Abstract
Introduction: Vitamin D deficiency has become quite prevalent and is known to cause a great many diseases. Numerous studies have investigated the association between vitamin D deficiency and gestational diabetes, and the results are somewhat contradictory. The present study deals with the relationship between the vitamin D deficiency and gestational diabetes.
Methods: Two researchers made use of MeSH, Scopus, PubMed database, Science Direct, the Cochrane Library, the Web of Science, CINAHL, and Google Scholar search engines to identify qualified studies and articles carried out and published before August 2017 and reported the risk of gestational diabetes developing as a result of vitamin D deficiency. The association between the two conditions was measured using odds ratios (ORs) with 95% confidence intervals (CIs). Funnel plots, Egger’s, and Begg’s tests were also used to assess publication bias. All analysis was done by STATA (version 11.2).
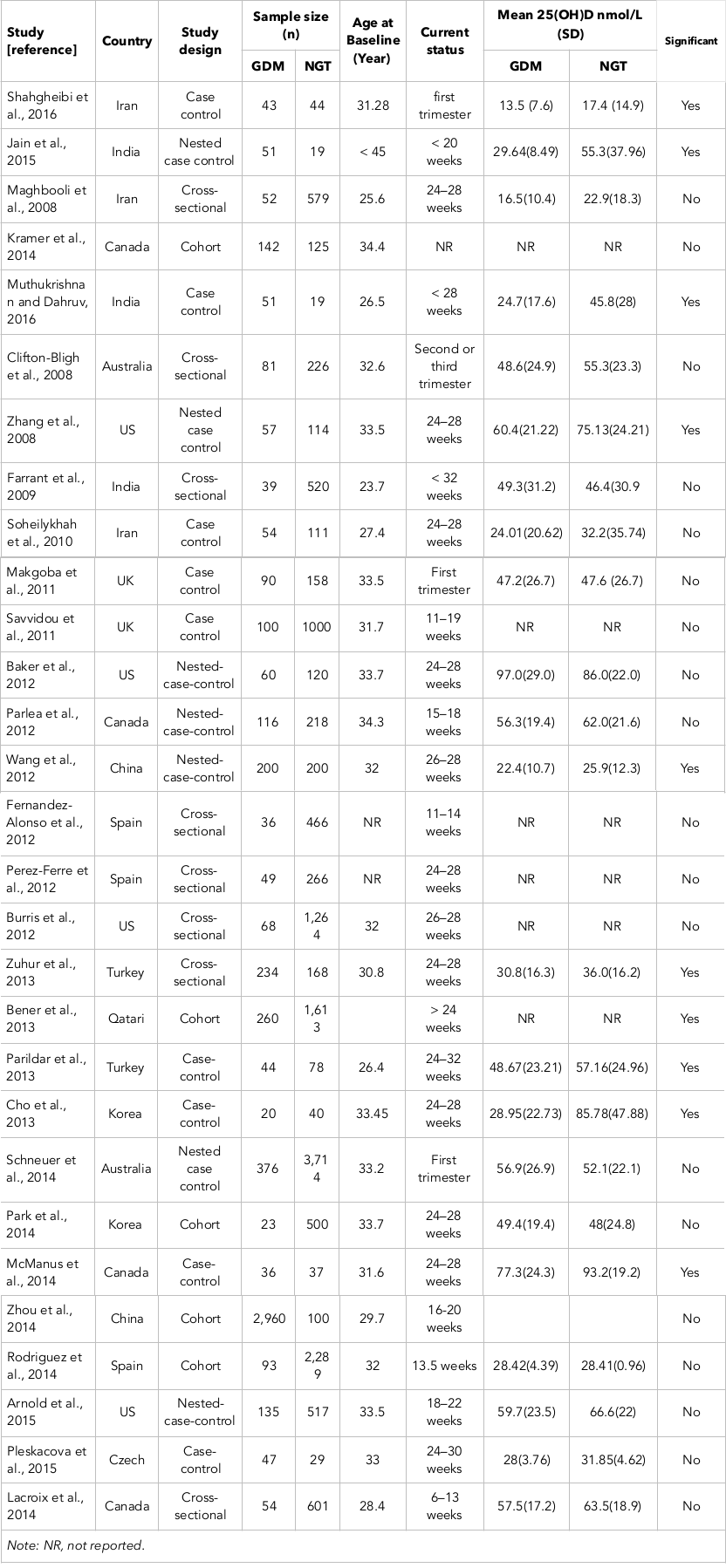
Results: Twenty-nine eligible studies with a total of 14,497 participants were included in the final analysis. Overall, maternal vitamin D insufficiency was significantly associated with a greater risk of gestational diabetes (OR = 1.15; 95% CI, 1.00-1.30; p<0.001). Serum 25OHD was significantly lower in participants with gestational diabetes mellitus than in those with natural glucose tolerance (−29.7 nmol/L, 95% CI, −36.6 to −22.8; p=0.000).
Conclusion: According to the current meta-analysis results, vitamin D deficiency is correlated with the risk of gestational diabetes development.
Introduction
Vitamin D deficiency is a costly health problem worldwide, and about one billion individuals in the world suffer from it Holick, 2010. Vitamin D is necessary agent for the regulation of mineral metabolism and skeletal health. Therefore, it plays a significant role in the health, growth, and fertility of humans Brown et al., 1999Hagenau et al., 2009.
Vitamin D deficiency during pregnancy can be accompanied by numerous maternal and fetal symptoms, including, inter alia, insulin resistance, gestational diabetes mellitus (GDM), increased risk of preeclampsia, bacterial vaginitis, and increased rate of cesareans among mothers and autism, Type I diabetes, increased rate of fetal growth delay, increased rate of respiratory infection, low birth weight, increased rate of HIV transmission from mother to fetus, asthma, and eczema in infants Aghajafari et al., 2013Palacios et al., 2016. The effect of vitamin D deficiency on the emergence of certain chronic diseases like autoimmune diseases Arnson et al., 2007, systemic lupus erythematosus (SLE) Amital et al., 2010, multiple sclerosis (MS) Ascherio et al., 2010, and malignancies Trump et al., 2010 has also been recognized.
A poor vitamin D status has been proposed as one risk factor associated with the incidence of GDM. The need for vitamin D is higher in some stages of life, including the period of rapid growth for fetuses in the embryonic stage, infancy, early stages of childhood, puberty, and pregnancy Shahgheibi et al., 2016. Vitamin D deficiency is common in pregnancy, and it significantly increases the risk for preeclampsia, cesarean section (C/S), and GDM in pregnancies Gernand et al., 2014Grant et al., 2014Merewood et al., 2009Robinson et al., 2010Yap et al., 2014.
Recent evidence suggests that vitamin D receptors are expressed in a large number of other tissues, including those involved in the regulation of glucose metabolism such as muscle and pancreatic beta cells Jain et al., 2015. These receptors have a direct effect on pancreatic beta cells and are required for the normal production and secretion of insulin by the endocrine pancreas Kramer et al., 2014Maghbooli et al., 2008. Thus, vitamin D deficiency is related to alterations in blood glucose and insulin concentrations and in target tissue sensitivity to insulin Shahgheibi et al., 2016. Vitamin D replenishment restores insulin secretion and sensitivity in patients with Type 2 diabetes with established vitamin D deficiency Muthukrishnan and Dhruv, 2015. Therefore, it was hypothesized that GDM might result from pregnancy-induced insulin resistance and impaired secretion to compensate for it.
