Mesenchymal Stem Cell-Derived Peptides in Regenerative Medicine: Mechanisms and Therapeutic Potential
- OXYZ Health & Wellness Academy, Research and Development Department, 59000, Kuala Lumpur, Malaysia
- Faculty of Medicine, University of Cyberjaya, Persiaran Bestari, Cyber 11, 63000 Cyberjaya, Selangor Darul Ehsan, Malaysia
- Faculty of Pharmacy, MAHSA University, Bandar Saujana Putra, 42610 Jenjarom, Selangor, Malaysia
- Atta-ur-Rahman Institute for Natural Products Discovery, Universiti Teknologi MARA, Puncak Alam Campus, 42300 Puncak Alam, Selangor, Malaysia
- Department of Food Science and Technology, Faculty of Agriculture, Ibb University, Aldhehar,70270, Ibb, Yemen
- Department of Biology, College of Sciences, University of Mosul, Iraq
Abstract
Mesenchymal stem cell-derived peptides (MSC-DPs) are increasingly recognized as pivotal mediators of MSC paracrine activity, providing a strictly defined, cell-free alternative to conventional whole-cell therapies. Current evidence, largely derived from in vitro and preclinical animal models, indicates that these low-molecular-weight peptides exert potent immunomodulatory, angiogenic, cytoprotective, and metabolic effects facilitating tissue repair. Peptidomic analyses of both human and xenogenic MSC sources have identified conserved peptide motifs enriched in cysteine-, glycine-, and proline-rich sequences, indicating robust potential for cross-species biological activity. Experimental data support the efficacy of MSC-DPs in enhancing epithelial repair, modulating macrophage polarization, and improving vascular responses; however, early translational observations remain limited and largely exploratory. Despite growing interest, progress toward clinical translation is constrained by substantial heterogeneity in MSC sources, isolation protocols, culture conditions, and peptide characterization methods, which complicate systematic comparisons across studies. Furthermore, a limited understanding of sequence–function relationships, coupled with challenges in peptide stability and in vivo delivery, further restricts therapeutic development. Continued advances in proteomics, computational prediction, and biomaterial-based delivery systems are expected to significantly improve the identification, standardization, and formulation of MSC-DPs. Overall, MSC-DPs represent a rapidly developing frontier in regenerative medicine with robust mechanistic foundations, yet their clinical translation will require rigorous validation and the harmonization of methodological approaches.
Introduction
Regenerative medicine has emerged as a transformative discipline aiming to restore, replace, or regenerate damaged tissues and organs through the integration of cellular, molecular, and bioengineering approaches1,2. Mesenchymal stem cells (MSCs) have garnered widespread attention as powerful biological tools due to their capacity for self-renewal, multilineage differentiation, and robust paracrine secretory activity3,4,5. The therapeutic action of MSCs was historically attributed to their ability to engraft, differentiate, and substitute for damaged cells6,7. An overview of the major components of the MSC secretome is presented in Figure 1. However, a growing body of evidence over the past decade indicates that most of their therapeutic effects are exerted indirectly via the release of soluble factors, such as cytokines, growth factors, extracellular vesicles, and, more recently, bioactive peptides8.
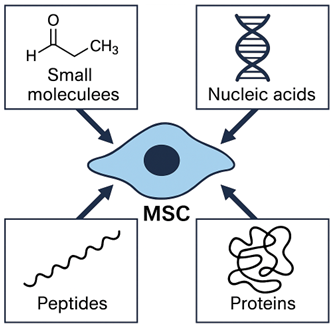
Overview of the mesenchymal stem cell (MSC) secretome. The MSC secretome comprises a diverse array of bioactive components—including small molecules (e.g., PGE₂, lactate), nucleic acids (e.g., miRNAs), peptides, and proteins (e.g., growth factors and cytokines)—which collectively mediate the paracrine activity and therapeutic effects of MSCs.
MSC-derived peptides (MSC-DPs) represent a unique and biologically active component of the MSC secretome; they comprise small amino acid sequences with diverse biological activities9,10. These peptides enhance tissue repair by modulating inflammation, oxidative stress, apoptosis, angiogenesis, and metabolism11,12. Due to their small size, they can penetrate tissues effectively; furthermore, they are stable in the extracellular environment, and their immunogenicity is significantly lower than that of full-length proteins or intact cells. Most importantly, they offer a cell-free therapeutic alternative that overcomes the limitations associated with live-cell transplantation, including low survival rates, potential tumorigenicity, and ethical or regulatory constraints13,14.
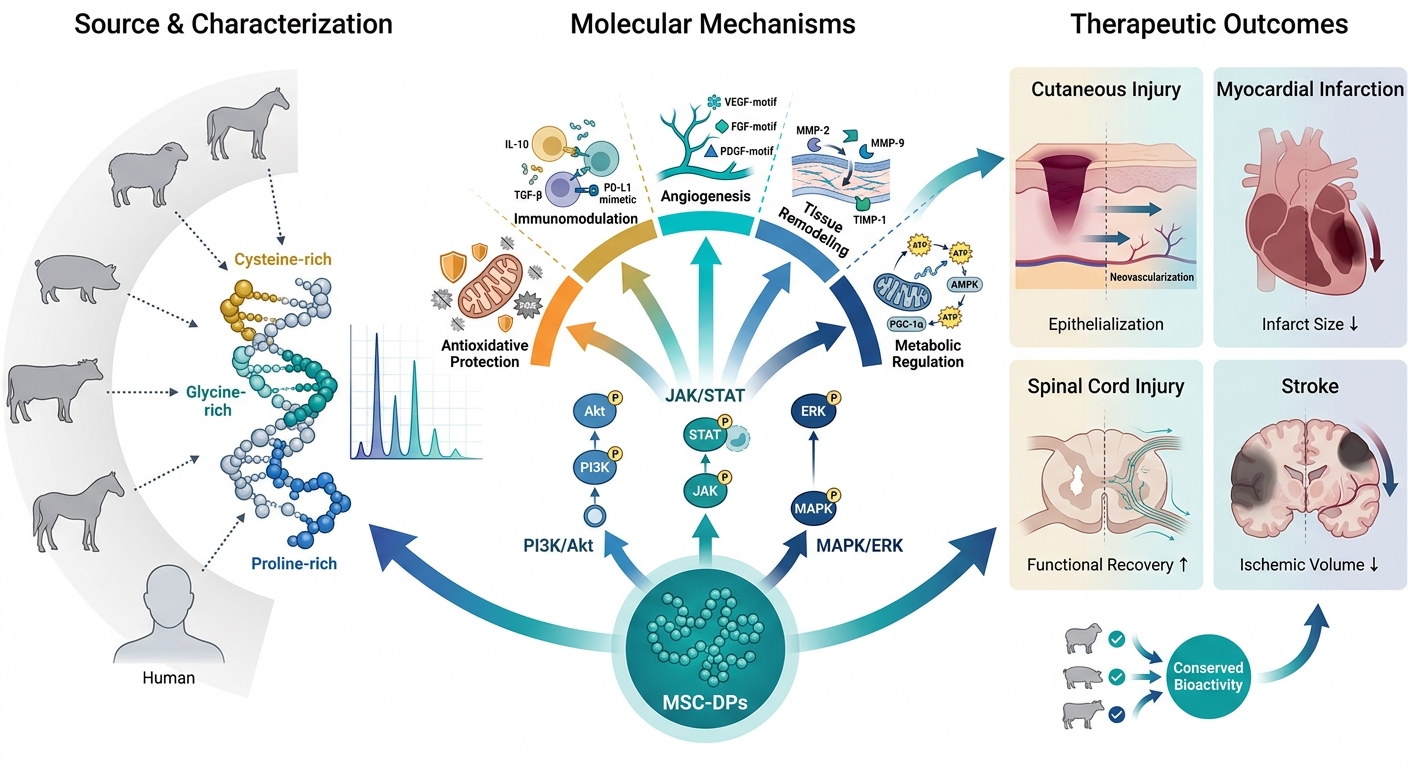
Both human and xenogenic MSCs secrete a complex secretome composed of cytokines, growth factors, extracellular vesicles, and bioactive peptides. Among these, MSC-DPs act as core paracrine mediators of regeneration, promoting angiogenesis, immunomodulation, antioxidation, and metabolic regulation across species (cf. Figure 2). Expanding beyond human-derived MSC-DPs, emerging evidence indicates that xenogenic MSCs—particularly those derived from ovine, porcine, and bovine tissues—represent promising alternative reservoirs of bioactive peptides with comparable immunomodulatory and regenerative capacities15. Studies on equine MSCs have confirmed the secretion of antimicrobial and pro-regenerative peptides with significant therapeutic activity across species boundaries16. Similarly, bovine and porcine MSCs have been characterized as rich peptide sources supporting tissue repair and translational research17,18.
