the potential of the ethanol fraction of Tridax procumbens leaf (EETP) in modulation of CVS-induced ED.
Methods: Twenty-five male Wistar rats were divided into five groups of five rats each. Groups 1 & 2 (without stress) were treated with normal saline (vehicle) and 100 mg/kg of EETP, respectively. Groups 3?5 were treated as stress groups, with Group 3 co-treated with 100 mg/kg of EETP, and group 4 co-treated with vitamin C (7 mg/kg). Treatments were administered by oral gavage once daily for seven weeks. Oxidative biomarkers, cortisol, testosterone, and sperm parameters were determined, as well as the contractile mechanism of the corpus cavernosa to cumulative doses of calcium chloride, potassium chloride, acetylcholine, and sodium nitroprusside. Furthermore, the contractile mechanism was also determined after incubation in acetovanillone, nicorandil, methyl blue, and glibenclamide.
Results: Serum cortisol was significantly reduced, while testosterone was significantly increased in the EETP supplemented groups when compared to the CVS-only exposed group. Furthermore, malonaldehyde activity was decreased while superoxide dismutase concentration was increased in the EETP- and vitamin C-supplemented groups when compared to the CVS-only exposed group. Contraction (%) responses to calcium chloride and potassium chloride were also significantly reduced in the CVS-only exposed group when compared to the EETP-supplemented groups. The relaxation responses (%) to acetylcholine and SNP were significantly increased in the CVS group supplemented with EETP and vitamin C when compared to the CVS-only exposed group. The incubation of the cavernosa tissues in acetovanillone and nicorandil resulted in increased relaxation (%) in the CVS-only group, while incubation in glibenclamide caused increased relaxation in the EETP-supplemented groups compared to CVSonly exposed group. Sperm motility (%) was significantly reduced while abnormal spermatozoa was increased in the CVS-only exposed group when compared to the groups supplemented with EETP and Vitamin C.
Conclusion: Variable stress-induced dysfunctions in erectile mechanism were attenuated through supplementation with EETP.
Introduction
For decades, increasing incidence of erectile dysfunction (ED) has been a problem for public health1. Currently, a prevalence of 52% is reported in the United States, with approximately 40% of men having it at 40 years old and almost 70% by the age of 70 years1, 2. Occurrence in Nigeria is also high. A cross-sectional descriptive study previously revealed a prevalence of 45.7%3. Other studies revealed a prevalence of 41.5% in the Niger Delta4, 66.4% in Abuja5, 46.9% in Ilorin6, and 58.9% in Ogbomosho, in the southwestern region7 of the country.
The development of ED is multifactorial and usually involves neurologic, cardiovascular, hormonal, psychological. and local anatomic systems1. Chronic variable stress resulting in oxidative stress is also a known cause of erectile dysfunction and male infertility8. Numerous orthodox treatments are in common use, as some herbs and supplements have been found to be effective in the treatment of ED9. Furthermore, complementary and alternative medicine is hugely popular in Africa due to cultural beliefs, affordability, and accessibility10.
Tridax procumbens is a natural plant of the Asteraceae family that occurs naturally in tropical Africa, Asia, Australia, and India. Bioactive compounds, such as hydroxycinnamates, flavonoids, alkaloids, phytosterols, vitamin C, linoleic acid, proteins, tannins, and carotenoids, have been reported to be present in the plant11.
There is evidence that extracts of Tridax procumbens have potentials for modulating ED caused by experimental hypertension12 as well as paroxetine-induced13 ED. Vitamin C, a synthetic antioxidant, has been reported to attenuate chronic variable stress-induced ED14. Several reports have shown that Tridax procumbens has potent antioxidant properties15, 16, 17, 18.
Tridax procumbens leaf extract has shown potential in attenuating erectile dysfunction in experimental hypertensive and paroxetine-induced rats. However, the function of Tridax procumbens leaf extract in erectile dysfunctions of chronic variable stress etiology is unknown. This study investigates the potential of EETP in modulating and/or attenuating CVS-induced ED and sperm impairments in male Wistar rats.
