Abstract
Background: Colorectal cancer is one of the most common malignancies with a high rate of liver metastasis, and various gene mutations have been proven to be involved in its pathogenesis. One of them is BRAF gene mutations, the most frequent of which—V600E—has a poor prognosis in patients with colon cancer.
Case presentation: By applying the FOLFIRINOX regimen and a specific maintenance strategy containing vemurafenib, we achieved disappearance of metastatic lesions, no recurrence, and good stability in a Kurdish patient with metastatic colon cancer harboring RASwt/ BRAF V600E.
Conclusion: Our therapeutic strategy allows the selection of the most appropriate treatment approach for patients.
Introduction
Colorectal cancer (CRC) is the second leading cause of cancer death and the third most common cancer worldwide1. Distant metastases occur in approximately 65% of cases. The liver is the most common site of CRC metastasis (20 — 30%), followed by the lungs (10 — 25%)2, 3. The survival rate is lower in men than in women, while the 5-year survival rate is poorer for rectal cancer than for colon cancer4. Generally, CRC affects 71% of the colon and 29% of the rectum5. Sporadic cases are observed in approximately 70% of patients, and environmental and dietary factors are mainly involved. Hereditary predisposition is noted in < 10% of patients and occurs owing to several genetic changes, which can be identified through specific genetic tests5, 6.
Based on the different molecular mechanisms involved in CRC, many treatment methods can be used. The most important molecular mechanisms underlying different subtypes of CRC are microsatellite instability, chromosomal instability, and DNA repair machinery error7. As a serine/threonine kinase, v-raf murine sarcoma viral oncogene homolog B1 (BRAF) is located downstream of the mitogen-activated protein kinase (MAPK)/epidermal growth factor receptor (EGFR) signaling pathway8. The V600E mutation of this gene permanently activates the MAPK pathway and is associated with epigenetic activation of MLH1, resulting in the microsatellite instability phenotype in patients with CRC9, 10. This mutation occurs in a specific subtype of CRC with a poor prognosis. After performing molecular and diagnostic tests, we determined that our patient had RAS-wt/BRAF V600E mutation; we immediately treated her with the FOLFIRINOX regimen and a specific maintenance therapy and obtained promising results.
CASE PRESENTATION
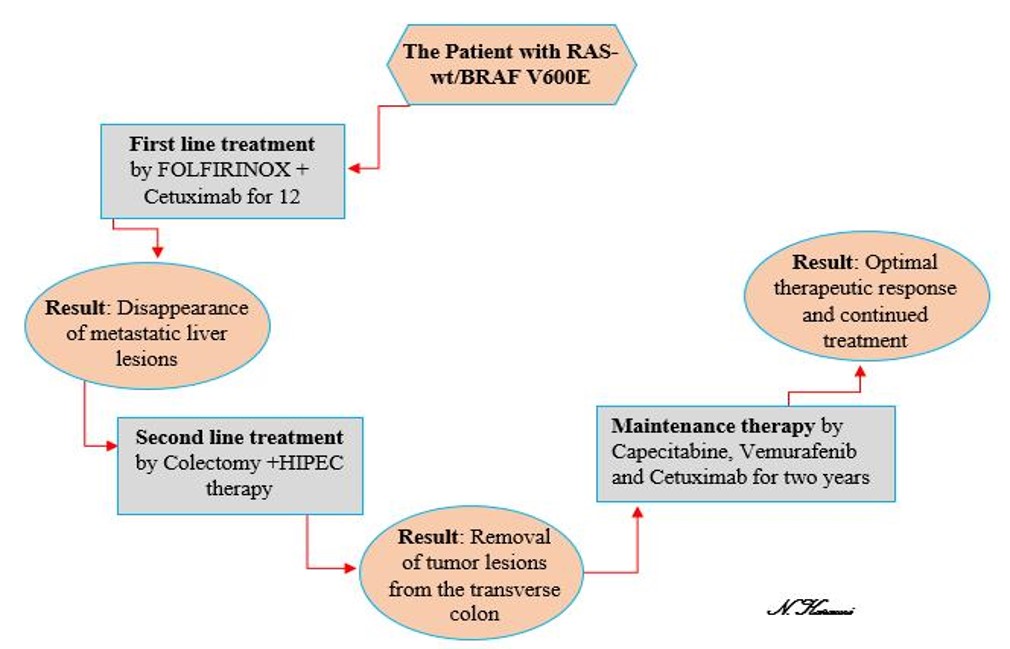
In February 2019, a 45-year-old Kurdish woman was referred to the Kermanshah Hematology and Oncology Clinic with symptoms of weight loss and abdominal pain. The patient had no relevant clinical history. Following a physical examination of the patient, the physician requested an abdominal CT. Laboratory examination indicated an increase in the CEA and CA19-9 levels and a decrease in the RBC indices. The complete blood count (CBC) testing results were as follows: platelet count: 284 (normal range: 120 – 450) × 109/L, hemoglobin level: 9.1 (normal range: 11.5 – 16.5) g/dL, and white blood cell (WBC) count: 6.6 (normal range: 3.5 – 11) × 109/L. CT illustrated a fungating tumor with a diameter of 40 × 26 mm seen in the transverse colon. Subsequently, PET-CT displayed primary lesions as mural thickening in the splenic flexure of the transverse colon with mesenteric lymph node nvolvement and multiple metastatic lesions on the right side of the liver. A panel of genetic tests, including tests for KRAS, NRAS, PLD1, and BRAF mutations, was performed; only BRAF mutation (V600E) tested positive. Based on these findings, the patient was diagnosed with disseminated colon cancer and treated with 12 cycles of the FOLFIRINOX regimen (leucovorin, 5-fluorouracil [FU], irinotecan, and oxaliplatin) and cetuximab (Erbitux) therapy. The metastatic lesions resolved within 2 weeks after the chemotherapy courses. Thereafter, total colectomy and hyperthermic intraperitoneal chemotherapy were conducted in the transverse colon, and the tumor lesions were removed.