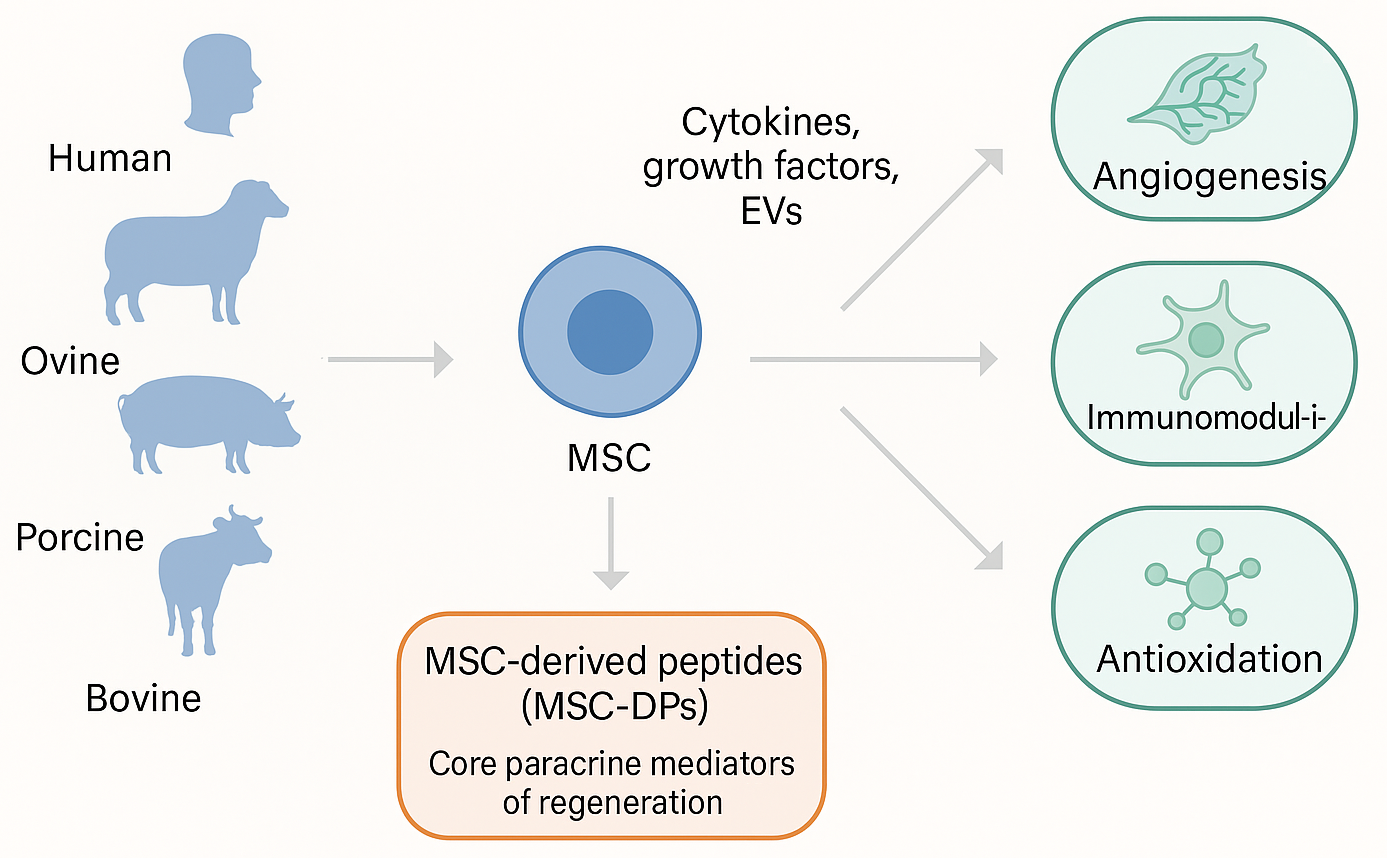
Biological sources and therapeutic functions of MSC-derived peptides (MSC-DPs). Mesenchymal stem cells (MSCs) isolated from both human and diverse xenogenic sources (e.g., ovine, porcine, and bovine) produce a complex secretome. Alongside cytokines, growth factors, and extracellular vesicles (EVs), MSC-DPs serve as core paracrine mediators that drive highly conserved therapeutic outcomes across species, including angiogenesis, immunomodulation, and antioxidation.
Evidence also demonstrates that xenogenic MSCs and their secreted factors can modulate immune responses across species, as observed in feline and canine models treated with xenogeneic MSC secretomes and cells19,20,21. Ovine umbilical cord MSCs have likewise been established as stable, immortalized sources suitable for scalable peptide isolation and comparative xenogenic studies22. These MSC-DPs demonstrate bioactivity equivalent to their human counterparts, with some exhibiting anti-inflammatory23, angiogenic24, and antioxidative activities across species25. Further evidence reinforces that xenogenic MSCs and their secreted factors can modulate the immune system across species, highlighting the translational potential of such peptides for preclinical and veterinary applications26,27. However, interspecies molecular compatibility, immunogenic epitopes, and peptide stability remain critical challenges requiring systematic research prior to clinical application28,29.
The isolation of MSC-DPs has been facilitated by advances in proteomics and peptidomics, particularly high-resolution mass spectrometry and bioinformatics-based peptide mapping. These technologies have enabled the identification of numerous peptides originating from MSC cytosolic proteins, membrane components, and extracellular matrix modulators30,31,32. Functionally, they replicate most of the classical MSC functions, such as promoting angiogenesis via vascular endothelial growth factor (VEGF)-like sequences or dampening inflammation through interleukin-10 (IL-10)–mimetic domains, thereby enhancing the treatment repertoire of MSC-based therapies33,34. Cell-to-peptide regenerative approaches bridge the translational gap in the emerging shift towards next-generation biologics with enhanced safety, scalability, and molecular accuracy35,36. MSC-DPs constitute a standardized, reproducible, and ethically sound therapeutic platform applicable across a wide range of pathological conditions, including cardiovascular, neurodegenerative, musculoskeletal, and metabolic disorders37.