There has been a rapidly growing interest in the association between vitamin D and the risk of gestational diabetes mellitus, and many studies with various populations and outcomes have been conducted on this topic. It is very important to have an overall estimation of its association. Also, understanding the breadth and quality of the conducted studies is critical. Recently some meta-analyses have found that vitamin D insufficiency is associated with increased GDM risk Aghajafari et al., 2013Lu et al., 2016Poel et al., 2012Zhang et al., 2015. Despite these findings, the knowledge and understanding of the clinical importance and implications of this association are limited. Moreover, since the publication of these meta-analyses, additional studies on this topic have been published. These studies have other clinically important outcomes that have not yet been effectively summarized. In order to authenticate these studies, an updated meta-analysis seems to be imperative. Thus, the current study aimed to quantitatively evaluate the association between vitamin D status and risk of gestational diabetes.
Materials - Methods
Search Method
The present review study was conducted based on systematic article review protocol and meta-analysis (PRISMA) Moher et al., 2009. To prevent the occurrence of any error or mistake during the search phase of the study, a quality evaluation and data extraction were carried out by two independent researchers. The compliance between the results and the discrepancies of the findings, if any, were investigated by a third researcher. To identify relevant studies, two independent researchers performed an internet-based search of such databases as PubMed, Scopus, Science Direct, the Cochrane Library, the Web of Science, CINAHL, and Google Scholar search engines with the exertion of no time limitation until August 2017. Subsequently, the references cited in the articles were investigated to access other related studies. To perform searches in the relevant databases, the researchers used MeSH-equivalent keywords, including “vitamin D”, “25, 1-dehydroxy cholecalciferol”, “25-hydroxy Vitamin D”,and“25(OH)D” along with “gestational diabetes”. The meta-analyses were limited to studies published in English.
Inclusion and exclusion criteria
To do further research, the abstracts and titles were studied by two arbiters. In order for screening studies to be included in the current meta-analysis, they had to have examined the relationship between vitamin D and the risk of developing gestational diabetes; have studied healthy pregnant women or pregnant women diagnosed with no chronic symptoms; have made use of blood samples for laboratory tests; and have compared women with gestational diabetes with women featuring natural glucose tolerance (NGT). Studies done on pregnant women with chronic illness were out of the scope of the current study. Studies implemented on non-human creatures (i.e. animal studies),those in languages other than English, those that were meta-analyses or systematic considerations as well as those that presented insufficient data or were duplicate publications were also excluded.
Study quality was evaluated using the STROBE (strengthening the reporting of observational studies in epidemiology) statement von Elm et al., 2008 and assessed based on variables related to the study objectives, characteristics of the study population, clearly explained inclusion/exclusion criteria, and the data collection method as well as the validity, explicit findings, and appropriate data analysis methods of the studies. Non-qualified studies were excluded.
Data extraction
Data was extracted from qualifying papers according to standard protocol. The data collected included the name of the first author, country, publication year, sample size, age groups, current status, study design, assessment of vitamin D levels, and the effect estimate with 95% CI. When necessary, the authors of articles were contacted for supplementary data or clarification. Information was extracted from the authors by two reviewers working independently, and their findings were compared afterwards. Disagreements on the eligibility of a study were resolved through group discussion. The data was entered into a standardized data extraction form and eventually into Microsoft Excel.
Data synthesis and analysis
The effect estimates (RR and OR) reported with 95% CI were used as the measure of association between vitamin D status and the risk of GDM. To account for the variance in the ways in which and degrees to which studies control for potential confounding factors, the risk estimates were maximally adjusted for potential confounders. The statistical heterogeneity between studies was evaluated using Cochran’s Q and I2 statistics. Wherever the results of studies were heterogeneous, a random effects model was used in the meta-analysis. A subgroup analysis was carried out in search for possible causes of the heterogeneity. The pooled estimates and corresponding 95% confidence intervals were visually assessed with the use of Forest plots. To investigate the influence of each study on the overall risk estimate, a sensitivity analysis was performed by removing individual studies in turn. Publication bias was assessed qualitatively using Funnel plots, Egger’s regression, and the Begg-Mazumdar rank correlation tests.
Values of p<0.05 were considered to denote significance in the heterogeneity tests. Statistical analyses were carried out in the software applications R (version 3.2.1) and STATA (version 11.1). All statistical tests were two-sided.
Results
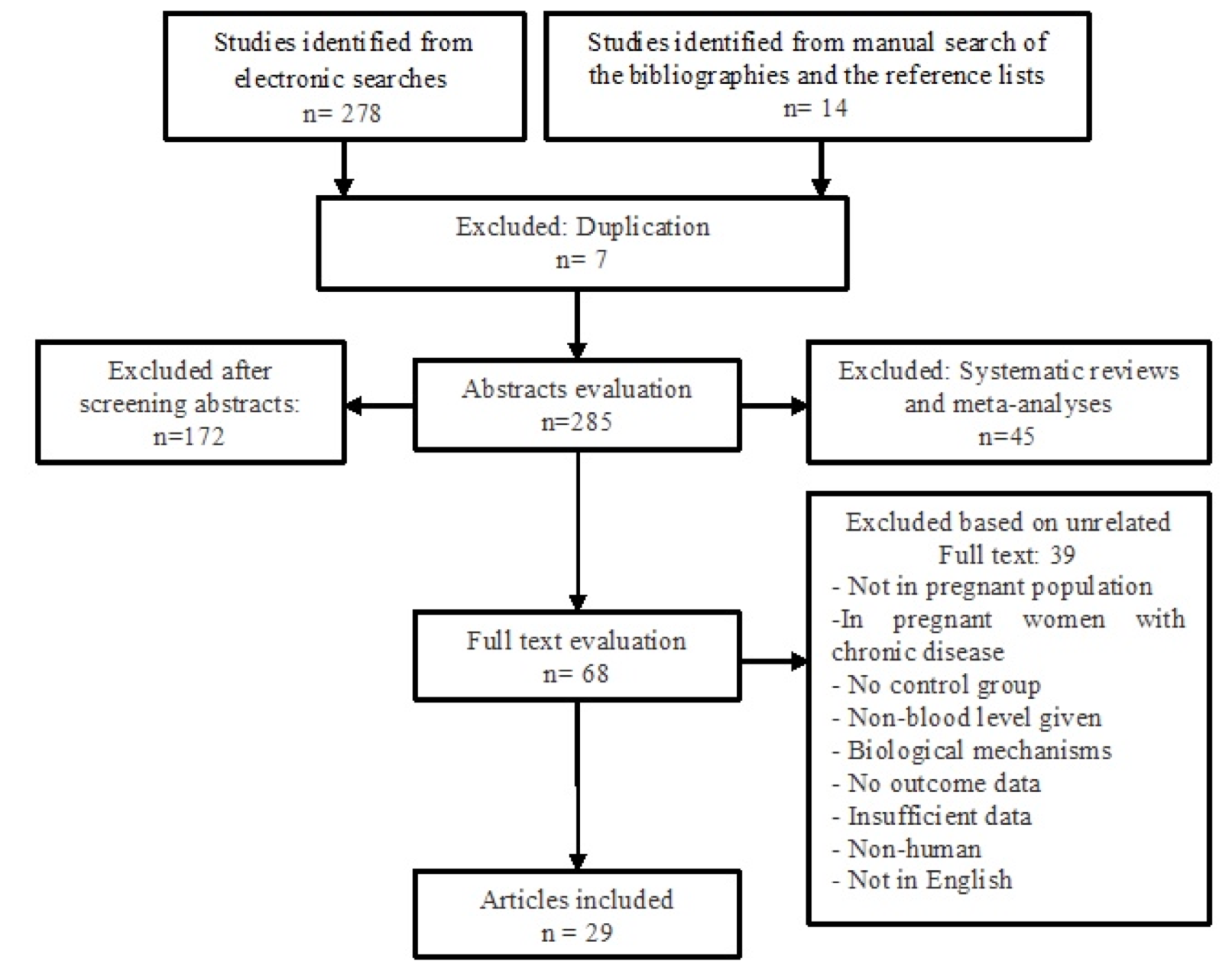
In the initial electronic search, a total of 278 potential articles were identified. A hand search of the bibliographies and reference lists of these articles identified 14 additional articles for a total of 292 articles identified through the literature search. After the initial screening of abstracts and titles, 224 papers were excluded based on the inclusion criteria, leaving 68 papers for a full text review. In a secondary screening and after the full text reviews, another 39 articles were excluded (four studies not in a pregnant population, eight studies in pregnant women with chronic disease, six studies for non-blood sampling, three studies for biological mechanisms, four studies for no outcome data, six studies for insufficient data, two studies for non-humans, and one study that was not in English). In total, 29 studies were selected for the final analysis ( Figure 1 ).