Methods
Animals, acclimatization and ethical approval
Twenty-five healthy adult male Wistar rats (180–200 g) were purchased from the Lagos State University College of Medicine animal house. Animals underwent acclimatization for two weeks within temperatures of 21–26 0C and 30%–70 % relative humidity. They were allowed access to standard food (Ladokun feeds Nigeria Ltd.) in pellet form and drinking water ad libitum. The NIH guide procedures for laboratory animal safety and use was adhered to at all times throughout the study and the procedures used on the animals were certified by the Lagos State University College of Medicine Animal Ethics Committee (AREC/2021/025).
Collection and preparation of ethanol extract of Tridax procumbens leaves (EETP)
Leaves of Tridax procumbens were collected from Lagos State University, Ojo, Lagos state, Nigeria in May 2022. The collected plant samples were authenticated by a certified taxonomist, with voucher number FHI 1008876. The extraction was performed according to previous literature13. Leaves were air-dried and ground to produce 300 g of product in smooth, powdered form. This was placed in a clean, conical flask and mixed with 5 liters of 96% ethanol, creating a suspension that was left to stand for 48 hours. The mixture was then filtered and the filtrate was left to stand for another 48 hours. After 48 hours, the filtrate was decanted carefully and allowed to concentrate by evaporation in a rotary evaporator at 35–40 0C. The yield was 5.83% (17.5 g) of a light-brown powdery extract.
Drugs and chemicals for the study
Vitamin C produced by the Emzor pharmaceutical company, Lagos, Nigeria was used. The potassium chloride, glucose, sodium bicarbonate, potassium phosphate, magnesium sulphate, calcium chloride, and sodium chloride used for the physiological salt solution were manufactured by J.T Baker Chemical Company, USA. Drugs used to assess dose response of the tissues included phenylephrine (PHE) and acetylcholine (Ach) purchased from Tocris, UK. Sodium nitroprusside was manufactured by Suvidhinath Laboratories, India, while acetovanillone, nicorandil, and N-nitro-l-arginine methyl ester (L-NAME) were purchased from AK Scientific, Inc., CA, USA. Glibenclamide was manufactured by Emcure Pharmaceuticals Ltd., while methyl blue, calcium chloride, and potassium chloride was produced by J.T Baker Chemical Company, USA.
Study design and treatment
Twenty-five male Wistar rats were divided into five groups of five rats each. Groups 1 and 2 (non-stress groups) were treated with normal saline (vehicle) and 100 mg/kg of EETP, respectively12. Groups 3, 4, and 5 were stress groups, with groups 3 and 4 co-treated with 100 mg/kg of EETP and vitamin C (7 mg/kg)14, respectively, while Group 5 received no supplementation. These treatments were administered via oral gavage once daily for seven weeks.
Procedure for variable stress exposure
The stress model of Mueller and Bale19 was used with slight modifications, as reported by Salami et al.14. These included sleep deprivation initiated by saturating the beddings of the cage with tepid water through the night; immobilization by restraining each rat in a 50 cl cylindrical container of 3 cm diameter for 20 minutes; fear inducement by placing a predator (cat) in cages with rats separated by a wire mesh; noise stress created by exposing rats to 100 decibels of noise during light cycle for 4 hours; stress from multiple cage changes by changing rats’ cages every 20 minutes interval during a light cycle for a period of 2 hours; and finally, exposure to foreign objects by introducing rough marbles and luminous beads in the cages of the animals at night. A different stress model was used each day for the entire duration of the stress exposure.
Animal sacrifice and serum collection
Animals were anaesthetized with 30 mg/kg of pentobarbital and sacrificed by cervical dislocation. Blood samples were taken from heart ventricles using sterile 5 ml syringes and needles; samples were then placed in plain bottles and allowed to stand for 15 minutes at room temperature. They were then centrifuged using a cold centrifuge (Model SM112, Uniscope Laboratory Centrifuge, England) at 4000 rpm for 15 mins, yielding a supernatant serum, which was carefully aspirated into empty plain bottles and stored at –40C.
Collection and preparation of corpus cavernosa
The corpus cavernosa of the rats were surgically removed and placed in a petri dish that contained physiological salt solution (PSS).