This review aims to comprehensively address the biological origin, mechanism of action, and therapeutic potential of MSC-DPs. Emphasis is placed on their molecular mechanisms—ranging from paracrine signaling and immunomodulation to angiogenic and metabolic regulation—as well as their synergistic effects when combined with organ-specific peptides. Moreover, the discussion includes the relative advantages of peptide therapy over traditional cell therapy, existing limitations in peptide stability and delivery, and future prospects for clinical translation. Having outlined the scope of this review, the subsequent section explores the biological origin and composition of MSC-DPs, which form the basis of their functional properties.
Search Strategy
A comprehensive literature search was conducted to identify relevant studies elucidating the biological and therapeutic roles of mesenchymal stem cell–derived peptides (MSC-DPs). Multiple electronic databases, including PubMed, Scopus, and Web of Science, were systematically searched for peer-reviewed articles. The search strategy utilized varied combinations of keywords, such as “mesenchymal stem cells,” “MSC secretome,” “MSC-derived peptides,” “peptidome,” “extracellular vesicles,” “regenerative medicine,” and “cell-free therapy”. The literature search primarily focused on publications spanning the period from 2010 to 2025, although earlier seminal studies were included where contextually relevant. Only articles published in English that were directly pertinent to the topic were considered for inclusion. Both experimental and review studies were included to provide a comprehensive overview; conversely, studies lacking sufficient methodological clarity or direct relevance to MSC-derived peptides were excluded.
Biological Source and Composition of MSC-Derived Peptides
MSC-DPs originate from multiple intracellular and secretory compartments, collectively reflecting the dynamic proteolytic, metabolic, and paracrine activities of mesenchymal stromal cells38,39. Their structural diversity arises from regulated protein turnover, enzymatic cleavage of membrane structures, and active secretion into the extracellular space. These peptide subsets contribute to the multifunctional therapeutic actions attributed to the MSC secretome and constitute a core component of cell-free regenerative strategies40,41.
Cytosolic peptides are generated primarily through proteasomal degradation, autophagy-associated processing, and intracellular stress–response pathways42,43. They frequently contain cysteine-, glycine-, or proline-rich motifs that enhance peptide stability and support antioxidant activity. These peptides participate in redox regulation, mitochondrial homeostasis, and protection against apoptosis, particularly under inflammatory or oxidative microenvironmental stress44. Their biochemical signatures reflect the adaptive metabolic responses of MSCs and contribute to the cytoprotective, anti-inflammatory, and survival-enhancing properties observed in MSC-DP therapy45,46.
Membrane-associated peptides originate from transmembrane receptors, adhesion molecules, and extracellular domain-bearing proteins that undergo controlled enzymatic cleavage or ectodomain shedding47. These peptides retain functional ligand-binding or receptor-interactive sequences, enabling them to modulate immune cell recruitment, extracellular matrix remodeling, and intercellular signaling48. By influencing integrin activity, chemokine responsiveness, and stromal–immune interactions, membrane-derived peptides help shape the regenerative microenvironment and amplify MSC-driven paracrine mechanisms49.
Extracellular peptides represent the actively released fraction of the MSC secretome and include angiogenic, immunoregulatory, cytoprotective, and metabolic-regulating sequences50. These peptides are released via exocytosis, extracellular vesicle secretion, and non-vesicular paracrine pathways. Subsequently, they act directly on target tissues to modulate inflammation, stimulate angiogenesis, enhance metabolic recovery, and promote structural repair. Their demonstrated activity in xenogenic models highlights their evolutionary conservation and therapeutic potential across species51,52. A consolidated overview of representative MSC-DP classes, properties, and bioactivities is presented in Table 1.
Representative classes of MSC-derived peptides (MSC-DPs), their candidate sequences or functional motifs, cellular origin, and reported bioactivities identified across human and xenogenic MSC secretomes.
| Peptide Class / Compartment | MSC Source (Human / Xenogenic) | Candidate Sequence / Motif | Reported Bioactivity | References |
|---|---|---|---|---|
| Cytosolic mitochondrial stress–response peptide | Human placenta-derived MSCs | MOTS-c (MRWQEMGYIFYPRKLR) | Enhances mitochondrial homeostasis, activates AMPK, reduces ROS, protects MSCs under oxidative stress; contributes to cytoprotective & anti-inflammatory effects | |
| Cytosolic antioxidant / anti-apoptotic peptide | Human MSCs (aging-associated pathway) | Humanin (MAPRGFSCLLLL-TSEIDLPVKRRA) | Strong anti-apoptotic peptide; regulates oxidative stress, mitochondrial stability, survival pathways | |
| Membrane-associated antimicrobial peptide | Human bone marrow MSCs | LL-37 / hCAP-18 (LLGDFFRKSKEK…) | Direct antimicrobial activity; modulates immune cell recruitment; supports wound defense; partially mediates MSC antimicrobial effects | |
| Membrane-associated cytoskeletal peptide | Human adipose MSC secretome | Thymosin β4 (actin-binding LKKTET motif) | Enhances migration, cytoskeletal remodeling, angiogenesis, wound repair; abundant in MSC conditioned medium | |
| Extracellular complement-derived immunoregulatory peptide | Human UC-MSC secretome | Complement C3 fragment peptide (e.g., AEGKGQGTL) | Regulates complement activity, reduces inflammatory signaling, contributes to immunomodulatory MSC secretome effects | |
| Extracellular angiogenic peptides (ECM-derived peptide fragments) | Human MSC secretome (systematic proteomic analysis) | Collagen-derived motifs (e.g., GPP/GVMGFO), thrombospondin fragments | Promote endothelial migration, tube formation, vascular stabilization; reflect high angiogenic potential of MSC secretome | |
| Extracellular broad immunoregulatory peptides (secretome) | Human MSC secretome | Small immunomodulatory ECM fragments; cathelicidin-related short peptides | Suppress pro-inflammatory cytokines, modulate T-cell phenotypes, regulate innate immunity; central to MSC cell-free therapy | |
| Extracellular peptides (xenogenic MSC secretome for clinical therapy) | Xenogeneic MSC secretome used in feline chronic gingivostomatitis | ECM-derived peptides; AMP-like motifs (short cationic sequences) | Promote mucosal healing, reduce oral inflammation, demonstrate cross-species peptide conservation & therapeutic efficacy | |
| Extracellular antimicrobial peptides (xenogenic) | Equine MSC secretome | Cathelicidin/CRAMP-like peptides | Strong antibacterial activity; disrupt microbial membranes; support innate defense mechanisms |
Mechanisms of Action of MSC-Derived Peptides
MSC-DPs execute their regenerative functions through multiple interconnected biological pathways that collectively reproduce many of the therapeutic effects typically attributed to mesenchymal stem cells67,68. A unifying feature of these mechanisms is their paracrine mode of action, whereby peptides—either freely secreted or packaged within extracellular vesicles—bind to receptors on neighboring cells and activate intracellular signaling cascades, such as PI3K/Akt, MAPK/ERK, and JAK/STAT69,70,71. Through these pathways, MSC-DPs promote fibroblast proliferation, endothelial migration, epithelial repair, and cellular survival under stress conditions, thereby establishing a pro-regenerative microenvironment that is independent of MSC engraftment72,73.