Description of the Studies
As seen in Table 1 , all 29 articles presented their findings as proportions. The studies used in this meta-analysis were published between 2008-2016 ( Table 1 ).
Main Analysis
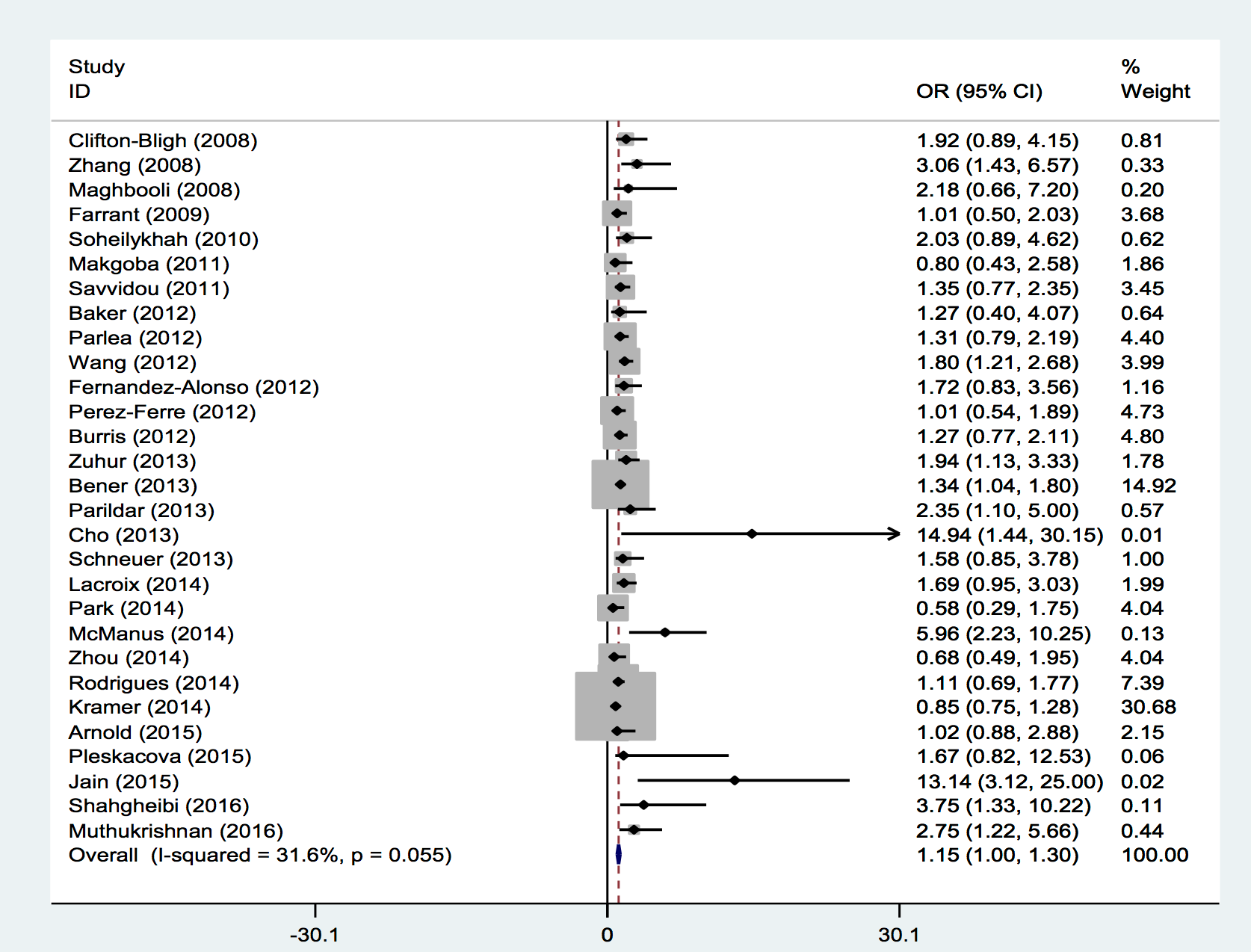
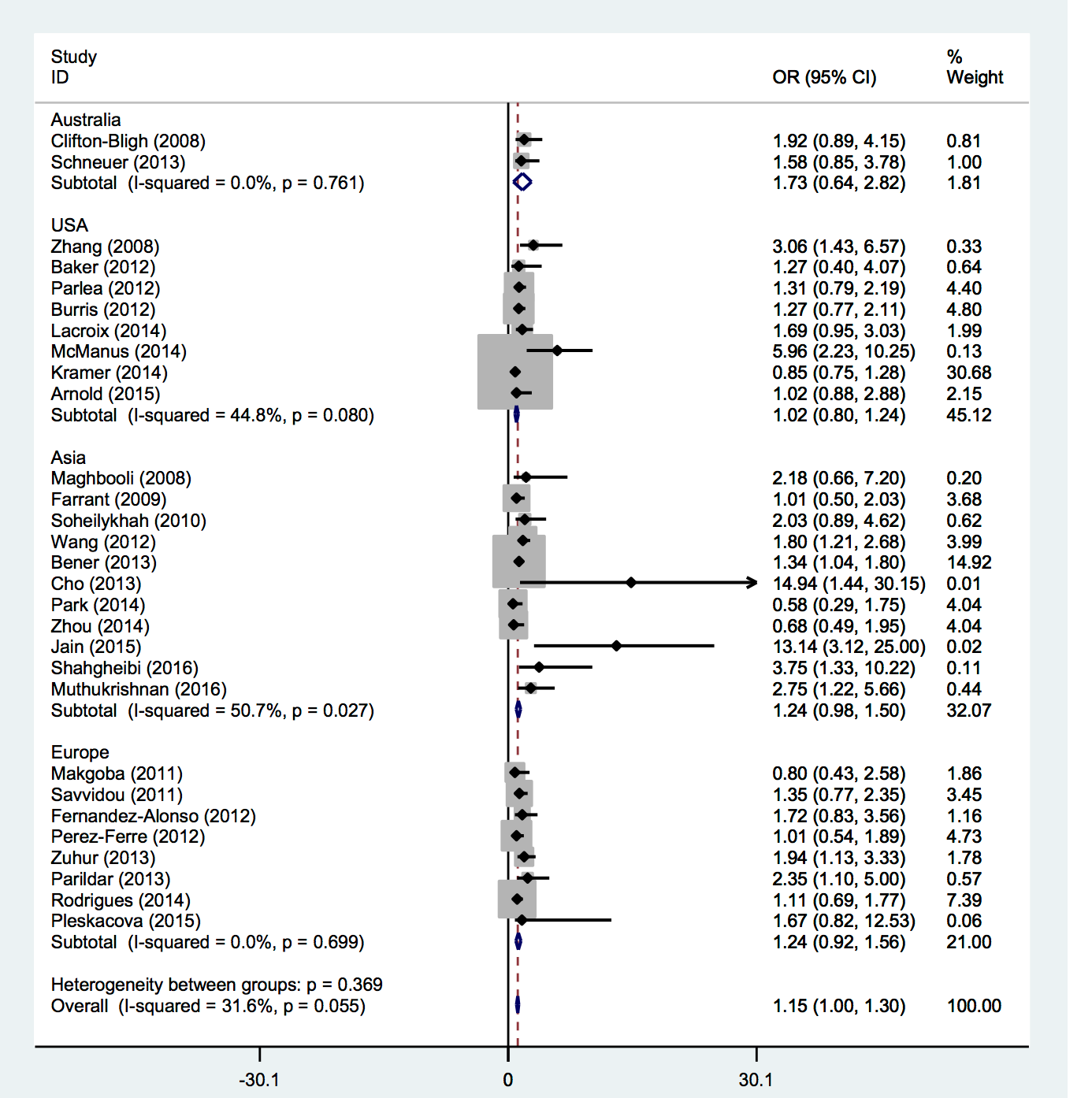
Those studies reporting odds ratios were pooled to quantify the association between 25-OHD insufficiency and GDM. Among the 29 studies, only ten showed a significant association between vitamin D status and risk of GDM; nineteen studies reported no significant association between vitamin D status and GDM. However, the present meta-analysis showed that vitamin D insufficiency was associated with increased gestational diabetes risk on a random effects model (OR = 1.15; 95% CI, 1.00-1.30; p<0.001) ( Figure 2 ). Little evidence of heterogeneity was observed among the studies (I2 = 31.6 %; p=0.055).
