Abstract
Introduction: Memory impairment is one of the most important complications in patients with HIV infection. The syndrome is caused by reductions in brain volume and the count of circulating CD4+ lymphocytes. This study was conducted to evaluate the relationship between CD4+ lymphocyte count and memory function in HIV+ patients.
Methods: This descriptive-analytical study was conducted on 150 HIV+ patients referred to the Behavioral Disorders Counseling Center of Kermanshah City. Memory function in patients was measured using the Wechsler memory scale. The patients' CD4+ cell counts and demographic information were extracted from their medical files. The data were recorded in STATA version 16 software and analyzed using regression and finite mixture models.
Results: The meansstandard deviations of memory function in three classes of patients were 63.99 +/- 7.02, 75.01 +/- 14.72, and 85.14 +/- 6.43. The results showed that a decrease in CD4+ cell count increased the risk of memory loss in patients (P < 0.001). In addition, higher age (P < 0.001), female gender (P < 0.001), and a lower education level (P < 0.001) were significantly associated with an increased risk of memory loss in HIV+ patients.
Conclusion: The results of the present study confirmed the findings of previous studies noting memory impairment in HIV+ patients as a result of immune system suppression, including the depletion of CD4+ cells. Therefore, it is necessary to monitor cognitive function in these patients and to implement measures to strengthen their memory performance.
Introduction
Medical knowledge concerning HIV is rapidly evolving, leading to a deeper understanding of the disease, including its immunology and clinical manifestations1. Memory impairment, a secondary effect due to a reduction in brain volume and a decreased CD4+ cell count, is one of the most severe complications in HIV+ patients2. The prevalence of this disorder in these patients varies from 30% to 60% and negatively affects their quality of life. The disruption of daily functioning, the development of clinical disorders, and non-compliance with treatment are among the complications associated with cognitive disorders in HIV+ patients3. The prevalence of non-compliance with treatment is six times higher in patients with memory impairment than in unaffected patients, leading to misuse of medications, treatment failure, drug resistance, increased viral load, increased risk of disease transmission to healthy people, and a higher mortality rate4.
The cells of the immune system, including CD4+ T lymphocytes, have a protective role in the brain, and the destruction of CD4+ helper T cells by the HIV virus compromises immunity against pathogenic agents. Immune system dysfunction and the depletion of CD4+ T cells predispose HIV+ patients to neurocognitive disorders and memory loss5. The number of CD4+ T cells and the viral load (HIV RNA) are among the laboratory markers that are routinely used to manage HIV/AIDS patients and to predict disease progression and/or treatment outcomes6.
A study by Sanford et al. in 2018 showed that there was a relationship between the number of CD4+ cells and the development of cognitive impairment, and this link was more prominent in patients who had CD4+ T cell counts between 200 and 350 and below 2007. Moreover, Fitri et al. in 2018 concluded that the number of CD4+ T cells, as a clinical indicator, could be used to predict the development of cognitive disorders, including memory impairment in HIV+ patients8. According to the above studies, a reduction in CD4+ T cells can be a predictor of neurocognitive defects and memory impairment in HIV+ patients9, 10. The early initiation of antiretroviral treatment can reduce the incidence and extent of cognitive impairment by elevating the number of CD4+ T cells11.
Finite mixture models are widely used in scientific investigation12. For example, Hong et al. (2021) formalized the concept of individualized mechanical ventilation (MV) strategy by combining finite mixture modeling (FMM) and a dynamic treatment regime (DTR)13.
As the complete treatment of infected patients is the most effective method of preventing the propagation of HIV in the wider population, care provided to patients should be among the priorities of AIDS control and prevention programs. Therefore, the aim of this study was to evaluate the relationship between the number of CD4+ T cells and memory impairment in HIV+ patients.