In this context, “mimetic” peptides refer primarily to endogenous low-molecular-weight peptides derived from the MSC secretome that exhibit functional similarity to established growth factors or cytokines, rather than to full-length proteins30,31,60. These must be clearly distinguished from synthetic or bioengineered analogs, which replicate similar biological activities but do not strictly fall within the definition of MSC-derived peptides. Specific peptide motifs contribute to these effects; for example, endogenous VEGF-mimetic peptide sequences stimulate endothelial tube formation, while IGF-like domains enhance cell survival under oxidative or inflammatory conditions74,75. Collectively, MSC-DPs exert immunomodulatory, antioxidative, angiogenic, and metabolic regulatory effects76,77, reflecting their role as key molecular mediators that translate the complex paracrine signals of MSCs into concise biochemical cues69.
Immunomodulatory and Anti-Inflammatory Effects
MSC-DPs regulate both innate and adaptive immunity, thereby promoting tissue tolerance and limiting inflammatory damage38,78. Peptides exhibiting IL-10, TGF-β, or PD-L1 mimicry inhibit pro-inflammatory cytokines, such as TNF-α, IL-6, and IFN-γ, while inducing anti-inflammatory mediators79,80,81. In macrophages, these peptides skew polarization towards an M2 pro-healing phenotype, inhibiting nitric oxide production and promoting the phagocytic clearance of cellular debris82,83,84. In lymphocytes, they suppress overactive T-cell proliferation and promote regulatory T-cell induction, effectively inhibiting autoimmune and fibrotic reactions85,86,87. Overall, this immunomodulatory action recapitulates the immune-privileged character of the parental MSCs, accounting for a significant portion of their therapeutic benefit in inflammatory and autoimmune diseases76.
Anti-Oxidative and Anti-Apoptotic Protection
Oxidative stress represents a potent barrier to tissue regeneration88. MSC-DPs modulate redox homeostasis by scavenging free radicals and boosting endogenous antioxidant defenses. Cysteine- and glycine-rich peptides mimic the activity of glutathione precursors and antioxidant enzymes, such as superoxide dismutase (SOD) and catalase38,89. In stressed or ischemic tissues, these peptides inhibit mitochondrial cytochrome C release and activate Bcl-2/Bcl-xL signaling to prevent the apoptosis of resident cells90. This mechanism significantly augments parenchymal and endothelial cell survival during hypoxia or inflammation, facilitating long-term structural repair.
Angiogenesis and Tissue Remodeling
One of the most prominent effects of MSC-DPs is their potent induction of angiogenesis and extracellular matrix (ECM) remodeling91,92. VEGF-, FGF-, or PDGF-motif peptides induce endothelial growth, migration, and capillary sprouting through the activation of VEGFR2 and FGFR193. Concurrently, matrix-modulating peptides regulate MMP-2, MMP-9, and TIMP-1 activity, maintaining a balance between ECM degradation and deposition. These effects facilitate vascular integration, collagen alignment, and improved wound closure14,94. In vivo models have validated that neovascularization is facilitated by MSC-DPs at a level equivalent to recombinant growth factors, but with a significantly reduced immunogenic risk95.
Metabolic Regulation
Emerging data demonstrate that MSC-DPs also contribute to metabolic homeostasis by regulating glucose uptake, lipid metabolism, and mitochondrial biogenesis96,97. Certain peptides enhance insulin sensitivity by activating AMPK and Akt in skeletal muscle and adipocytes98,99, whereas others facilitate lipid oxidation by regulating PGC-1α signaling100. Within hepatic or pancreatic tissues, MSC-DPs counteract oxidative damage and promote β-cell survival, offering significant therapeutic potential in models of metabolic syndrome and diabetes96,101. The coordinated regulation of the inflammatory, oxidative, and metabolic axes supports the systemic reparative potential of the MSC peptidome97,102.
In summary, MSC-DPs exert their effects through five interconnected mechanisms—paracrine signaling, immunomodulation, antioxidative protection, angiogenesis and tissue remodeling, and metabolic regulation—each linked by continuous molecular crosstalk (Figure 3). These mechanisms converge through the activation of key signaling cascades, such as PI3K/Akt, JAK/STAT, and AMPK, collectively enhancing cellular survival, immune homeostasis, redox balance, and vascular and metabolic restoration. Together, these pathways illustrate how MSC-DPs recapitulate the multifunctional therapeutic actions of their parent MSCs through selective and targeted molecular interactions. By orchestrating paracrine communication, immune regulation, redox control, angiogenic remodeling, and metabolic reprogramming, MSC-DPs represent a concentrated, controllable, and next-generation cell-free alternative to traditional MSC-based therapy. Importantly, these mechanistic insights are strongly supported by extensive experimental and preclinical evidence, underscoring the translational relevance and emerging therapeutic value of MSC-DPs.
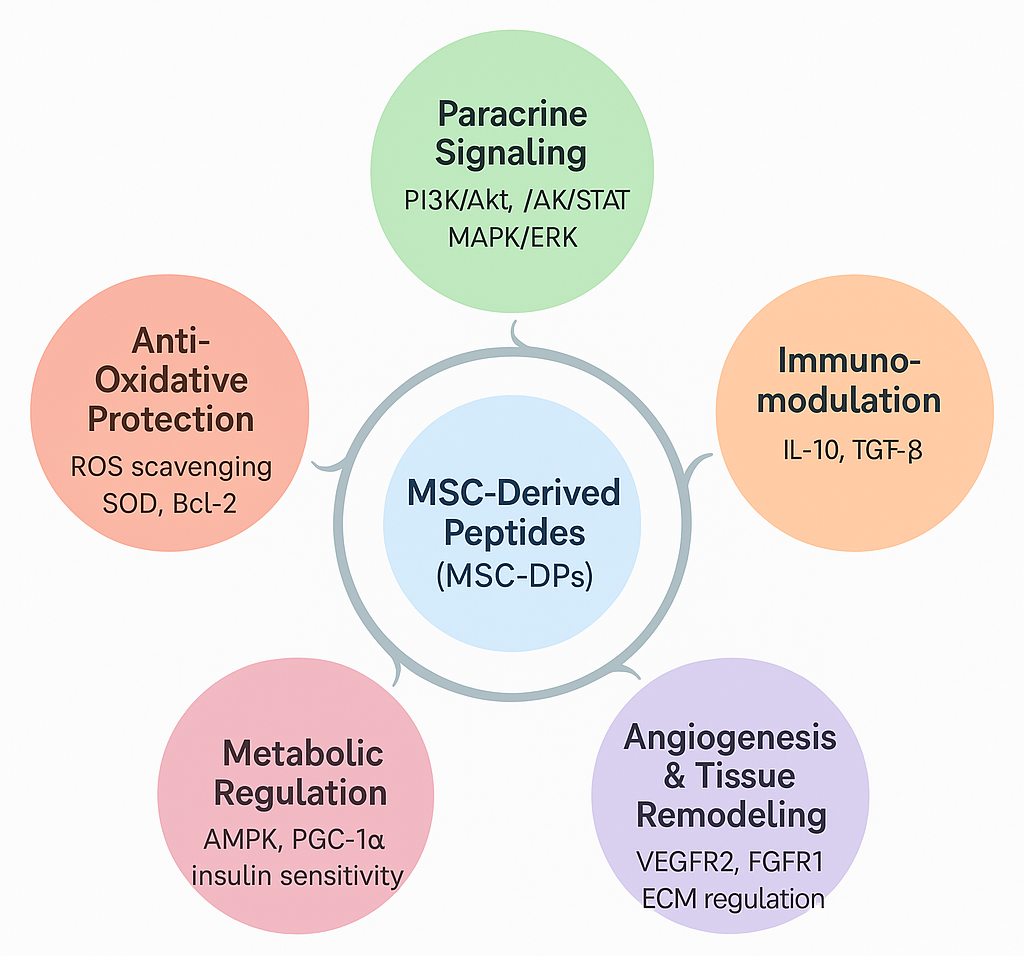
Molecular mechanisms of action of MSC-derived peptides (MSC-DPs) in regenerative medicine. MSC-DPs orchestrate tissue repair and cellular homeostasis through five interconnected biological axes. By activating key paracrine signaling cascades (e.g., PI3K/Akt, JAK/STAT, and MAPK/ERK), these peptides simultaneously drive immunomodulation, stimulate angiogenesis and extracellular matrix (ECM) remodeling, regulate cellular metabolism, and confer potent antioxidative and anti-apoptotic protection.