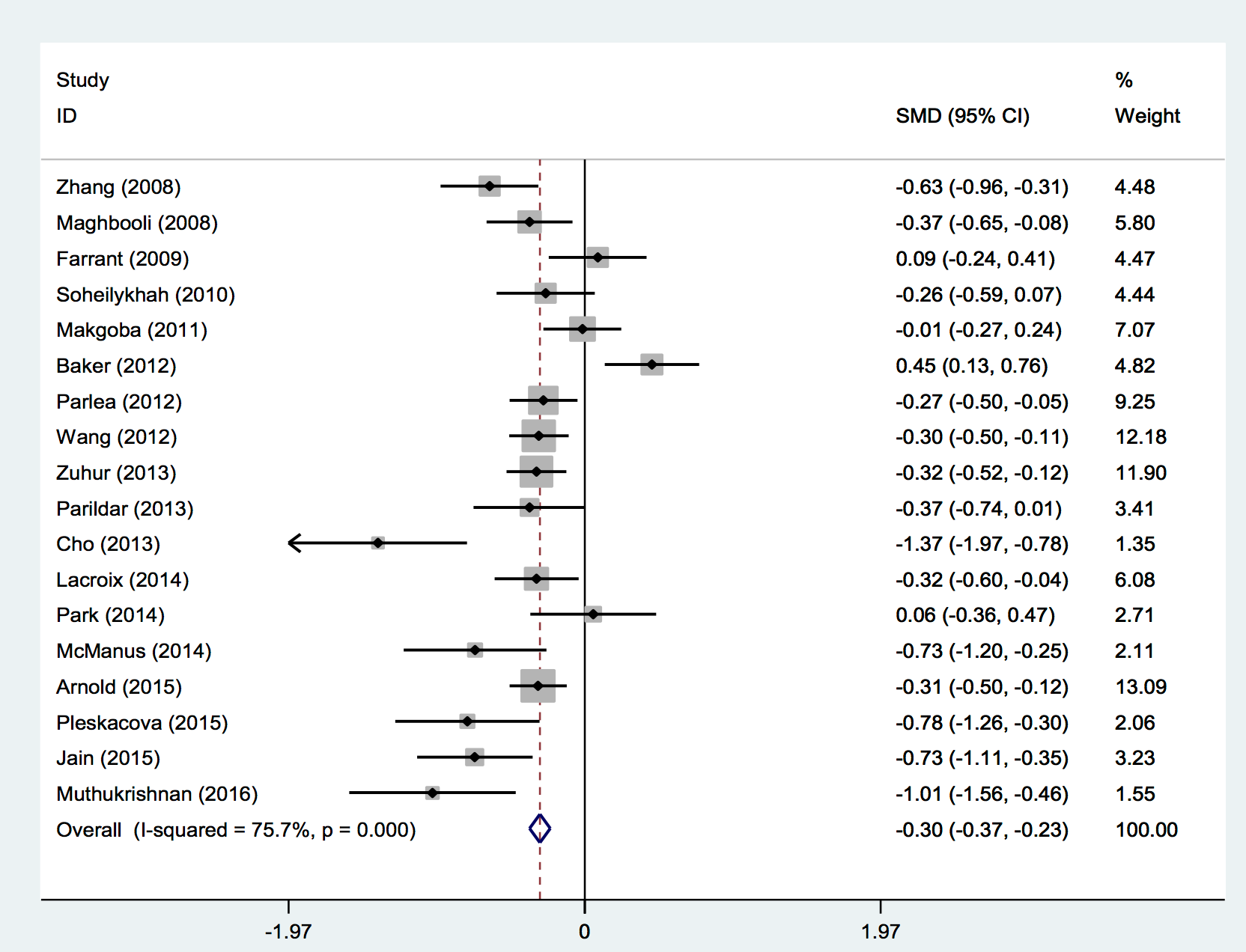
A meta-analysis was performed to determine whether there were significant differences between mean vitamin D levels among women with and without GDM. Figure 3 shows the results of a comparison of the mean differences of these studies. Based on a random-effects model meta-analysis, the pooled weighted mean difference was −29.7 nmol/L (95% CI −36.6 to −22.8), and significant heterogeneity was observed (I2 = 75.7%, p=0.000) ( Figure 3 ). These results indicated that pregnant women with GDM in these studies had significantly lower vitamin D levels than the comparison group and further demonstrated that vitamin D deficiency is significantly associated with an increased risk of GDM.