Pathological examination did not show any tumors. Owing to the good response and poor prognosis of BRAF mutation in the colon tumor, the patient received a maintenance therapy regimen consisting of capecitabine, vemurafenib, and Erbitux for more than 2 years. During this time, the initial response to maintenance therapy was relatively good, and the CBC testing results were as follows: platelet count: 150 × 109/L, hemoglobin level: 11.6 g/dL, and WBC count: 5.4 × 109/L. Similarly, there was no evidence of residual invasive tumor in the colon pathology report. The patient currently receives continual treatment with gradual tapering of these combinations and is followed up regularly.
DISCUSSION
The BRAF gene has 18 exons, and its gene locus is on chromosome 7 and functions as a proto-oncogene11. This serine/threonine protein kinase is an important signaling molecule in the EGFR-mediated MAPK pathway, the activation of which is mediated by GTPase/RAS8. This pathway and the closely related phosphatidylinositol 3-kinase/AKT signaling pathway play a substantial role in the transcription of genes involved in cancer progression. Therefore, abnormalities involved in the activation of these pathways are among the causes of many types of human cancer12. Approximately 70% of BRAF-activating mutations occur in malignant melanomas, and the most common mutation is V600E, which changes valine to glutamic acid in codon 600 and accounts for approximately 95% of mutations in this gene13. This substitution has been observed with a lower frequency in other types of human cancer, including CRC at 10 – 20%14. As revealed in a recent meta-analysis, tumors with BRAF V600E mutations occur mainly on the right side of the colon, with a higher prevalence in women and elderly populations (over 60 years of age)15.
Notably, there is a mutually exclusive relationship between KRAS mutation and BRAF V600E16. Accordingly, the treatment strategies for patients can differ. In patients with KRAS-wt, the recommended first-line treatment is the administration of FOLFOX or FOLFIRI regimens, cetuximab or panitumumab, and doublet chemotherapy. These may be considered for some patients with right-sided RAS-wt tumors to reduce cytotherapy17.
In our study, the patient had RAS-wt/BRAF V600E mutation, which was observed with transverse colon involvement and liver metastasis. Thus, we started treatment with FOLFIRINOX (FFX) plus cetuximab. FOLFIRINOX has been recommended and extensively prescribed as a first-line treatment for patients with pancreatic ductal adenocarcinoma since 201118. FFX includes leucovorin, FU, irinotecan, and oxaliplatin19, and this regimen plus cetuximab can be used in the first-line treatment of patients with metastatic CRC20. After 12 cycles of this regimen, the liver metastatic lesions of our patient resolved, and colectomy was then performed. Because of the favorable response and poor prognosis of BRAF V600E mutations in the colon tumor, the patient received maintenance therapy, which included capecitabine (chemotherapy drug), vemurafenib (BRAF inhibitor), and cetuximab (EGFR inhibitor) and continued for more than 2 years; to date, the results have been promising.
Maintenance therapy generally consists of a less toxic portion of the previous regimen or an entirely disparate combination to which the patient has not formerly been exposed20. This therapeutic strategy is performed in patients who have not responded to salvage therapy and is intended to extend the positive results achieved with first-line induction treatment21. Treatment continuation or medication break is left to the doctor’s discretion. The advantages of drug vacations or therapeutic breaks include less toxicity, fewer hospital visits, better quality of life, and cost saving20, 22.