Experimental and Clinical Evidence
The biological relevance of MSC-DPs is supported by a growing body of in vitro, in vivo, and preclinical evidence demonstrating their regenerative, immunomodulatory, and cytoprotective properties71. Cumulatively, these studies indicate that MSC-DPs replicate the majority of the therapeutic functions of intact MSCs, but with improved safety, reproducibility, and scalability93.
Notably, a significant proportion of current experimental and preclinical evidence is derived from studies employing the whole MSC secretome, conditioned media, or extracellular vesicle-associated formulations rather than purified MSC-derived peptides (MSC-DPs). Given the compositional complexity of the secretome, the therapeutic effects observed in these studies cannot be attributed solely to peptides, but are more likely the result of synergistic interactions among multiple bioactive components. Accordingly, such findings should be interpreted with caution when considered as direct evidence of MSC-DP efficacy.
In Vitro Experimental Studies
Early peptidomic and proteomic analyses have identified numerous low-molecular-weight peptides in MSC supernatants, confirming active secretion beyond the canonical protein factors103. In vitro analyses have demonstrated that VEGF-like and FGF-mimetic peptides from MSCs evoke significant endothelial cell growth and tube formation, indicating robust angiogenic activity104,105.
Furthermore, IL-10-analog peptides inhibit NF-κB activation and decrease pro-inflammatory cytokine production in LPS-activated macrophages, whereas TGF-β-mimetic fragments stimulate fibroblast migration and extracellular matrix synthesis, thereby promoting wound closure in in vitro scratch-assay models103. In addition, cysteine- and proline-rich antioxidant peptides have been shown to antagonize reactive oxygen species (ROS) accumulation and upregulate endogenous antioxidant genes, such as SOD1 and GPX4, during oxidative stress106. Cumulatively, these in vitro data confirm that MSC-DPs operate via paracrine molecular networks, mimicking much of the protective and trophic effects that were previously ascribed to the parent cells.
In Vivo and Preclinical Animal Models
The therapeutic potential of MSC-derived peptide-containing formulations has been investigated across a range of preclinical disease models, including wound healing, cardiovascular repair, neuroregeneration, and metabolic disorders. In rodent models of cutaneous injury, the administration of MSC-derived peptide-enriched or secretome-based preparations has been associated with enhanced epithelial regeneration, increased collagen deposition, and improved neovascularization. These effects partially recapitulate the regenerative outcomes observed with whole-cell MSC therapy, while potentially reducing risks, such as ectopic tissue formation107.
In rodent models of myocardial infarction, treatment with an MSC-DP cocktail reinstated cardiac contractility, reduced infarct size, and suppressed post-ischemic inflammation by activating the PI3K/Akt and STAT3 signaling pathways108. Similarly, in rat models of spinal cord injury and stroke, MSC-secreted neurotrophic peptides improved neurite outgrowth and neuronal survival, which correlated with functional recovery109.
Studies utilizing xenogenic sources of peptides have also supported translational safety. For instance, MSC-DPs from ovine and porcine sources exhibited conserved bioactivity across species, reducing inflammation and improving tissue remodeling in rabbit and canine wound models110. This therapeutic consistency across species supports the hypothesis that many MSC-DPs mediate their effects through evolutionarily conserved molecular targets.
Clinical and Translational Investigations
Most clinical and translational studies are based on the MSC secretome or conditioned media rather than purified MSC-DPs. Given the complexity of these formulations, the observed therapeutic effects cannot be attributed solely to peptides, but instead reflect the combined action of multiple bioactive components. While clinical investigations involving purified MSC-DPs remain limited, early exploratory studies using secretome- and peptide-enriched preparations have been reported in both human and veterinary contexts111.
The topical application of MSC-conditioned media, which contains peptides alongside proteins, cytokines, and extracellular vesicles, has been associated with improved epithelialization and reduced inflammatory cytokine levels in patients with chronic non-healing ulcers; however, these observations are derived from small, uncontrolled studies and require further validation112.
Similarly, in a pilot study of osteochondral defects, the intra-articular administration of MSC-derived peptide fractions was linked to increased cartilage thickness and reduced pain over a six-month follow-up period; although, the sample size was small and the study lacked randomization113.
In veterinary settings, xenogeneic MSC secretome and peptide formulations have demonstrated symptomatic improvement in canine osteoarthritis and feline chronic oral inflammatory disease, providing preliminary proof-of-concept for cross-species feasibility rather than definitive evidence of clinical efficacy114. Taken together, these early findings suggest the potential regenerative activity of MSC-related secretome and peptide formulations, but they do not yet confirm a clinically meaningful therapeutic benefit, and further controlled studies are required.
Key in vitro, preclinical, and translational investigations supporting the regenerative and cross-species efficacy of MSC-DPs are summarized in Table 2. Notably, well-controlled clinical trials exclusively evaluating purified or synthetic MSC-derived peptides remain extremely limited. This represents a critical gap in the field, as the specific therapeutic contribution of peptides, independent of other secretome components, has yet to be rigorously established in clinical settings.