Methods
This was a descriptive-analytical study aiming to determine the relationship between CD4+ T cell count and memory function in HIV+ patients. After obtaining the necessary permissions and ethical approval, the subjects were selected by simple random sampling (using codes available in patients’ profiles) among HIV+ patients referred to the Behavioral Disorders Counseling Center of Kermanshah City. The appropriate sample size was estimated as n = 150 according to a study by Diranchi et al.14. The required data, including the CD4+ T cell counts and demographic information, were extracted from the patients’ files.
Inclusion criteria for participating in this study were age over 18 years, giving consent to participate, and having good physical and mental health. Learning disabilities, neurological diseases, psychological disorders, suffering from opportunistic infections, and a history of head trauma were regarded as exclusion criteria. A total of 150 patients were selected for enrollment in the study, and none were excluded from the study due to our exclusion criteria.
In this study, the Wechsler memory scale was used to assess memory function15. The test was administered and completed by the psychology expert of the center. This tool provides a visual scale for evaluating memory and identifies conditions such as dissociative identity disorder and memory deficits. The scale assesses learning ability, immediate retrieval, concentration and attention, orientation, and long-term memory retrieval. The subscales of the test include personal awareness, orientation, mental control, logical memory, and spatial memory. The total score for memory function is obtained by summing the scores for each of these subscales.
Statistical analysis
The data concerning memory function were divided into three classes using the regression method of finite mixture models (FMMs), and the results of the regression model were then interpreted for all three classes. STATA software (version 16) was used for data analysis, and the error rate for statistical significance was considered as P < 0.05.