A sensitivity analysis was also conducted to examine the influence of various exclusion criteria on the overall risk estimate. After each study was sequentially excluded from the pooled analysis, the overall combined relative risk did not alter. The exclusion of any single study yielded similar results, and the conclusion was not affected by excluding any specific study.
We also evaluated the association between vitamin D status and the risk of GDM based on region. Selected studies were conducted in different countries. Of the 29 studies, 2 were conducted in Australia Clifton-Bligh et al., 2008Schneuer et al., 2014, eight were conducted in North America Arnold et al., 2015Baker et al., 2012Burris et al., 2012Kramer et al., 2014Lacroix et al., 2014McManus et al., 2014Parlea et al., 2012Zhang et al., 2008, eleven were conducted in Asia Bener et al., 2013Cho et al., 2013Farrant et al., 2009Jain et al., 2015Maghbooli et al., 2008Muthukrishnan and Dhruv, 2015Park et al., 2014Shahgheibi et al., 2016Soheilykhah et al., 2010Wang et al., 2012Zhou et al., 2014 and the remaining 8 were performed in Europe Fernandez-Alonso et al., 2012Makgoba et al., 2011Parildar et al., 2013Perez-Ferre et al., 2012Pleskacova et al., 2015Rodriguez et al., 2015Savvidou et al., 2011Zuhur et al., 2013. The result of our meta analysis showed no significant associations between vitamin D status and GDM risk based on geographic area (P for heterogeneity [Phet] =0.171, Figure 4 ).
Publication Bias
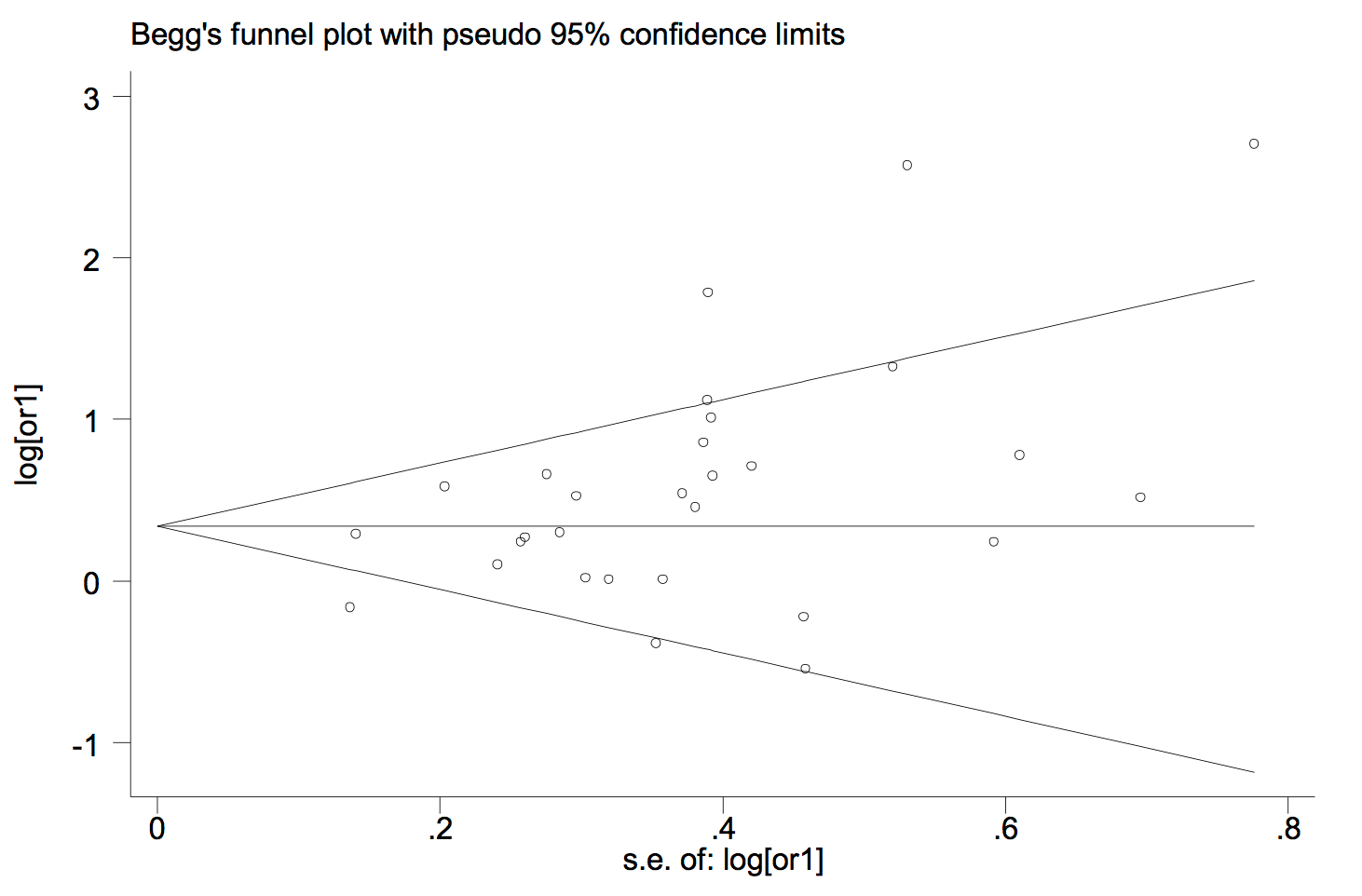
According to the publication bias tests, the effect of bias in these studies was not significant. No sign of publication bias was observed when Begg’s funnel plot was examined. Figure 5 presents the Begg’s funnel plot of the included trials related to vitamin D deficiency in GDM patients.
Discussion
The literature on vitamin D insufficiency in pregnancy is growing rapidly.The current meta-analysis was conducted to comprehensively review the literature and explore possible correlations between vitamin D status and the risk of GDM.
In this systematic review and quantitative meta-analysis of 29 observational studies, an association was found between insufficient vitamin D levels and the incidence of GDM. Women with GDM appear to have a significant lower serum 25OHD than women with NGT. The current results suggest that vitamin D deficiency may be an independent risk factor for GDM. These results are consistent with those of previous meta-analyses Aghajafari et al., 2013Lu et al., 2016Poel et al., 2012Zhang et al., 2015. Since the publication of themeta-analyses included in this research, further studies have been published. Moreover, the previous meta-analyses did not include several important observational studies.The current study attempts to fill this gap.
Obesity as well as a high maternal age are risk factors for both vitamin D deficiency and gestational diabetes; thus, they are potential confounders of the association between vitamin D deficiency and the incidence of GDM Poel et al., 2012. Most of the studies included in the current meta-analysis had been adjusted for maternal age and BMI; however, the association between vitamin D status and GDM was statistically significant.
Recent evidence has suggested that vitamin D deficiency or insufficiency is more common among pregnant women with limited sun exposure (e.g., those who live in cold climates or in northern latitudes) which may affect their vitamin D status; Those women who wear sunscreen or protective clothing and those from ethnic minority groups with darker skin Bodnar et al., 2007Hollis and Wagner, 2004Lee et al., 2007. The majority of vitamin D is generated in the skin under the influence of ultraviolet B radiation, which makes serum 25OHD levels dependent on seasons, with higher levels seen in spring and summer Poel et al., 2012.