Evidence landscape for MSC-DPs and xenogenic sources (
| MSC source & product | Model / Indication | Dose / Route | Key readouts (summary) | Reference |
|---|---|---|---|---|
| Equine MSCs; secreted antimicrobial peptides | In-vitro antibacterial activity vs. common wound pathogens | Not applicable (secreted factors tested in vitro) | MSC-secreted peptides inhibited growth of multiple skin-wound bacteria; supports bioactive peptide modality from non-human MSCs | |
| Xenogeneic equine MSCs | Homing to naturally occurring canine joint lesion (post-IV) | IV, scintigraphy tracking | First confirmation of xenogeneic MSC homing to lesion in vivo; foundational for cross-species secretome/peptide relevance | |
| Xenogeneic MSCs in dogs with OA | Preclinical canine OA (surgically induced) | IV, low-dose regimen | Analgesic, anti-inflammatory, cartilage-protective effects over 42 days; supports cross-species immunomodulation | |
| Xenogeneic MSC secretome | Veterinary clinical: feline chronic gingivostomatitis (case series, n=8) | Intramucosal injections, 2–3 sessions / 3-month intervals | Clinical improvement in refractory FCGS; first use of xenogeneic MSC secretome in cats | |
| Ovine (sheep) UC-MSCs; immortalized line (TERT) | Source enablement for scalable peptide/secretome research | Not applicable | Established TERT-immortalized ovine UC-MSCs maintaining stemness; practical platform for xenogenic peptide production |
Emerging Trends and Challenges
Current studies focus on the discovery of specific peptide sequences responsible for certain therapeutic effects. Quantitative proteomics, high-throughput peptidomics, and bioinformatic analyses of motifs are facilitating the identification of the most significant regenerative peptides and their target molecules117,118. Nevertheless, several challenges remain:
-
The requirement for standardized purification and isolation techniques119.
-
The optimization of peptide stability and delivery vehicles38.
-
The establishment of regulatory definitions to differentiate between natural peptides and synthetic analogues120.
Well-defined peptide cocktails or biomaterial delivery systems, to be used in subsequent preclinical and future clinical trials, will be required to determine long-term safety, dosage parameters, and therapeutic persistence103.
Cumulative experimental and clinical evidence firmly supports the concept that MSC-DPs are potent, multifunctional mediators capable of driving tissue regeneration through defined molecular mechanisms. Their successful transition from cell-based to cell-free biologic therapy positions them as a promising next-generation platform for regenerative medicine and precision therapeutics. The translational progression of MSC-DP research—from cell-culture validation to animal models and early human and veterinary trials—is summarized in Figure 4. Beyond their independent effects, MSC-DPs demonstrate enhanced therapeutic performance when combined with organ-specific peptides, as outlined in the next section.
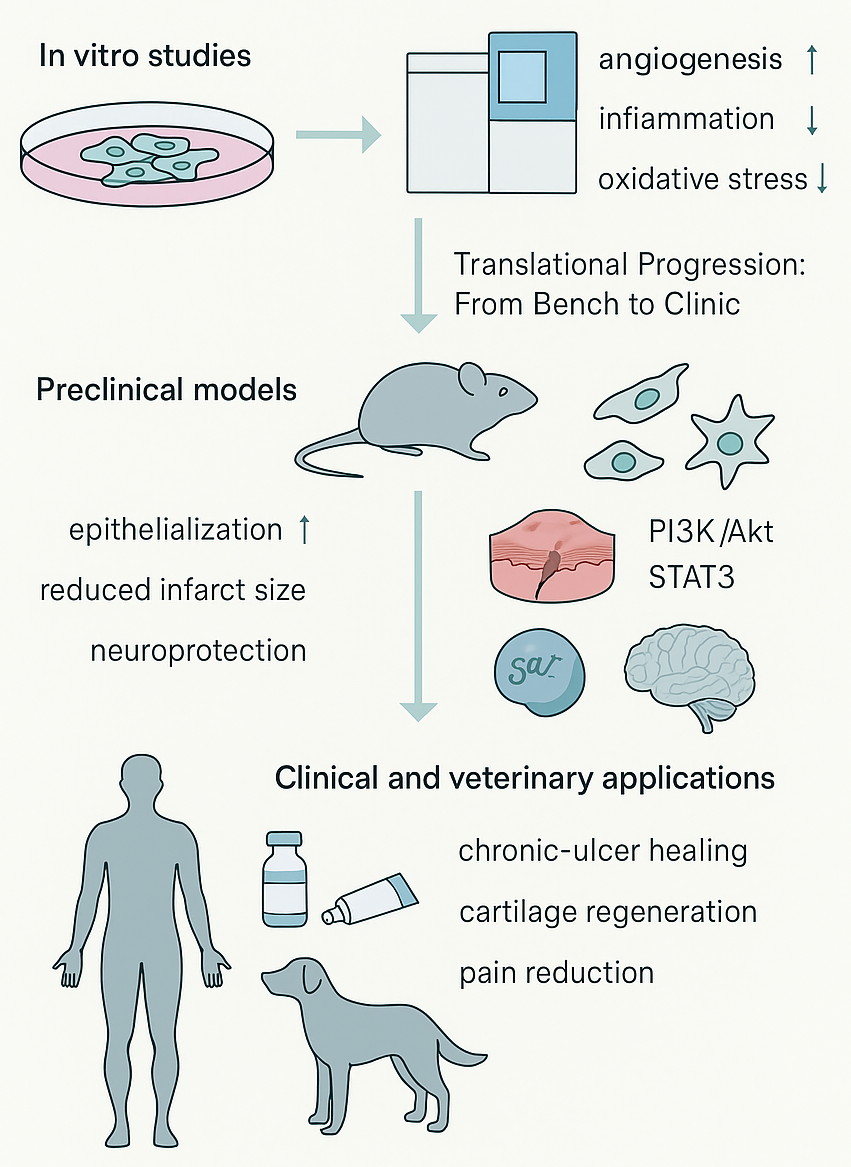
Translational progression of MSC-derived peptides (MSC-DPs) from bench to clinic. The therapeutic efficacy of MSC-DPs is supported by a growing body of multi-tiered evidence.
Synergistic Effects with Organ-Specific Peptides
The therapeutic efficacy of MSC-DPs can be significantly enhanced by coupling them with organ-specific or tissue-targeting peptides that direct their biological functions to precise cellular niches121. This synergy exploits the pleiotropic signaling of MSC-DP-mediated angiogenic, immunomodulatory, and antioxidative functions, coupled with the specificity and selectivity of organ-specific sequences, thereby creating multifunctional therapeutic platforms with immense potential for tissue-specific repair and systemic regulation122.
Molecular Synergy and Mechanistic Interplay
MSC-DPs, whether secreted freely or enclosed within extracellular vesicles (EVs), function as global regulators, controlling immune modulation, angiogenesis, and matrix remodeling123. The cocktail administration or fusion of MSC-DPs with peptides that display organ or receptor specificity—such as arginyl-glycyl-aspartic acid (RGD) peptides that bind selectively to integrins on endothelial cells124, C-peptide variants for insulin signaling125, or N-cadherin-binding fragments that stimulate cardiomyocytes126—translates into a cumulative and highly targeted therapeutic index. Both approaches further potentiate signaling pathways, such as the PI3K/Akt, ERK 1/2, and TGF-β/Smad cascades, ultimately enhancing cellular repair mechanisms127.
Synthetically or naturally designed cell-specific peptides coupled with MSC-DP fractions have demonstrated substantial success in enhancing homing efficiency while minimizing off-target effects. For example, the fusion of angiogenic MSC-DPs with cardiac-targeting peptides resulted in a significant increase in neovascularization within ischemic cardiac tissue as compared to non-targeted control groups121. Additionally, the fusion of a liver-targeting peptide with hepatoprotective MSC-DPs facilitated preferential accumulation within liver tissue, improving its antioxidant state and prompting a concurrent decline in profibrotic factors128.