| Class | N | Mean | SD | Percent | Cum. |
|---|---|---|---|---|---|
| I | 11 | 63.99 | 7.01 | 7 | 7 |
| II | 50 | 75.01 | 14.72 | 33 | 40 |
| III | 89 | 85.14 | 6.43 | 58 | 100 |
| Total | 150 | 80.25 | 13.77 | 100 | 100 |
| Class: I Response: Wechsler Memory Scale (WMS). Model: regression | |||||
| Coefficient Std. err. | z | P>|z| | [95% conf. interval] | ||
| WMS | |||||
| CD4 | .01 0.00 | 18.5 | <0.001 | .01 .01 | |
| Gender (Female/Male) | -9.38 0.49 | -19.06 | <0.001 | -10.34 -8.41 | |
| Age | .193 0.03 | 6.04 | <0.001 | .13 .25 | |
| Education | 4.02 0.29 | 13.58 | <0.001 | 3.44 4.60 | |
| intercept | 55.83 1.58 | 35.23 | <0.001 | 52.72 58.93 | |
| Class: II Response: WMS. Model: regression | |||||
| Coefficient Std. err. | z | P>|z| | [95% conf. interval] | ||
| WMS | |||||
| CD4 | 0 .00 0.00 | 0.6 | 0.950 | -.00 .01 | |
| Gender (Female/Male) | 9.68 2.38 | 4.21 | <0.001 | 5.95 14.31 | |
| Age | -0.33 0.14 | -2.22 | 0.026 | -.62 - .03 | |
| Education | 8.92 1.18 | 7.69 | <0.001 | 6.56 11.00 | |
| intercept | 54.58 7.52 | 7.25 | <0.001 | 39.83 69.33 | |
| Class: III Response: WMS. Model: regression | |||||
| WMS | |||||
| CD4 | 0.00 0.00 | 1.4 | 0.138 | -.00 .01 | |
| Gender (Female/Male) | 4.83 3.40 | 1.4 | 0.156 | -1.84 11.51 | |
| Age | .342 0.20 | 1.65 | 0.099 | -.066 0.750 | |
| Education | -3.26 2.14 | -1.52 | 0.127 | -7.46 0.93 | |
| intercept | 68.45 2.14 | 5.58 | <0.001 | 44.40 92.50 | |
Results
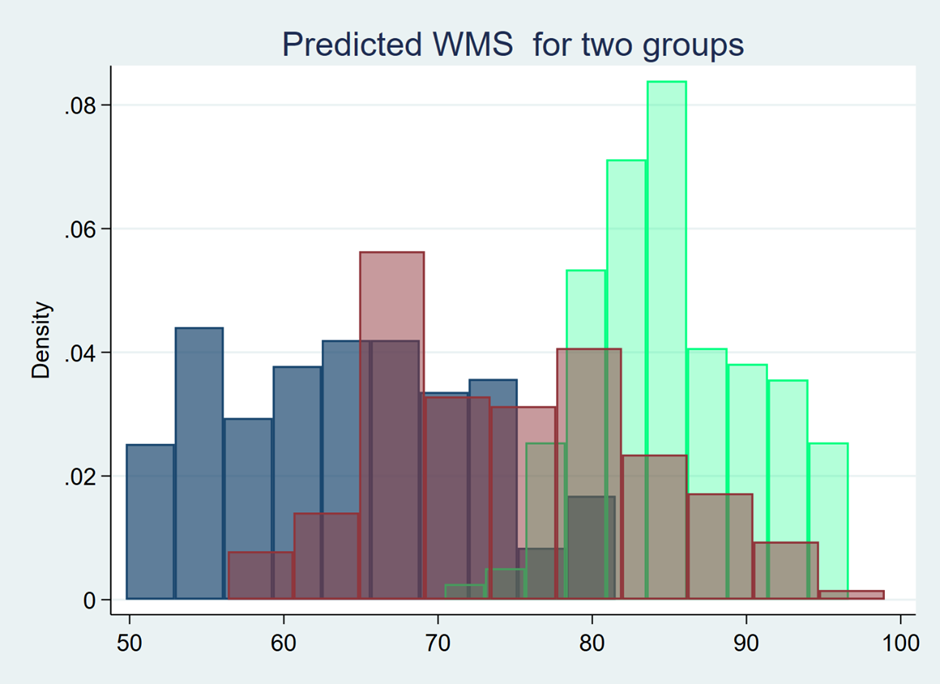
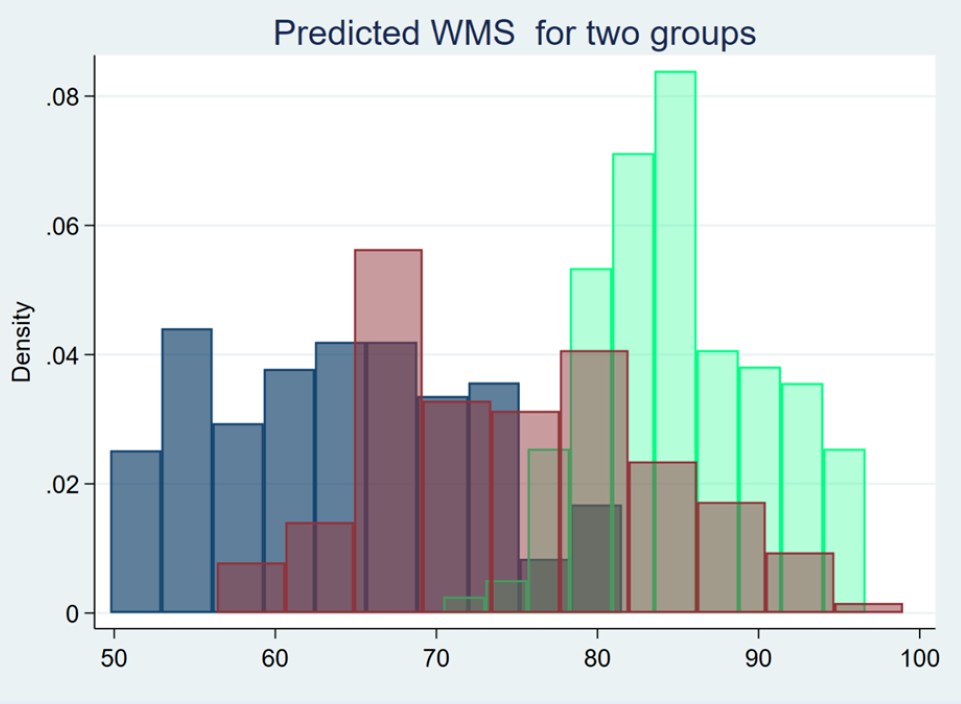
The present study evaluated the memory functions of 150 patients with HIV infection. The descriptive analysis of the data using a density plot showed that the distribution of memory function scores in these patients possessed three separate modes. Therefore, three regression models using FMM were utilized to assess the risk of memory impairment in these patients (Figure 1).