The corpus cavernosa of each animal across the groups were later suspended by a thread in a 50 ml chamber of the organ bath with PSS. Each corpus cavernosum was anchored with a stainless-steel hook to an electronic transducer (7004 model, Ugo-Basile, Varese, Italy) connected to a data capsule (model 17400) for isometric contraction recordings12. The organ bath was bubbled with a gas mixture of 95% oxygen and 5% carbon dioxide at a pH of 7.35–7.40, with temperature maintained at 370C.
Experiments regarding the contractile activity of the cavernosa tissue
The corpus cavernosa were allowed to equilibrate in the PSS for a period of 90 minutes. During this period, cavernosa tissue was stimulated three times at 30-minute intervals with 10-7 M phenylephrine. After equilibration, contractile responses of the cavernosa tissue to cumulative doses of acetylcholine (10-9–10-5 M), sodium nitroprusside (10-9–10-5 M), and potassium chloride (10–60 mM) were determined and recorded. Furthermore, the following were also investigated:
Contractile responses were allowed to occur in a steady state before the addition of further doses and tissues were washed thrice before the use of a separate drug.
Determination of serum superoxide dismutase (SOD) and MDA concentration
Serum SOD was determined as described by Sun and Zigman20, while the method of Buege and Aust21 was used to determine MDA (i.e., an index of lipids peroxidation).
Determination of serum cortisol and testosterone
Serum cortisol was determined using the cortisol ELISA kit (Calbiotech, CA 92020, USA), while and testosterone was determined using the ELISA kit (Accu-Bind ELISA Lake Forest, CA 92630, USA).
Analysis of sperm motility, and morphology
The cauda epididymis was macerated and then immersed in 10 ml of PSS in a sterile specimen bottle. Using a pipette, an aliquot of the suspension was placed on a slide. The percentage of motility was then evaluated based on five different fields with a microscope (Cetti reset microscope). Five different fields were checked and recorded for progressive sperm motility in percentages, while morphological abnormalities were determined using a portion of sperm suspension placed on a glass slide and smeared out with another slide and stained with Leishman’s stain for morphological examination. Abnormalities of the sperm cells noted included coiled tail, headless, rotated head, and microcephaly conditions.
Statistical analysis
Results are presented here as mean ± standard error of mean in percentages. Tension was expressed as a percentage of the initial contraction to phenylephrine. Prism GraphPad (version 8.0.2) was the statistical software used for data analysis. Data were analyzed using the ANOVA test, with p values of less than 0.05 considered statistically significant.
| Control | EETP | EETP+CVS | EETP+VIT C | CVS | |
|---|---|---|---|---|---|
| Cortisol (ng/ml) | 28.5 ± 0.4 | 27.9 ± 0.6 | 25.2 ± 0.5* | 31.5 ± 1.0 | 34.9 ± 1.3 |
| Testosterone (ng/ml) | 3.8 ± 0.2 | 4.0 ± 0.2 | 5.7 ± 0.3* | 5.2 ± 0.4* | 3.6 ± 0.3 |
| MDA (µm/ml) | 0.5 ± 0.01 | 0.5 ± 0.1 | 0.6 ± 0.1 | 0.7 ± 0.1 | 2.6 ± 0.7* |
| SOD (µm/ml) | 1.64 ± 0.05 | 1.67 ± 0.03 | 1.73 ± 0.05 | 1.71 ± 0.04 | 1.44 ± 0.22 |
| Control | EETP | EETP+CVS | EETP+VIT C | CVS | |
|---|---|---|---|---|---|
| Motility (%) | 55.22 ± 3.39 | 58.10 ± 2.96 | 76.25 ± 4.71 | 68.06 ± 7.65 | 12.00 ± 3.39* |
| Abnormal morphology (%) | 5.40 ± 0.93 | 5.60 ± 1.29 | 4.80 ± 0.80 | 5.80 ± 1.77 | 8.00 ± 1.61* |
| Weeks | Control | EETP | CVS+EETP | CVS+VIT C | CVS |
|---|---|---|---|---|---|
| 1 st | 146.0 + 11.9 | 144.0 + 15.2 | 142.4 + 13.5 | 141.4 + 12.4 | 141.2 + 8.9 |
| 4 th | 167.0 + 9.2 | 187.0 + 10.5 | 178.4 +14.8 | 174.0 + 12.4 | 147.6 + 7.9* |