Synergistic Regeneration in Tissue-Specific Systems
In dermal and epithelial models, the co-delivery of MSC-DP-containing MSC secretome/EVs with binding peptides or laminin-derived peptides promoted cell migration, cell proliferation, and the production of ECM, which resulted in faster wound healing with reduced scarring129. For neuroregeneration, MSC-DPs with a high concentration of neuroregulatory motifs were shown to work in combination with brain-targeting peptides, like the rabies virus glycoprotein (RVG) or SynB peptide (SynB), to enable efficient blood-brain barrier (BBB) passage, as well as to promote axonal extension, neuronal differentiation, and synapse restoration in animal models of stroke or spinal cord injury (SCI)130. In musculoskeletal and osteochondral tissue repair, the combination of chondrogenic MSC-DPs with bone morphogenetic protein-2 (BMP-2) or cartilage oligomeric matrix protein (COMP) mimetic peptides promoted the production of extracellular matrix materials as well as cartilage structural integrity, indicating a potentially viable treatment for osteoarthritis or cartilaginous defects131.
Translational and Biomedical Integration
In translational contexts, these synergies offer a next-generation therapeutic paradigm: a customizable, cell-free, multi-peptide platform that is adaptable to diverse clinical needs14. By uniting the systemic regulatory functions of MSC-DPs with the spatial precision of tissue-specific motifs, optimal regenerative outcomes can be achieved with enhanced efficiency, reproducibility, and safety121.
Beyond simple co-delivery, it is highly feasible to create synergistic constructs by coordinating MSC-DPs from either the soluble secretome or EVs with organ-specific peptides132. Hydrogels containing MSC-DPs, either in free form or associated with EVs, possess high bioactivity, facilitating controlled release as well as prolonged retention at the injury site133. Multifactor-loaded constructs offer spatiotemporal control; for example, an initial immune modulation phase mediated by MSC-DPs can be succeeded by differentiation or vascularization driven by organ-specific peptides134.
In a translational context, such synergies represent a new generation of a therapeutic paradigm featuring a customizable, cell-free, multi-peptide platform that may be adapted for a variety of clinical needs135. Consequently, it will now be possible to optimize regenerative results with increased efficiency, predictability, and safety by integrating the systemic regulatory role of MSC-DPs with tissue-specific motifs. Ultimately, these synergistic interactions contribute directly to the several advantages of MSC-DPs over traditional cell-based therapies.
Advantages of (MSC-DPs)-based over Cell-Based Therapy
MSC-DPs, whether released in a soluble form or packaged within the secretome/EVs, represent a paradigm shift in regenerative medicine, designed to overcome the primary limitations associated with whole-cell transplantation38. Unlike living MSCs, which are encumbered by hurdles such as batch variability, low engraftment efficiency, senescence, and potential tumorigenesis, MSC-DPs offer distinct benefits; these include reduced immunogenicity, increased consistency, and the capacity for large-scale production under standardized conditions136.
Given that their active components are predominantly peptides, these therapeutics demonstrate efficient diffusion across extracellular matrices, facilitating rapid local pharmacodynamic effects at the site of injury137. Furthermore, as non-cellular bioproducts, peptide-rich MSC-DPs exhibit superior storage and distribution properties; they can be safely sterilized, lyophilized, and stored for extended periods with a highly stable shelf life under controlled conditions138. Consequently, these cell-free, peptide-rich formulations mitigate logistical concerns related to cell harvesting, expansion, and cryopreservation, ultimately rendering this form of regenerative medicine safer and more universally applicable139.
Beyond manufacturing advantages, MSC-DPs enable the precise regulation of regenerative mechanisms. Specifically, these bioactive components can selectively modulate angiogenesis, immune responses, oxidative balance, and metabolic pathways, thereby yielding more predictable and controllable outcomes compared to heterogeneous cell populations94. Pharmacologically, the structured and quantifiable composition of peptide-enriched secretome preparations facilitates standardized pharmacokinetic and pharmacodynamic assessments, thereby ensuring a more consistent dose–response relationship across batches140.
From an application standpoint, peptide-enriched cell-free formulations can be seamlessly integrated with biomaterial scaffolds, nanoscale delivery systems, or combined with small-molecule therapeutics, significantly enhancing their therapeutic versatility while maintaining high target specificity141.
In summary, MSC-DPs and peptide-enriched secretomes represent a straightforward, scalable, and ethically favorable platform that bridges the gap between complex cell-based therapies and conventional protein drugs142. By combining the biological sophistication of multicellular communication with the precision of molecular therapeutics, they truly embody the next generation of regenerative medicine.
Limitations, Challenges, and Future Perspectives
Despite their therapeutic promise, MSC-DPs—whether free or secretome/EV-associated—still face several scientific and translational barriers hindering their widespread application in a clinical context143. Currently, the most significant challenge complicating MSC-DP development is inherent peptide variability, driven by differences in starting cell characteristics, tissue and species origin, and cell culture conditions; this variability poses a major hurdle for reproducibility and complicates comparative analyses across studies. Furthermore, standardized preparation and purification techniques for MSC-DPs remain underdeveloped; different cell culture, processing, and analytical methodologies—including proteolysis, chromatography, and mass spectrometry quantification—yield highly variable peptide signatures, thereby impeding the development of MSC-DPs in compliance with FDA Good Manufacturing Practice (GMP) standards144.
Additionally, from a pharmacological perspective, rapid enzymatic turnover and renal clearance compromise the in vivo half-life of these peptides, requiring advanced formulations—such as nanoparticle encapsulation, hydrogel embedding, or PEGylation—to extend peptide availability145. Another major concern for the development of MSC-DPs is immunogenicity; although less immunogenic than whole-cell-derived products, the potential for immune reactions remains significant for xenogeneic-derived peptides, necessitating comprehensive immune toxicity studies to ensure safety prior to clinical application146. Moreover, although xenogeneic peptides are generally less immunogenic than full-length proteins, cross-species administration may still induce immune responses, particularly following repeated exposure. Regulatory agencies, such as the U.S. Food and Drug Administration and the European Medicines Agency, require the rigorous evaluation of immunogenicity, safety, and long-term effects, while challenges related to batch consistency and source traceability further complicate clinical translation147,148. Furthermore, ambiguous regulatory definitions distinguishing natural MSC-DPs from synthetic peptides may complicate intellectual property protection, requiring harmonized global frameworks for widespread therapeutic application149.
An additional key limitation is the frequent extrapolation of therapeutic outcomes from whole secretome or conditioned media studies directly to MSC-derived peptides. Given the compositional complexity of the secretome, the precise therapeutic contribution of low-molecular-weight peptides remains insufficiently resolved. Future studies must prioritize the isolation, characterization, and functional validation of defined peptide sequences to establish clear structure–function relationships and enable the accurate assessment of their independent therapeutic potential150.