There are also other risk factors that could be associated with GDM risk, including smoking, alcohol intake, gestational weight gain, and socioeconomic status. In the current study, it was observed that the adjusted models differed across the included studies; some of them did not adjust for several important confounding factors. The contribution of these confounding factors to the risk of gestational diabetes can go some distance in explaining conflicting results among different studies.
Other important points noted were that the included studies used different methods and criteria for the diagnosis of GDM, serum 25OHD levels were measured in different trimesters of pregnancy, definitions of cut-offs for 25-OHD insufficiency varied, and different techniques were used. The studies suggest that the methods used to quantify 25-OHD levels and to diagnose GDM may be important factors which could influence the final results Agarwal et al., 2005Shirazian et al., 2008.
There were several limitations in the current meta-analysis. First, the included studies varied in their definitions of the cut-off for 25-OHD insufficiency and in diagnostic criteria of GDM, and that could have influenced the pooled effect. Secondly, the researchers were unable to evaluate the impact of some important factors such as gestational weight gain, skin tone, and socioeconomic status on the correlation between maternal vitamin D status and the risk of GDM because of insufficient data in some studies. Also, data on sunlight exposure and dietary vitamin D intake was not available. Thirdly, the most adjusted odds ratio was used in meta-analysis. However, the adjusted models differed across the included studies. In some studies, the potential confounding factors could not be adjusted for, and therefore, the findings could not be pooled by adjusting confounding factors. Furthermore, inherent to any meta-analysis is the possibility of publication bias which is an inevitable problem. Finally, some studies associated with vitamin D status and GDM were not accessible.
Conclusion
The present meta-analysis indicated that pregnant women with gestational diabetes had significantly lower vitamin D levels than did those with normal glucose tolerance. The results further demonstrated a statistically significant association between maternal vitamin D insufficiency and the incidence of gestational diabetes. In conclusion, these findings suggest that low levels of 25(OH)D may be a risk factor in pregnancy. However, given the variety of study designs and the heterogeneity between the included studies, it is not reasonable to derive a definite conclusion.Further clinical trials are needed to verify this association and determine the explicit effect of vitamin D supplementation on the prevention of gestational diabetes.
Abbreviations
CIs: Confidence intervals
GDM: Gestational Diabetes Mellitus
NGT: Natural glucose tolerance
NR: Not reported
OR: Odds ratios
RR: Ratios risk
Author Contribution
Shahin Nargesi and Mansour Amraei designed the study and participated in writing the paper, Ayub Ghorbani and Ehsan Shirzadpour performed the meta-analysis, Seyedeh Fatemeh Mousavi, Ehsan Shirzadpour and Mahmoud Mohamadpour participated in writing the paper, Mansour Amraei provided data analysis and participated in writing the paper. All authors read and approved the manuscript.
References
-
M. M.
Agarwal,
G. S.
Dhatt,
J.
Punnose,
G.
Koster.
Gestational diabetes: Dilemma caused by multiple international diagnostic criteria. Diabetic Medicine.
2005;
22(12)
:
1731-1736
.
View Article PubMed Google Scholar -
F.
Aghajafari,
T.
Nagulesapillai,
P. E.
Ronksley,
S. C.
Tough,
M.
O’Beirne,
D. M.
Rabi.
Association between maternal serum 25-hydroxyvitamin D level and pregnancy and neonatal outcomes: Systematic review and meta-analysis of observational studies. BMJ (Clinical Research Ed.).
2013;
346(mar26 4)
:
f1169
.
-
H.
Amital,
Z.
Szekanecz,
G.
Szücs,
K.
Dankó,
E.
Nagy,
T.
Csépány,
Y.
Shoenfeld.
Serum concentrations of 25-OH vitamin D in patients with systemic lupus erythematosus (SLE) are inversely related to disease activity: Is it time to routinely supplement patients with SLE with vitamin D?. Annals of the Rheumatic Diseases.
2010;
69(6)
:
1155-1157
.
-
D. L.
Arnold,
D. A.
Enquobahrie,
C.
Qiu,
J.
Huang,
N.
Grote,
A.
VanderStoep,
M. A.
Williams.
Early pregnancy maternal vitamin D concentrations and risk of gestational diabetes mellitus. Paediatric and Perinatal Epidemiology.
2015;
29(3)
:
200-210
.
View Article PubMed Google Scholar -
Y.
Arnson,
H.
Amital,
Y.
Shoenfeld.
Vitamin D and autoimmunity: New aetiological and therapeutic considerations. Annals of the Rheumatic Diseases.
2007;
66(9)
:
1137-1142
.
View Article PubMed Google Scholar -
A.
Ascherio,
K. L.
Munger,
K. C.
Simon.
Vitamin D and multiple sclerosis. Lancet Neurology.
2010;
9(6)
:
599-612
.
View Article PubMed Google Scholar -
A. M.
Baker,
S.
Haeri,
C. A. Jr.
Camargo,
A. M.
Stuebe,
K. A.
Boggess.
First-trimester maternal vitamin D status and risk for gestational diabetes (GDM) a nested case-control study. Diabetes/Metabolism Research and Reviews.
2012;
28(2)
:
164-168
.
View Article PubMed Google Scholar -
A.
Bener,
A. O.
Al-Hamaq,
N. M.
Saleh.
Association between vitamin D insufficiency and adverse pregnancy outcome: Global comparisons. International Journal of Women’s Health.
2013;
5
:
523-531
.
View Article PubMed Google Scholar -
L. M.
Bodnar,
H. N.
Simhan,
R. W.
Powers,
M. P.
Frank,
E.
Cooperstein,
J. M.
Roberts.
High prevalence of vitamin D insufficiency in black and white pregnant women residing in the northern United States and their neonates. The Journal of Nutrition.
2007;
137(2)
:
447-452
.
View Article PubMed Google Scholar -
A. J.
Brown,
A.
Dusso,
E.
Slatopolsky.
Vitamin D. The American Journal of Physiology.
1999;
277(2 Pt 2)
:
F157-F175
.
PubMed Google Scholar -
H. H.
Burris,
S. L.
Rifas-Shiman,
K.
Kleinman,
A. A.
Litonjua,
S. Y.
Huh,
J. W.
Rich-Edwards,
M. W.
Gillman.
Vitamin D deficiency in pregnancy and gestational diabetes mellitus. American Journal of Obstetrics and Gynecology.
2012;
207(3)
:
182.e1-182.e8
.
-
G. J.
Cho,
S. C.
Hong,
M. J.
Oh,
H. J.
Kim.
Vitamin D deficiency in gestational diabetes mellitus and the role of the placenta. American Journal of Obstetrics and Gynecology.
2013;
209(6)
:
560.e1-560.e8
.
-
R. J.
Clifton-Bligh,
P.
McElduff,
A.
McElduff.
Maternal vitamin D deficiency, ethnicity and gestational diabetes. Diabetic Medicine.
2008;
25(6)
:
678-684
.