| 7 th | 186.6 + 12.78 | 191.0 + 10.1 | 182.8 + 13.4 | 179.4 + 10.6 | 146.8 + 8.1* |
Results
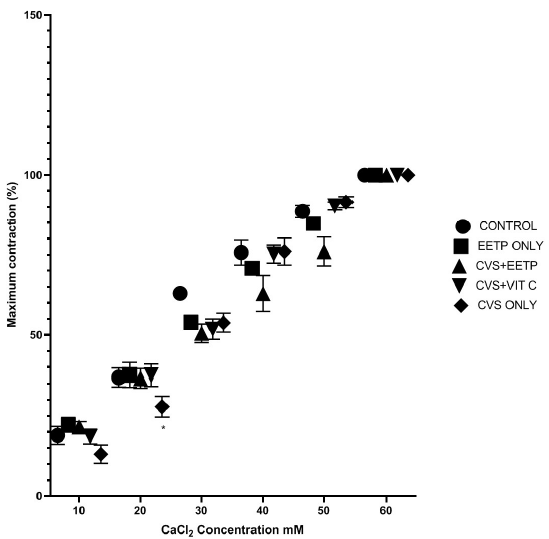
There was a significant reduction in the contraction response to calcium influx in the CVS-only exposed group (13%, 27%) when compared to the EETP supplemented group (21%, 36.6%) and the control (19%, 36.9%) (Figure 1). Furthermore, contraction (%) response to KCl influx was also reduced in the CVS-only group when compared to the EETP-supplemented and control groups (Figure 2). Furthermore, relaxation responses (%) to acetylcholine and SNP were significantly increased in the CVS group supplemented with EETP and vitamin C when compared to CVS-only exposed group (Figure 3 and Figure 4, respectively).
As shown in Figure 5, the incubation of the cavernosa tissues across groups with acetovanillone resulted in increased relaxation (%) in the CVS-only group when compared to EETP supplemented groups and control. Similar observations were recorded after the incubation of the cavernosa tissues in nicorandil (Figure 8). Incubation of cavernosa tissues in glibenclamide and L-NAME resulted in increased relaxation response to cumulative doses of ACh in the EETP supplemented groups as compared to the CVS-only exposed group (Figure 6 and Figure 7, respectively). Relaxation (%) was significantly reduced in the CVS-only exposed group after incubation in methyl blue as compared to control and EETP supplemented groups (Figure 9).
Serum cortisol was significantly reduced in the CVS group supplemented with EETP compared to the CVS-only exposed group. Serum testosterone also significantly increased in the EETP supplemented groups compared to the CVS-only exposed group. Furthermore, serum malonaldehyde activity significantly increased in the CVS-only exposed group when compared to the EETP- and vitamin C-supplemented CVS groups. Superoxide dismutase concentration was reduced in the CVS-only exposed group relative to the EETP supplemented group (Table 1).
As shown in Table 2, motility (%) was significantly reduced in the CVS-only exposed group when compared to the groups supplemented with EETP and vitamin C. Additionally, numbers of abnormal spermatozoa were significantly increased in the CVS-only group as compared to groups supplemented with EETP and vitamin C.
Discussion
This study hypothesized that EETP supplementation during variable stress exposure should ameliorate erectile dysfunctions and reproductive impairments due to its potent antioxidant activity. We observed that EETP supplementation during CVS exposure largely attenuated impaired sperm and contractile mechanisms of the corpus cavernosa. The profound importance of the antioxidant activity of EETP in this attenuation was buttressed by the observation that serum malonaldehyde activity was significantly increased in the CVS-only exposed group when compared to the EETP and vitamin C supplemented CVS groups. Furthermore, superoxide dismutase concentration was reduced in the CVS-only exposed group as compared to the EETP supplemented group (Table 1). Similar protective activity of EETP, as observed in this study, has been previously suggested by other studies13, 22. A recent study also reported a similar observation regarding vitamin C supplementation (alone) during CVS exposure13, 14 in male and gravid female rats. Tridax procumbens is known to be rich in naturally occurring antioxidants, such as poly phenols and flavonoids, which are anti-oxidative in nature. Habilah et al.15 previously observed that Tridax procumbens antioxidant activity was about 96.70% when compared to the 94.81% of vitamin C and 92.92% of garlic.