The means ± standard deviations of the memory function scores of HIV+ patients in classes I, II, and III were 63.99 ± 7.02, 75.01 ± 14.72, and 85.14 ± 6.43, respectively, with the lowest score in class I compared to the other two classes. The ratios of patients with memory impairment in the three classes were 7%, 33%, and 58%, respectively (Table 1).
Regarding the risk factors for memory impairment in HIV+ patients, the predictor variables for the regression model were CD4+ T cell count, gender, age, and education level. The results showed that in patients with poorer memory (i.e., those in class I), scores decreased with increasing age (P < 0.001). Also, memory loss was more prominent in women than in men (P < 0.001), and a higher level of education had a positive effect on memory function (P < 0.001). The results also showed that a reduction in CD4+ T cell count increased the risk of memory loss in these patients (P < 0.001). In patients in classes II and III, those who had higher memory function scores than their peers in class I showed no significant relationship between CD4+ T cell count and memory loss (Table 2).
Discussion
This study was conducted in order to investigate the relationship between the number of CD4+ T cells and memory function in HIV+ patients. The results showed that the patients could be divided into three classes in terms of memory status, where the mean score for memory function was significantly lower in class I patients compared to those in classes II and III. According to the results of the FMM regression model, CD4+ T cells had a protective role against memory loss in HIV+ patients, as indicated by a significantly lower mean number of CD4+ T cells in class I patients than in their counterparts in classes II and III. Previous studies investigating the relationship between memory function and the number of CD4+ T cells have also confirmed this observation, noting that a drop in CD4+ T cell count below 200 could aggravate memory impairment in HIV+ patients8, 16, 17, 18, 19.
In this study, the average CD4+ cell count was 326, slightly below the mean count of 350 reported to be protective against memory loss in an investigation by Nicoletta et al. on 150 patients with HIV20. Progressive decline in CD4+ cell counts is associated with human immunodeficiency virus (HIV) disease progression21. Recent WHO guidelines advise that a CD4+ threshold of ≤ 200 cells/µL should be used to define patients who have advanced HIV disease22.
According to the results of the present study, memory function in HIV+ patients decreased with increasing age. Schouten et al. (2014) showed that HIV-infected individuals were more susceptible to inflammatory diseases that could directly affect multiple organs and indirectly affect cellular metabolic pathways, the circulatory system, and the neuronal system23. Studies using neuroimaging have demonstrated continuing cerebral atrophy in HIV+ patients with disease progression and failure to control the viral load24. The theory that HIV infection accelerates aging is based on a series of results obtained from studies of infected older adults, and the effect is thought to be associated with persistent stimulation of the immune system, chronic low-grade inflammation, and T-cell dysfunction, symptoms that are assumed to be age-related events25. Our findings are consistent with the observation that HIV infection and aging both activate the immune cells residing in the brain, triggering neuroinflammation and neurodegeneration and leading to the loss of cognitive function. Previous studies have also noted an association between increasing age and a decline in verbal memory in HIV+ patients, where the disease stage was reported to be one of the most important predictors of the intensity of verbal memory impairment26. In another study on individuals less than 25 years old, a reduced CD4+ T cell count was associated with a lower level of verbal learning. In addition, increasing age was associated with speech delay and poor cognitive memory27.