CONCLUSION
The use of FOLFIRINOX for complete response and resolution of metastatic lesions in patients with colon cancer with RAS-wt/BRAF V600E mutation is rarely reported. Our study showed that this combination plus a maintenance therapy strategy can lead to a complete and sustained therapeutic response. This strategy allows the selection of the most appropriate therapeutic method for the patient, yielding benefits such as cost savings and fewer hospital referrals. Our findings emphasize the importance of further investigations through larger prospective studies.
Abbreviations
BRAF: V-raf murine sarcoma viral oncogene homolog B1, CBC: complete blood count, CRC: Colorectal Cancer, EGFR: Epidermal growth factor receptor, FFX: FOLFIRINOX, Hb; hemoglobin, HIPEC: Hyperthermic Intraperitoneal Chemotherapy, IHC: Immunohistochemistry, MAPK: mediated mitogen-activated protein kinase, PI3K: phosphatidylinositol 3-kinase, WBC: white blood cell.
Acknowledgments
The authors are grateful to all colleagues in the Clinic of Hematology and Oncology.
Author’s contributions
Mehrdad Payandeh & Noorodin Karami: Literature search, Clinical studies, Data acquisition, Data analysis; Noorodin Karami: Manuscript preparation, Manuscript review, Guarantor; Afshin Karami: Concepts, Design, Definition of intellectual content, Literature search, Manuscript editing. All authors read and approved the final manuscript.
Funding
None.
Availability of data and materials
None
Ethics approval and consent to participate
The procedures have been reviewed and approved by the ethics committee. All procedures were in accordance with the 1964 Declaration of Helsinki and its subsequent amendments or comparable ethical standards. Written informed consent was obtained from the patient to publish this case report and any accompanying images.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
-
Bray
F.,
Ferlay
J.,
Soerjomataram
I.,
Siegel
R.L.,
Torre
L.A.,
Jemal
A.,
Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a Cancer Journal for Clinicians.
2018;
68
(6)
:
394-424
.
View Article PubMed Google Scholar -
Deshwar
A.,
Margonis
G.A.,
Andreatos
N.,
Barbon
C.,
Wang
J.,
Buettner
S.,
Double KRAS and BRAF mutations in surgically treated colorectal cancer liver metastases: an international, multi-institutional case series. Anticancer Research.
2018;
38
(5)
:
2891-5
.
View Article PubMed Google Scholar -
Cirocchi
R.,
Trastulli
S.,
Abraha
I.,
Vettoretto
N.,
Boselli
C.,
Montedori
A.,
Non-resection versus resection for an asymptomatic primary tumour in patients with unresectable stage IV colorectal cancer. Cochrane Database of Systematic Reviews.
2012
.
View Article Google Scholar -
Maajani
K.,
Khodadost
M.,
Fattahi
A.,
Shahrestanaki
E.,
Pirouzi
A.,
Khalili
F.,
Survival rate of colorectal cancer in Iran: a systematic review and meta-analysis. Asian Pacific journal of cancer prevention. Asian Pacific Journal of Cancer Prevention.
2019;
20
(1)
:
13-21
.
View Article PubMed Google Scholar -
Ahmed
M.,
Colon cancer: a clinician's perspective in 2019. Gastroenterology Research.
2020;
13
(1)
:
1-10
.
View Article PubMed Google Scholar -
Hadjipetrou
A.,
Anyfantakis
D.,
Galanakis
C.G.,
Kastanakis
M.,
Kastanakis
S.,
Colorectal cancer, screening and primary care: A mini literature review. World Journal of Gastroenterology.
2017;
23
(33)
:
6049-58
.
View Article PubMed Google Scholar -
Molina-Cerrillo
J.,
San Román
M.,
Pozas
J.,
Alonso-Gordoa
T.,
Pozas
M.,
Conde
E.,
BRAF mutated colorectal cancer: new treatment approaches. Cancers (Basel).
2020;
12
(6)
:
1571
.
View Article PubMed Google Scholar -
Taieb
J.,
Lapeyre-Prost
A.,
Laurent Puig
P.,
Zaanan
A.,
Exploring the best treatment options for BRAF-mutant metastatic colon cancer. British Journal of Cancer.
2019;
121
(6)
:
434-42
.
View Article PubMed Google Scholar -
Ducreux
M.,
Chamseddine
A.,
Laurent-Puig
P.,
Smolenschi
C.,
Hollebecque
A.,
Dartigues
P.,
Molecular targeted therapy of BRAF-mutant colorectal cancer. Therapeutic Advances in Medical Oncology.
2019;
11
:
1758835919856494
.
View Article PubMed Google Scholar -
Wang
Z.,
Dai
W.P.,
Zang
Y.S.,
Complete response with fluorouracil and irinotecan with a BRAFV600E and EGFR inhibitor in BRAF-mutated metastatic colorectal cancer: a case report. OncoTargets and Therapy.