View Article PubMed Google Scholar -
H. J.
Farrant,
G. V.
Krishnaveni,
J. C.
Hill,
B. J.
Boucher,
D. J.
Fisher,
K.
Noonan,
C. H.
Fall.
Vitamin D insufficiency is common in Indian mothers but is not associated with gestational diabetes or variation in newborn size. European Journal of Clinical Nutrition.
2009;
63(5)
:
646-652
.
View Article PubMed Google Scholar -
A. M.
Fernández-Alonso,
E. C.
Dionis-Sánchez,
P.
Chedraui,
M. D.
González-Salmerón,
F. R.
Pérez-López,
the Spanish Vitamin D Women’s Health Research Group..
First-trimester maternal serum 25-hydroxyvitamin D₃ status and pregnancy outcome. International Journal of Gynaecology and Obstetrics: the Official Organ of the International Federation of Gynaecology and Obstetrics.
2012;
116(1)
:
6-9
.
View Article PubMed Google Scholar -
A. D.
Gernand,
H. N.
Simhan,
S.
Caritis,
L. M.
Bodnar.
Maternal vitamin D status and small-for-gestational-age offspring in women at high risk for preeclampsia. Obstetrics and Gynecology.
2014;
123(1)
:
40-48
.
View Article PubMed Google Scholar -
C. C.
Grant,
A. W.
Stewart,
R.
Scragg,
T.
Milne,
J.
Rowden,
A.
Ekeroma,
C. A. Jr.
Camargo.
Vitamin D during pregnancy and infancy and infant serum 25-hydroxyvitamin D concentration. Pediatrics.
2014;
133(1)
:
e143-e153
.
View Article PubMed Google Scholar -
T.
Hagenau,
R.
Vest,
T. N.
Gissel,
C. S.
Poulsen,
M.
Erlandsen,
L.
Mosekilde,
P.
Vestergaard.
Global vitamin D levels in relation to age, gender, skin pigmentation and latitude: An ecologic meta-regression analysis. Osteoporosis International.
2009;
20(1)
:
133-140
.
View Article PubMed Google Scholar -
M. F.
Holick.
The vitamin D deficiency pandemic: A forgotten hormone important for health. Public Health Reviews.
2010;
32(1)
:
267-263
.
View Article Google Scholar -
B. W.
Hollis,
C. L.
Wagner.
Assessment of dietary vitamin D requirements during pregnancy and lactation. The American Journal of Clinical Nutrition.
2004;
79(5)
:
717-726
.
PubMed Google Scholar -
M.
Jain,
S.
Kapry,
S.
Jain,
S.K.
Singh,
T.B.
Singh.
Maternal Vitamin D Deficiency: A Risk Factor for Gestational Diabetes Mellitus in North India. 2015
.
-
C. K.
Kramer,
B.
Swaminathan,
A. J.
Hanley,
P. W.
Connelly,
M.
Sermer,
B.
Zinman,
R.
Retnakaran.
Vitamin D and parathyroid hormone status in pregnancy: Effect on insulin sensitivity, β-cell function, and gestational diabetes mellitus. The Journal of Clinical Endocrinology and Metabolism.
2014;
99(12)
:
4506-4513
.
View Article PubMed Google Scholar -
M.
Lacroix,
M. C.
Battista,
M.
Doyon,
G.
Houde,
J.
Ménard,
J. L.
Ardilouze,
P.
Perron.
Lower vitamin D levels at first trimester are associated with higher risk of developing gestational diabetes mellitus. Acta Diabetologica.
2014;
51(4)
:
609-616
.
View Article PubMed Google Scholar -
J. M.
Lee,
J. R.
Smith,
B. L.
Philipp,
T. C.
Chen,
J.
Mathieu,
M. F.
Holick.
Vitamin D deficiency in a healthy group of mothers and newborn infants. Clinical Pediatrics.
2007;
46(1)
:
42-44
.
View Article PubMed Google Scholar -
M.
Lu,
Y.
Xu,
L.
Lv,
M.
Zhang.
Association between vitamin D status and the risk of gestational diabetes mellitus: A meta-analysis. Archives of Gynecology and Obstetrics.
2016;
293(5)
:
959-966
.
View Article PubMed Google Scholar -
Z.
Maghbooli,
A.
Hossein-Nezhad,
F.
Karimi,
A. R.
Shafaei,
B.
Larijani.
Correlation between vitamin D3 deficiency and insulin resistance in pregnancy. Diabetes/Metabolism Research and Reviews.
2008;
24(1)
:
27-32
.
View Article PubMed Google Scholar -
M.
Makgoba,
S. M.
Nelson,
M.
Savvidou,
C. M.
Messow,
K.
Nicolaides,
N.
Sattar.
First-trimester circulating 25-hydroxyvitamin D levels and development of gestational diabetes mellitus. Diabetes Care.
2011;
34(5)
:
1091-1093
.
View Article PubMed Google Scholar -
R.
McManus,
K.
Summers,
B.
de Vrijer,
N.
Cohen,
A.
Thompson,
I.
Giroux.
Maternal, umbilical arterial and umbilical venous 25-hydroxyvitamin D and adipocytokine concentrations in pregnancies with and without gestational diabetes. Horumon To Rinsho.
2014;
80(5)
:
635-641
.
View Article PubMed Google Scholar -
A.
Merewood,
S. D.
Mehta,
T. C.
Chen,
H.
Bauchner,
M. F.
Holick.
Association between vitamin D deficiency and primary cesarean section. The Journal of Clinical Endocrinology and Metabolism.
2009;
94(3)
:
940-945
.
View Article PubMed Google Scholar -
D.
Moher,
A.
Liberati,
J.
Tetzlaff,
D. G.
Altman,
P.
Group,
the PRISMA Group..
Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Medicine.
2009;
6(7)
:
e1000097
.
View Article PubMed Google Scholar -
J.
Muthukrishnan,
G.
Dhruv.
Vitamin D status and gestational diabetes mellitus. Indian Journal of Endocrinology and Metabolism.
2015;
19(5)
:
616-619
.
View Article PubMed Google Scholar -
C.
Palacios,
L. M.
De-Regil,
L. K.
Lombardo,
J. P.
Peña-Rosas.
Vitamin D supplementation during pregnancy: Updated meta-analysis on maternal outcomes. The Journal of Steroid Biochemistry and Molecular Biology.
2016;
164
:
148-155
.
View Article PubMed Google Scholar -
H.
Parildar,
A.
Dogruk Unal,
G.
Aksan Desteli,
O.
Cigerli,
N.
Guvener Demirag.
Frequency of Vitamin D deficiency in pregnant diabetics at Baskent University Hospital, Istanbul. Pakistan Journal of Medical Sciences.
2013;
29(1)
:
15-20
.
PubMed Google Scholar -
S.
Park,
H. K.
Yoon,
H. M.
Ryu,
Y. J.
Han,
S. W.
Lee,
B. K.
Park,
S. H.
Kim.
Maternal vitamin D deficiency in early pregnancy is not associated with gestational diabetes mellitus development or pregnancy outcomes in Korean pregnant women in a prospective study. Journal of Nutritional Science and Vitaminology.
2014;
60(4)
:
269-275
.
View Article PubMed Google Scholar -
L.
Parlea,
I. L.
Bromberg,
D. S.
Feig,
R.