In the present study, the level of memory deterioration was more prominent in women compared to men, and a higher educational level had a positive impact on memory retention. Lumbanraja et al. (2018) showed that there was a relationship between red blood cell indices and CD4+ T cell counts in HIV-infected women of childbearing age28. Another study demonstrated that neurological symptoms indicating cognitive disability were more common in HIV+ women than in HIV+ men and that measures of viral load were higher for women than for men29. Despite limited available evidence to date, there are arguments suggesting differences in immune system responses between men and women, and this may explain the phenomena of a greater decrease in the number of CD4+ T cells in HIV+ females and a more pronounced decrease in their cognitive functions.
Conclusions
The results of the present study support the findings of previous studies suggesting a link between CD4+ T cell depletion and memory loss in HIV+ patients. The HIV infection suppresses aspects of the immune system, including CD4+ T helper cells, and leads to memory impairment. Thus, it is necessary for healthcare providers and practitioners to pay attention to memory deterioration and its association with CD4+ T cell count. Patients should be monitored for memory status in parallel with regular checking of their CD4+ T cell counts, and they should receive appropriate supportive and therapeutic measures if necessary. Our findings suggest that future studies should investigate the signaling pathways responsible for the differences in viral load and cognitive disorders between HIV+ men and HIV+ women.
Abbreviations
None.
Acknowledgments
None.
Author’s contributions
All authors equally contributed to this work, read and approved the final manuscript.
Funding
This research was financially supported by the Research and Technology Deputy of Ilam University of Medical Sciences.
Availability of data and materials
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The present study was approved by the research ethics committee of Ilam University of Medical Sciences under the code IR.MEDILAM.REC.1398.116.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
-
Melhuish
A.,
Lewthwaite
P.,
Natural history of HIV and AIDS. Medicine (Abingdon).
2018;
46
(6)
:
356-61
.
View Article Google Scholar -
Ripamonti
E.,
Clerici
M.,
The association of memory disorders and chronic HIV disease in the antiretroviral therapy era: a systematic literature review. HIV Medicine.
2020;
21
(1)
:
9-20
.
View Article PubMed Google Scholar -
Cohen
R.A.,
Harezlak
J.,
Schifitto
G.,
Hana
G.,
Clark
U.,
Gongvatana
A.,
Effects of nadir CD4 count and duration of human immunodeficiency virus infection on brain volumes in the highly active antiretroviral therapy era. Journal of Neurovirology.
2010;
16
(1)
:
25-32
.
View Article PubMed Google Scholar -
Vassallo
M.,
Fabre
R.,
Durant
J.,
Lebrun-Frenay
C.,
Joly
H.,
Ticchioni
M.,
A decreasing CD4/CD8 ratio over time and lower CSF-penetrating antiretroviral regimens are associated with a higher risk of neurocognitive deterioration, independently of viral replication. Journal of Neurovirology.
2017;
23
(2)
:
216-25
.
View Article PubMed Google Scholar -
Miranda
C.S.,
Roque
S.,
Santos
N.C.,
Nunes
J.C.L.P.,
Costa
P.S.,
Palha
J.A.,
Effector memory CD4+ T cells are associated with cognitive performance in a senior population. Neurology-Neuroimmunology Neuroinflammation.
2015;
2
(1)
:
1-8
.
View Article Google Scholar -
Hoffmann
C.J.,
Charalambous
S.,
Thio
C.L.,
Martin
D.J.,
Pemba
L.,
Fielding
K.L.,
Hepatotoxicity in an African antiretroviral therapy cohort: the effect of tuberculosis and hepatitis B. AIDS (London, England).
2007;
21
(10)
:
1301-8
.