Interestingly, the protective potential described for EETP above may help explain the modulatory effect of EETP supplementation on the impaired contractile mechanism of the CVS-exposed group in this study. The significant reduction in the contraction (%) by calcium and potassium influx in the CVS group was attenuated significantly in the EETP-supplemented group (Figure 1, Figure 2). Calcium and potassium chloride additions to calcium- and potassium-free solution were conducted to assess the integrity of the contractile mechanism mediated by calcium influx through the voltage-dependent calcium channels and the inhibitory effect of the receptor-operated calcium channels, respectively. We found that CVS exposure clearly impaired these contraction mechanisms and that EETP supplementation was able to reduce impairment through its protective properties against corpus cavernosa tissue damage.
The relaxation mechanism in the corpus cavernosa mediated by parasympathetic agonist (acetylcholine) and endothelia nitric oxide donor (SNP) were significantly increased in the corpus cavernosa of the CVS groups supplemented with EETP and vitamin C as compared to the CVS-only exposed groups (Figure 3, Figure 4). This suggests that EETP supplementation during CVS exposure can protect or enhance relaxation mechanisms mediated by acetylcholine and SNP ways that have not yet been elucidated. The SNP pathway in the activity of EETP is supported, however, by the fact that incubation of corpus cavernosa tissues of the CVS-exposed group in nicorandil (nitric oxide donor and ATP-sensitive potassium channel agonist) resulted in enhanced relaxation of corpora tissues (comparable to EETP supplemented and control groups) (Figure 8). Furthermore, in this study, the antioxidant hypothesis regarding the activity of EETP is highlighted by the enhanced relaxation in the cavernosa tissue of the CVS-exposed group (comparable to EETP supplemented groups) when the tissues were incubated in acetovanillone (antioxidant and NADPH oxidase inhibitor).
This study also found that the degrees of impairment in relaxation were mediated by the activity of ATP-sensitive K+ channels, and that the activity of the soluble guanyl cyclase were pronounced in the cavernosa of the CVS-only exposed group when compared to EETP-supplemented groups. Incubation of cavernosa tissues in glibenclamide and methyl blue also respectively reduced acetylcholine-mediated relaxation significantly in the CVS group as compared to EETP supplemented groups. This indicates the benefits of EETP for the erectile tissues of the CVS-exposed groups.
Quantification of the expression of the specific channel proteins identified in this study would have shed more light on the specific mechanisms involved in the observed ameliorations of erectile dysfunctions by EETP. This is a limitation that should be investigated in future studies.
Serum cortisol was significantly reduced in the CVS group supplemented with EETP when compared with the CVS-only exposed group. However, serum testosterone was also significantly increased in the EETP supplemented groups when compared to the CVS-only exposed group.
Elevated cortisol level during stress exposure is usually a stress survival mechanism that involves hypothalamic-pituitary-adrenocortical (HPA) axis feedback activation23. Previous literature has shown that single24, 25 and variable stressors14 usually potentiate cortisol levels in human and animal models. The ability of EETP to reduce cortisol level in a supplemented group highlights its antioxidant potential for reducing stress and cortisol elevation. Previous studies have also shown that synthetic antioxidant (vitamin C) led to reduced cortisol levels in acute and chronic sleep-deprived stressed rats24 and chronic variable stress exposed male rats14.
The elevated serum testosterone in EETP-supplemented groups may be indicative of hormonal activity in EETP. EETP is known to contain phytosterols11, which can act as steroids or activate other steroids in the steroidogenic pathway in vivo. Other studies have reported elevation in testosterone level after EETP treatment26, 27. Thus, the reduced testosterone level observed in the CVS group could be attributable to stress-induced elevation of cortisol levels acting via glucocorticoid receptors in testicular interstitial cells to suppress the testicular response to gonadotrophin28.