Vieth,
E.
Merman,
L. L.
Lipscombe.
Association between serum 25-hydroxyvitamin D in early pregnancy and risk of gestational diabetes mellitus. Diabetic Medicine.
2012;
29(7)
:
e25-e32
.
View Article PubMed Google Scholar -
N.
Perez-Ferre,
M. J.
Torrejon,
M.
Fuentes,
M. D.
Fernandez,
A.
Ramos,
E.
Bordiu,
A. L.
Calle-Pascual.
Association of low serum 25-hydroxyvitamin D levels in pregnancy with glucose homeostasis and obstetric and newborn outcomes. Endocrine Practice.
2012;
18(5)
:
676-684
.
View Article PubMed Google Scholar -
A.
Pleskačová,
V.
Bartáková,
L.
Pácal,
K.
Kuricová,
J.
Bělobrádková,
J.
Tomandl,
K.
Kaňková.
Vitamin D status in women with gestational diabetes mellitus during pregnancy and postpartum. BioMed Research International.
2015;
2015
:
260624
.
View Article PubMed Google Scholar -
Y. H.
Poel,
P.
Hummel,
P.
Lips,
F.
Stam,
T.
van der Ploeg,
S.
Simsek.
Vitamin D and gestational diabetes: A systematic review and meta-analysis. European Journal of Internal Medicine.
2012;
23(5)
:
465-469
.
View Article PubMed Google Scholar -
C. J.
Robinson,
M. C.
Alanis,
C. L.
Wagner,
B. W.
Hollis,
D. D.
Johnson.
Plasma 25-hydroxyvitamin D levels in early-onset severe preeclampsia. American Journal of Obstetrics and Gynecology.
2010;
203(4)
:
366.e1-366.e6
.
-
A.
Rodriguez,
R.
García-Esteban,
M.
Basterretxea,
A.
Lertxundi,
C.
Rodríguez-Bernal,
C.
Iñiguez,
E.
Morales.
Associations of maternal circulating 25-hydroxyvitamin D3 concentration with pregnancy and birth outcomes. BJOG.
2015;
122(12)
:
1695-1704
.
View Article PubMed Google Scholar -
M. D.
Savvidou,
R.
Akolekar,
R. B.
Samaha,
A. P.
Masconi,
K. H.
Nicolaides.
Maternal serum 25-hydroxyvitamin D levels at 11(+0) -13(+6) weeks in pregnant women with diabetes mellitus and in those with macrosomic neonates. BJOG.
2011;
118(8)
:
951-955
.
View Article PubMed Google Scholar -
F. J.
Schneuer,
C. L.
Roberts,
C.
Guilbert,
J. M.
Simpson,
C. S.
Algert,
A. Z.
Khambalia,
N.
Nassar.
Effects of maternal serum 25-hydroxyvitamin D concentrations in the first trimester on subsequent pregnancy outcomes in an Australian population. The American Journal of Clinical Nutrition.
2014;
99(2)
:
287-295
.
View Article PubMed Google Scholar -
S.
Shahgheibi,
F.
Farhadifar,
B.
Pouya.
The effect of vitamin D supplementation on gestational diabetes in high-risk women: Results from a randomized placebo-controlled trial. Journal of Research in Medical Sciences.
2016;
21(1)
:
2
.
View Article PubMed Google Scholar -
N.
Shirazian,
M.
Mahboubi,
R.
Emdadi,
R.
Yousefi-Nooraie,
Z.
Fazel-Sarjuei,
N.
Sedighpour,
R.
Mozaffari-Kermani.
Comparison of different diagnostic criteria for gestational diabetes mellitus based on the 75-g oral glucose tolerance test: A cohort study. Endocrine Practice.
2008;
14(3)
:
312-317
.
View Article PubMed Google Scholar -
S.
Soheilykhah,
M.
Mojibian,
M.
Rashidi,
S.
Rahimi-Saghand,
F.
Jafari.
Maternal vitamin D status in gestational diabetes mellitus. Nutrition in Clinical Practice.
2010;
25(5)
:
524-527
.
View Article PubMed Google Scholar -
D. L.
Trump,
K. K.
Deeb,
C. S.
Johnson.
Vitamin D: Considerations in the continued development as an agent for cancer prevention and therapy. Cancer Journal (Sudbury, Mass.).
2010;
16(1)
:
1-9
.
-
E.
Elm,
D. G.
Altman,
M.
Egger,
S. J.
Pocock,
P. C.
Gøtzsche,
J. P.
Vandenbroucke,
the Iniciativa STROBE..
[The Strengthening the Reporting of Observational Studies in Epidemiology [STROBE] statement: Guidelines for reporting observational studies]. Gaceta Sanitaria.
2008;
22(2)
:
144-150
.
View Article PubMed Google Scholar -
O.
Wang,
M.
Nie,
Y. Y.
Hu,
K.
Zhang,
W.
Li,
F.
Ping,
X. P.
Xing.
Association between vitamin D insufficiency and the risk for gestational diabetes mellitus in pregnant Chinese women. Biomedical and Environmental Sciences.
2012;
25(4)
:
399-406
.
PubMed Google Scholar -
C.
Yap,
N. W.
Cheung,
J. E.
Gunton,
N.
Athayde,
C. F.
Munns,
A.
Duke,
M.
McLean.
Vitamin D supplementation and the effects on glucose metabolism during pregnancy: A randomized controlled trial. Diabetes Care.
2014;
37(7)
:
1837-1844
.
View Article PubMed Google Scholar -
C.
Zhang,
C.
Qiu,
F. B.
Hu,
R. M.
David,
R. M.
van Dam,
A.
Bralley,
M. A.
Williams.
Maternal plasma 25-hydroxyvitamin D concentrations and the risk for gestational diabetes mellitus. PLoS One.
2008;
3(11)
:
e3753
.
View Article PubMed Google Scholar -
M. X.
Zhang,
G. T.
Pan,
J. F.
Guo,
B. Y.
Li,
L. Q.
Qin,
Z. L.
Zhang.
Vitamin D Deficiency Increases the Risk of Gestational Diabetes Mellitus: A Meta-Analysis of Observational Studies. Nutrients.
2015;
7(10)
:
8366-8375
.
View Article PubMed Google Scholar -
J.
Zhou,
L.
Su,
M.
Liu,
Y.
Liu,
X.
Cao,
Z.
Wang,
H.
Xiao.
Associations between 25-hydroxyvitamin D levels and pregnancy outcomes: A prospective observational study in southern China. European Journal of Clinical Nutrition.
2014;
68(8)
:
925-930
.
View Article PubMed Google Scholar -
S. S.
Zuhur,
R. S.
Erol,
I.
Kuzu,
Y.
Altuntas.
The relationship between low maternal serum 25-hydroxyvitamin D levels and gestational diabetes mellitus according to the severity of 25-hydroxyvitamin D deficiency. Clinics (São Paulo).
2013;
68(5)
:
658-664
.
View Article PubMed Google Scholar
Comments
Downloads
Article Details
Volume & Issue : Vol 5 No 3 (2018)
Page No.: 2078-2095
Published on: 2018-03-19
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 8656 times
- Download PDF downloaded - 2220 times
- View Article downloaded - 0 times
 Biomedpress
Biomedpress