View Article PubMed Google Scholar -
Sanford
R.,
Fellows
L.K.,
Ances
B.M.,
Collins
D.L.,
Association of brain structure changes and cognitive function with combination antiretroviral therapy in HIV-positive individuals. JAMA Neurology.
2018;
75
(1)
:
72-9
.
View Article PubMed Google Scholar -
Fitri
F.I.,
Rambe
A.S.,
Fitri
A.,
Correlation between lymphocyte CD4 count, treatment duration, opportunistic infection and cognitive function in human immunodeficiency virus-acquired immunodeficiency syndrome (HIV-AIDS) patients. Open Access Macedonian Journal of Medical Sciences.
2018;
6
(4)
:
643-7
.
View Article PubMed Google Scholar -
Grauer
O.M.,
Reichelt
D.,
Grüneberg
U.,
Lohmann
H.,
Schneider-Hohendorf
T.,
Schulte-Mecklenbeck
A.,
Neurocognitive decline in HIV patients is associated with ongoing T-cell activation in the cerebrospinal fluid. Annals of Clinical and Translational Neurology.
2015;
2
(9)
:
906-19
.
View Article PubMed Google Scholar -
Subra
C.,
Trautmann
L.,
Role of T lymphocytes in HIV neuropathogenesis. Current HIV/AIDS Reports.
2019;
16
(3)
:
236-43
.
View Article PubMed Google Scholar -
Ellis
R.J.,
Badiee
J.,
Vaida
F.,
Letendre
S.,
Heaton
R.K.,
Clifford
D.,
Group
CHARTER,
CD4 nadir is a predictor of HIV neurocognitive impairment in the era of combination antiretroviral therapy. AIDS (London, England).
2011;
25
(14)
:
1747-51
.
View Article PubMed Google Scholar -
Chen
J.,
On finite mixture models. Statistical Theory and Related Fields.
2017;
1
(1)
:
15-27
.
View Article PubMed Google Scholar -
Hong
Y.,
Chen
L.,
Pan
Q.,
Ge
H.,
Xing
L.,
Zhang
Z.,
Individualized Mechanical power-based ventilation strategy for acute respiratory failure formalized by finite mixture modeling and dynamic treatment regimen. EClinicalMedicine.
2021;
36
:
100898
.
View Article PubMed Google Scholar -
De Ronchi
D.,
Faranca
I.,
Berardi
D.,
Scudellari
P.,
Borderi
M.,
Manfredi
R.,
Risk factors for cognitive impairment in HIV-1-infected persons with different risk behaviors. Archives of Neurology.
2002;
59
(5)
:
812-8
.
View Article PubMed Google Scholar -
Drozdick
L.W.,
Raiford
S.E.,
Wahlstrom
D.,
Weiss
L.G.,
The Wechsler Adult Intelligence Scale—Fourth Edition and the Wechsler Memory Scale—Fourth Edition. 2018.. 2018
.
-
Avci
G.,
Sheppard
D.P.,
Tierney
S.M.,
Kordovski
V.M.,
Sullivan
K.L.,
Woods
S.P.,
A systematic review of prospective memory in HIV disease: from the laboratory to daily life. The Clinical Neuropsychologist.
2018;
32
(5)
:
858-90
.
View Article PubMed Google Scholar -
Balaini
N.,
Sharma
A.,
Sharma
S.,
Sharma
A.,
HIV associated neurocognitive dysfunction and its association with CD4 count in HIV positive patients-a hospital based study. International Journal of Research in Medical Sciences.
2017;
5
(10)
:
4259
.
View Article Google Scholar -
Long short-term memory neural networks for chinese word segmentation.. {P}roceedings of the 2015 conference on empirical methods in natural language processing.
2015
.
View Article Google Scholar -
Doyle
K.L.,
Weber
E.,
Morgan
E.E.,
Loft
S.,
Cushman
C.,
Villalobos
J.,
Neurobehavioral Research Program (HNRP) Group
HIV,
Habitual prospective memory in HIV disease. Neuropsychology.