As shown in Table 2, sperm motility (%) was significantly reduced in the CVS-only exposed group as compared to the groups supplemented with EETP and Vitamin C. Additionally, the percentage of abnormal spermatozoa was significantly increased in the CVS-only group when compared to groups supplemented with EETP and vitamin C. These findings support the enhanced antioxidant activities in the EETP supplemented groups found in this study and the elevated lipid peroxidation in the CVS-only group. Lipid peroxidation is known to cause damage in sperm structure, function, and DNA integrity29, which would have in turn compromised motility and elevated abnormal spermatozoa, as observed in this study. Furthermore, testosterone level was elevated in the EETP groups and this may have possibly also improved sperm characteristics and functions26.
In the final analysis, mean body weight at the 4th and 7th weeks were significantly reduced in the CVS-only exposed group when compared to the CVS groups supplemented with EETP and vitamin C. A similar reduction in body weight as a result of stress exposure has been reported in previous studies30. However, the specific mechanism through which EETP was able to increase body weight in this study is unclear. Nonetheless, EETP is known to be rich in vitamins, proteins11, and other substances that we suggest may have enhanced the utilization of food at the cellular and tissue levels in EETP-treated groups.
Conclusions
Chronic variable stress-impaired contractile mechanisms mediated by nitric oxide, calcium channels, ATP-sensitive K+ channels, parasympathetic activation, and cyclic guanylyl cyclase activity. EETP supplementation, however, ameliorated these impaired mechanisms through its potent antioxidant and hormonal activity.
Abbreviations
Ach: Acetylcholine, ATP: Adenosine triphosphate, CaCl2: Calcium chloride, CVS: Cardiovascular system, ED: Erectile dysfunction, EETP: Ethanol fraction of Tridax procumbens, KAT P: adenosine triphosphate sensitive potassium channel, KCl: Potassium chloride, L-NAME: N-nitro-L-arginine methyl ester, MAD: Malondialdehyde, NADPH: Nicotinamide adenine dinucleotide phosphate hydrogenase, PHE: Phenylephrine, SOD: Serum superoxide dismutase
Acknowledgments
None
Author’s contributions
Conceptualization: SAS, Methodology: SAS & HMS; Software: SAS, HMS & BAM; Validation: SAS, HMS; Formal Analysis: SAS, HMS & BAM; Investigation: ACI, BAM; Resources: SAS, HMS, ACI; Data Curation: SAS, HMS, ACI, BAM, AMO, Writing –Original Draft: SAS; Writing – Review &, Editing: SAS, HMS, AMO, BAM; Supervision: SAS. All authors read and approved the final manuscript.
Funding
None
Availability of data and materials
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval
The NIH guide procedures for the laboratory animal safety and use, was adhered to all through the study and the procedures used on the animals were certified by the Lagos State University College of Medicine Animal Ethics Committee (AREC/2021/025).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
-
Milton
L.,
Hadley
W.,
Erectile Dysfunction. 2018. Retrieved from https://www.clevelandclinicmeded.com/medicalpubs/diseasemanagement/endocrinology/erectile-dysfunction/. 2018
.
-
Johannes
C.B.,
Araujo
A.B.,
Feldman
H.A.,
Derby
C.A.,
Kleinman
K.P.,
McKinlay
J.B.,
Incidence of erectile dysfunction in men 40 to 69 years old: longitudinal results from the Massachusetts male aging study. The Journal of Urology.
2000;
163
(2)
:
460-3
.
View Article PubMed Google Scholar -
Umuerri
E.M.,
Ayandele
C.O.,
Prevalenve and Associations of Erectile Dysfunctions and Premature Ejaculation among Nigerian Men: an online survey. Highland Medical Research Journal.
2021;
21
(2)
:
24-30
.
-
Idung
A. U.,
Abasiubong
F.,
Ukott
I. A.,
Udoh
S. B.,
Unadike
B. C.,
Prevalence and risk factors of erectile dysfunction in Niger delta region, Nigeria. African Health Sciences.
2012;
12
(2)
:
160-5
.
View Article Google Scholar -
Abu
S.,
Atim
T.,
Ripiye
N.,
Prevalence of Erectile Dysfunction and Awareness of Its Treatment in Abuja, Nigeria. International Journal of TROPICAL DISEASE & Health.
2019;
37
(3)
:
1-0
.