2019;
12
:
443-7
.
View Article PubMed Google Scholar -
Mehta
A.,
Gupta
G.,
Nathany
S.,
The mala fides of BRAF in oncogenesis. Journal of Current Oncology..
2019;
2
(2)
:
37
.
View Article Google Scholar -
Wan
P.T.,
Garnett
M.J.,
Roe
S.M.,
Lee
S.,
Niculescu-Duvaz
D.,
Good
V.M.,
Cancer Genome Project
Mechanism of activation of the RAF-ERK signaling pathway by oncogenic mutations of B-RAF. Cell.
2004;
116
(6)
:
855-67
.
View Article PubMed Google Scholar -
Sahin
I.H.,
Kazmi
S.M.,
Yorio
J.T.,
Bhadkamkar
N.A.,
Kee
B.K.,
Garrett
C.R.,
Rare though not mutually exclusive: a report of three cases of concomitant KRAS and BRAF mutation and a review of the literature. Journal of Cancer.
2013;
4
(4)
:
320-2
.
View Article PubMed Google Scholar -
Taieb
J.,
Zaanan
A.,
Le Malicot
K.,
Julié
C.,
Blons
H.,
Mineur
L.,
Prognostic effect of BRAF and KRAS mutations in patients with stage III colon cancer treated with leucovorin, fluorouracil, and oxaliplatin with or without cetuximab: a post hoc analysis of the PETACC-8 trial. JAMA Oncology.
2016;
2
(5)
:
643-53
.
View Article PubMed Google Scholar -
Wang
J.,
Shen
J.,
Huang
C.,
Cao
M.,
Shen
L.,
Clinicopathological significance of BRAFV600E mutation in colorectal cancer: an updated meta-analysis. Journal of Cancer.
2019;
10
(10)
:
2332-41
.
View Article PubMed Google Scholar -
Caputo
F.,
Santini
C.,
Bardasi
C.,
Cerma
K.,
Casadei-Gardini
A.,
Spallanzani
A.,
BRAF-mutated colorectal cancer: clinical and molecular insights. International Journal of Molecular Sciences.
2019;
20
(21)
:
5369
.
View Article PubMed Google Scholar -
Folprecht
G.,
Martinelli
E.,
Mazard
T.,
Modest
D.P.,
Tsuji
A.,
Esser
R.,
Triplet chemotherapy in combination with anti-EGFR agents for the treatment of metastatic colorectal cancer: current evidence, advances, and future perspectives. Cancer Treatment Reviews.
2022;
102
:
102301
.
View Article PubMed Google Scholar -
Franck
C.,
Canbay
A.,
Malfertheiner
P.,
Venerito
M.,
Maintenance therapy with FOLFIRI after FOLFIRINOX for advanced pancreatic ductal adenocarcinoma: a retrospective single-center analysis. Journal of Oncology.
2019;
2019
:
5832309
.
View Article PubMed Google Scholar -
Hatachi
Y.,
Mohan
S.R.,
Kotake
T.,
Satake
H.,
Okita
Y.,
Yasui
H.,
FOLFIRINOX as First-line Chemotherapy in Japanese Patients Suffering from Metastatic Pancreatic Cancer (KOBE FOLFIRINOX Study). Cancer Diagnosis & Prognosis.
2022;
2
(1)
:
101-6
.
View Article PubMed Google Scholar -
Aprile
G.,
Giuliani
F.,
Lutrino
S.E.,
Fontanella
C.,
Bonotto
M.,
Rihawi
K.,
Maintenance therapy in colorectal cancer: moving the Artillery down while keeping an eye on the enemy. Clinical Colorectal Cancer.
2016;
15
(1)
:
7-15
.
View Article PubMed Google Scholar -
Van Cutsem
E.,
Cervantes
A.,
Nordlinger
B.,
Arnold
D.,
Guidelines Working Group
ESMO,
Metastatic colorectal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology : Official Journal of the European Society for Medical Oncology.
2014;
25
:
iii1-9
.
View Article PubMed Google Scholar -
Sonbol
M.B.,
Mountjoy
L.J.,
Firwana
B.,
Liu
A.J.,
Almader-Douglas
D.,
Mody
K.,
The role of maintenance strategies in metastatic colorectal cancer: A systematic review and network meta-analysis of randomized clinical trials. JAMA oncology.
2020;
6
(3)
:
e194489
.
View Article PubMed Google Scholar
Comments
Article Details
Volume & Issue : Vol 9 No 9 (2022)
Page No.: 5301-5305
Published on: 2022-09-30
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 3871 times
- PDF downloaded - 1449 times
- XML downloaded - 0 times
 Biomedpress
Biomedpress