2015;
29
(6)
:
909-18
.
View Article PubMed Google Scholar -
Ciccarelli
N.,
Grima
P.,
Fabbiani
M.,
Baldonero
E.,
Borghetti
A.,
Milanini
B.,
Baseline CD4(+) T-cell count and cardiovascular risk factors predict the evolution of cognitive performance during 2-year follow-up in HIV-infected patients. Antiviral Therapy.
2015;
20
(4)
:
433-40
.
View Article PubMed Google Scholar -
Kazemi
A.,
Djafarian
K.,
Speakman
J.R.,
Sabour
P.,
Soltani
S.,
Shab-Bidar
S.,
Effect of probiotic supplementation on CD4 cell count in HIV-infected patients: a systematic review and meta-analysis. Journal of Dietary Supplements.
2018;
15
(5)
:
776-88
.
View Article PubMed Google Scholar -
Organization
W.H.,
Guidelines for managing advanced HIV disease and rapid initiation of antiretroviral therapy, July 2017. 2017. 2017
.
-
Schouten
J.,
Wit
F.W.,
Stolte
I.G.,
Kootstra
N.A.,
van der Valk
M.,
Geerlings
S.E.,
AGEhIV Cohort Study Group
Cross-sectional comparison of the prevalence of age-associated comorbidities and their risk factors between HIV-infected and uninfected individuals: the AGEhIV cohort study. Clinical Infectious Diseases.
2014;
59
(12)
:
1787-97
.
View Article PubMed Google Scholar -
Pfefferbaum
A.,
Rogosa
D.A.,
Rosenbloom
M.J.,
Chu
W.,
Sassoon
S.A.,
Kemper
C.A.,
Accelerated aging of selective brain structures in human immunodeficiency virus infection: a controlled, longitudinal magnetic resonance imaging study. Neurobiology of Aging.
2014;
35
(7)
:
1755-68
.
View Article PubMed Google Scholar -
Pirrone
V.,
Libon
D.J.,
Sell
C.,
Lerner
C.A.,
Nonnemacher
M.R.,
Wigdahl
B.,
Impact of age on markers of HIV-1 disease. Future Virology.
2013;
8
(1)
:
81-101
.
View Article PubMed Google Scholar -
Seider
T.R.,
Luo
X.,
Gongvatana
A.,
Devlin
K.N.,
de la Monte
S.M.,
Chasman
J.D.,
Verbal memory declines more rapidly with age in HIV infected versus uninfected adults. Journal of Clinical and Experimental Neuropsychology.
2014;
36
(4)
:
356-67
.
View Article PubMed Google Scholar -
Nichols
S.L.,
Chernoff
M.C.,
Malee
K.,
Sirois
P.A.,
Williams
P.L.,
Figueroa
V.,
Memory
Executive Functioning Substudy of the Pediatric HIVAIDS Cohort Study
Learning and memory in children and adolescents with perinatal HIV infection and perinatal HIV exposure. The Pediatric Infectious Disease Journal.
2016;
35
(6)
:
649-54
.
View Article PubMed Google Scholar -
Association between red blood cell indices and CD4 count in HIV-positive reproductive women. IOP Conference Series: Earth and Environmental ScienceIOP Publishing 2018.
Google Scholar -
Royal
W.,
Cherner
M.,
Burdo
T.H.,
Umlauf
A.,
Letendre
S.L.,
Jumare
J.,
Associations between cognition, gender and monocyte activation among HIV infected individuals in Nigeria. PLoS One.
2016;
11
(2)
:
e0147182
.
View Article PubMed Google Scholar
Comments
Article Details
Volume & Issue : Vol 10 No 4 (2023)
Page No.: 5624-5629
Published on: 2023-04-30
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 3286 times
- PDF downloaded - 1349 times
- XML downloaded - 102 times
 Biomedpress
Biomedpress