View Article Google Scholar -
Oladiji
F.,
Kayode
O.O.,
Parakoyi
D.B.,
Influence of socio-demographic characteristics on prevalence of erectile dysfunction in Nigeria. International Journal of Impotence Research.
2013;
25
(1)
:
18-23
.
View Article PubMed Google Scholar -
Oyelade
B.O.,
Jemilohun
A.C.,
Aderibigbe
S.A.,
Prevalence of erectile dysfunction and possible risk factors among men of South-Western Nigeria: a population based study. The Pan African Medical Journal.
2016;
24
:
124
.
View Article PubMed Google Scholar -
Wingfield
J.C.,
Sapolsky
R.M.,
Reproduction and resistance to stress: when and how. Journal of Neuroendocrinology.
2003;
15
(8)
:
711-24
.
View Article PubMed Google Scholar -
Leisegang
K.,
Finelli
R.,
Alternative medicine and herbal remedies in the treatment of erectile dysfunction: A systematic review. Arab Journal of Urology.
2021;
19
(3)
:
323-39
.
View Article PubMed Google Scholar -
Asmerom
D.,
Kalay
T.H.,
Araya
T.Y.,
Desta
D.M.,
Wondafrash
D.Z.,
Tafere
G.G.,
Medicinal Plants Used for the Treatment of Erectile Dysfunction in Ethiopia: A Systematic Review. BioMed Research International.
2021;
2021
:
6656406
.
View Article PubMed Google Scholar -
Ikewuchi
J.,
Ikewuchi
C.,
Ifeanacho
M.,
Phytochemical Composition of Tridax procumbens Linn Leaves: Potential as a Functional Food. Food and Nutrition Sciences.
2015;
6
(11)
:
992-1004
.
View Article Google Scholar -
Salami
S.A.,
Salahdeen
H.M.,
Ugbebor
E.C.,
Murtala
B.A.,
Raji
Y.,
Effects of aqueous leaf extract of Tridax procumbens on contractile activity of corpus cavernosum in N-nitro-l-arginine methyl ester-induced hypertensive male rats. Journal of Integrative Medicine.
2018;
16
(1)
:
51-6
.
View Article PubMed Google Scholar -
Salami
S.,
Salahdeen
H.,
Balogun
Z.,
Murtala
B.,
Raji
Y.,
Mechanisms in Tridax procumbens leaf extract reversal of paroxetine-induced erectile dysfunction in corpus cavernosum of male Wistar rats. Biomedical Research and Therapy.
2021;
8
(6)
:
4405-16
.
View Article Google Scholar -
Salami
S.A.,
Salahdeen
H.M.,
Moronkola
O.T.,
Murtarla
B.A.,
Raji
Y.,
Vitamin C supplementation during chronic variable stress exposure modulates contractile functions of testicular artery and sperm parameters in male Wistar rats. Middle East Fertility Society Journal.
2020;
25
(8)
:
8
.
View Article Google Scholar -
Habila
J.D.,
Bello
I.A.,
Dzikwi
A.A.,
Musa
H.,
Abubakar
N.,
Total phenolics and antioxidant activity of Tridax procumbens Linn. African Journal of Pharmacy and Pharmacology.
2010;
4
(3)
:
123-6
.
View Article Google Scholar -
Pareek
H.,
Sharma
S.,
Jain
G.,
Evaluation of Ameliorative Efficacy of Tridax procumbens on the Sexual Performance of Male Rats with Alloxan-Induced Diabetes. CIBTech Journal of Pharmaceutical Sciences.
2014;
3
(4)
:
50-59
.
-
Akintunde
O.G.,
Oloye
A.A.,
Adetomiwa
A.,
Subuloye
W.,
Olaiya
C.O.,
Effects of ethanol extract of Tridax procumbens on spermiogram and reproductive hormones in Wistar rat. Sokoto Journal of Veterinary Sciences.
2017;
15
(3)
:
25-31
.
View Article Google Scholar -
Andriana
Y.,
Xuan
T.D.,
Quy
T.N.,
Minh
T.N.,
Van
T.M.,
Viet
T.D.,
Antihyperuricemia, antioxidant, and antibacterial activities of Tridax procumbens L. Foods.
2019;
8
(1)
:
21
.
View Article Google Scholar -
Mueller
B.R.,
Bale
T.L.,
Impact of prenatal stress on long term body weight is dependent on timing and maternal sensitivity. Physiology & Behavior.
2006;
88
(4-5)
:
605-14
.
View Article PubMed Google Scholar -
Sun
M.,
Zigman
S.,
An improved spectrophotometric assay for superoxide dismutase based on epinephrine autoxidation. Analytical Biochemistry.
1978;
90
(1)
:
81-9
.
View Article PubMed Google Scholar -
Buege
J.A.,
Aust
S.D.,
Microsomal lipid peroxidation. Methods in Enzymology.
1978;
52
:
302-10
.
View Article PubMed Google Scholar -
Salahdeen
H.M.,
Salami
S.A.,
Paul
C.O.,
Murtala
B.A.,
Alada
A.A.,
Biochemical parameters as indicators of antihypertensive efficacy of leaf aqueous extract of Tridax procumbens (Lin) in L-NAME induced hypertensive rats. Journal of Molecular Pathophysiology..
2017;
6
(2)
:
30-7
.
View Article Google Scholar -
Sheng
J.A.,
Bales
N.J.,
Myers
S.A.,
Bautista
A.I.,
Roueinfar
M.,
Hale
T.M.,
The Hypothalamic-Pituitary-Adrenal Axis: Development, Programming Actions of Hormones, and Maternal-Fetal Interactions. Frontiers in Behavioral Neuroscience.
2021;
14
:
601939
.
View Article PubMed Google Scholar -
Olayaki
L.A.,
Sulaiman
S.O.,
Anoba
N.B.,
Vitamin
C.,
Vitamin C Prevents Sleep Deprivation-induced Elevation in Cortisol and Lipid Peroxidation in the Rat Plasma. Nigerian Journal of Physiological Sciences ; Official Publication of the Physiological Society of Nigeria.
2015;
30
(1-2)
:
5-9
.
PubMed Google Scholar -
Chu
X.,
Zhou
Y.,
Hu
Z.,
Lou
J.,
Song
W.,
Li
J.,
24-hour-restraint stress induces long-term depressive-like phenotypes in mice. Scientific Reports.
2016;
6
(1)
:
32935
.
View Article PubMed Google Scholar -
Salami
S.,
Salahdeen
H.,
Rahman
O.,
Murtala
B.,
Raji
Y.,
Oral administration of Tridax procumbens aqueous leaf extract attenuates reproductive function impairments in L-NAME induced hypertensive male rats. Middle East Fertility Society Journal.
2017;
22
(3)
:
219-25
.
View Article Google Scholar -
Akintunde
O.G.,
Oloye
A.A.,
Adetomiwa
A.,
Subuloye
W.,
Olaiya
C.O.,
Effects of ethanol extract of Tridax procumbens on spermiogram and reproductive hormones in Wistar rat. Sokoto Journal of Veterinary Sciences.
2017;
15
(3)
:
25-31
.
View Article Google Scholar -
Hampl
R.,
Stárka
L.,
Glucocorticoids affect male testicular steroidogenesis. Physiological Research.
2020;
69
(2)
:
205-10
.
View Article PubMed Google Scholar -
Kavita
M.,
Badade
Z.G.,
Narshetty
J.G.,
Joshi
D.S.,
Mukherjee
S.,
Deepak
A.D.,
Lipid Peroxidation, Sperm DNA Fragmentation Total Antioxidant Capacity and Semen Quality in Male Infertility. MGM Journal of Medical Sciences.
2014;
1
(1)
:
1-6
.
View Article Google Scholar -
Jeong
J.Y.,
Lee
D.H.,
Kang
S.S.,
Effects of chronic restraint stress on body weight, food intake, and hypothalamic gene expressions in mice. Endocrinology and Metabolism (Seoul, Korea).
2013;
28
(4)
:
288-96
.
View Article PubMed Google Scholar
Comments
Article Details
Volume & Issue : Vol 10 No 9 (2023)
Page No.: 5912-5923
Published on: 2023-09-30
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 4304 times
- PDF downloaded - 1207 times
- XML downloaded - 135 times
 Biomedpress
Biomedpress