Overcoming these obstacles will require the interdisciplinary integration of bioengineering, computational biology, and translational medicine to improve the development pipeline for MSC-DPs151. Future directions will rely on the development of standardized platforms for peptide identification, ensuring reproducible peptide signatures across cellular origins, in conjunction with advanced, bioinformatics-driven models for predicting peptide sequences with potential biological functions152. In this respect, the integration of synthetic biology and peptide engineering may provide insights for designing novel, "hybrid" peptides with improved stability, targeted delivery, and precisely controlled potency153. Simultaneously, the further development of biomaterial-based delivery formulations, including "intelligent" hydrogels or exosome-mimetic vesicles, could provide a robust paradigm for overcoming pharmacokinetic hurdles and improving in vivo dwell time154,155,156.
Ultimately, a concerted effort between bench researchers, practicing clinicians, and regulatory experts is imperative to successfully translate emerging MSC-derived peptides into standardized, approved biologics with predictable safety, efficacy, and potency across various regenerative therapies (cf. Figure 5).
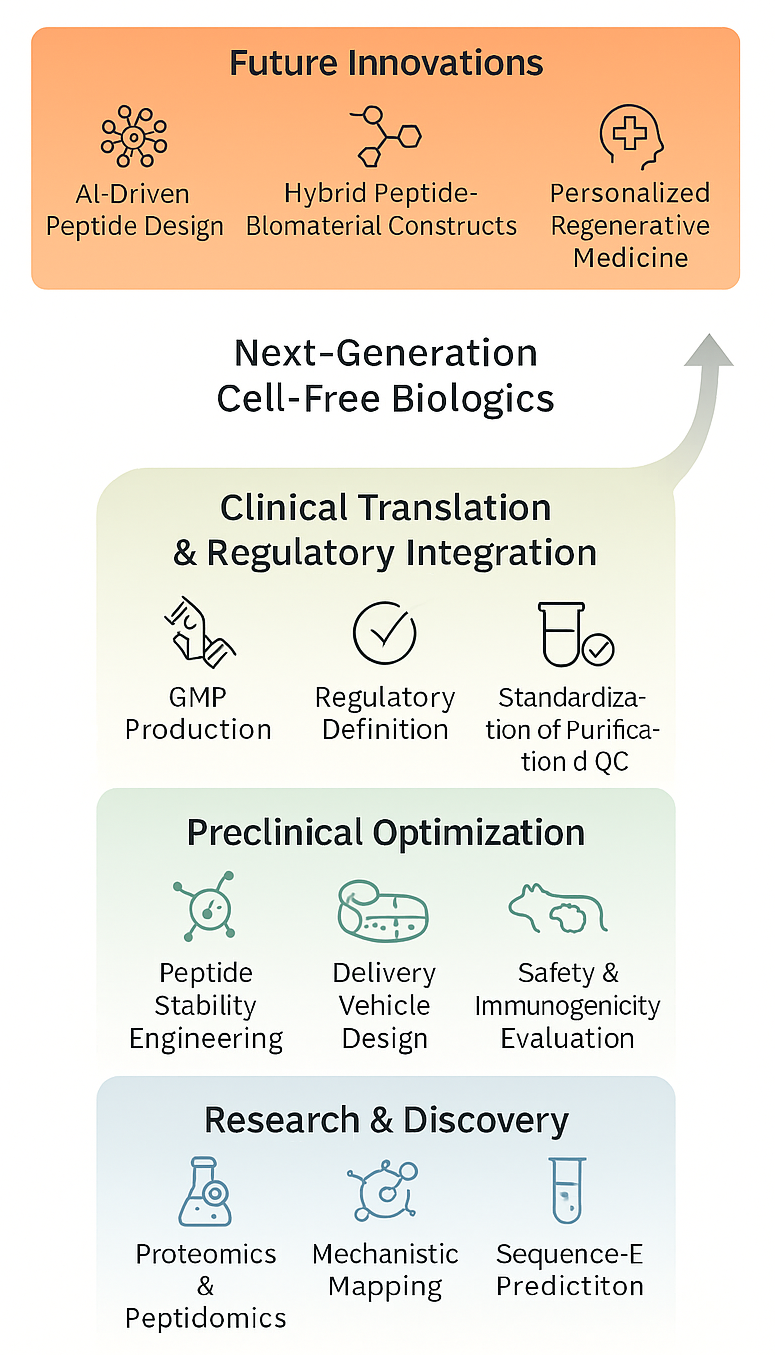
Translational outlook and future directions for MSC-DP therapeutics. The progression of mesenchymal stem cell-derived peptides (MSC-DPs) into next-generation cell-free biologics requires a structured, interdisciplinary pipeline. The process begins with foundational research and discovery utilizing high-resolution proteomics and bioinformatics-driven sequence prediction. Preclinical optimization must address current challenges by focusing on peptide stability engineering, comprehensive immunogenicity evaluations, and the design of advanced delivery vehicles. Advancing to clinical translation demands harmonized regulatory integration, standardized purification methods, and GMP-compliant production. Ultimately, future innovations will leverage artificial intelligence, hybrid peptide-biomaterial constructs, and personalized medicine to fully realize the therapeutic potential of MSC-DPs.
Conclusion
MSC-DPs are increasingly recognized as specialized mediators of MSC paracrine activity, offering a more defined and potentially scalable alternative to whole-cell therapy. Evidence from peptidomic profiling and early functional studies suggests that these peptides modulate several key regenerative pathways, including immune modulation, oxidative stress regulation, metabolic support, and tissue remodeling. Their small size, ease of synthesis, and potential for standardized formulation position them as highly promising candidates for next-generation, cell-free therapeutics.
Despite these advantages, research into MSC-DPs remains in its formative stages. Current findings are largely derived from heterogeneous experimental models, diverse MSC sources, and inconsistent culture or peptide-isolation protocols. These methodological differences limit direct comparisons across studies and generate uncertainty regarding the reproducibility of the reported peptide functions. Moreover, the mechanisms linking peptide sequences to specific biological effects remain poorly defined, while ongoing challenges related to peptide stability, targeted delivery, and in vivo persistence continue to hinder translational advancement.
In order to advance MSC-DPs toward clinical relevance, future research must prioritize standardized peptide-identification workflows, robust functional validation, and the development of delivery systems that enhance peptide stability and bioavailability. The integration of high-resolution proteomics, computational modeling, and biomaterial-based platforms will be critical in defining consistent peptide signatures and clarifying their mechanistic contributions.
Overall, MSC-DPs represent a promising, yet emerging, domain in regenerative medicine, requiring sustained and rigorous investigation before their therapeutic potential can be fully realized. Crucially, the definitive clinical validation of purified MSC-derived peptides remains lacking, further underscoring the need for rigorously designed, peptide-specific translational studies.
Abbreviations
AMPK: AMP-activated protein kinase; BBB: Blood-brain barrier; BMP-2: Bone morphogenetic protein-2; COMP: Cartilage oligomeric matrix protein; ECM: Extracellular matrix; ERK: Extracellular signal-regulated kinase; EVs: Extracellular vesicles; FDA: Food and Drug Administration; FGF: Fibroblast growth factor; FGFR1: Fibroblast growth factor receptor 1; IFN-γ: Interferon gamma; IL-10: Interleukin-10; JAK/STAT: Janus kinase/Signal transducer and activator of transcription; MAPK: Mitogen-activated protein kinase; MMP: Matrix metalloproteinase; MSC: Mesenchymal stem cell; MSC-DPs: Mesenchymal stem cell-derived peptides; PDGF: Platelet-derived growth factor; PD-L1: Programmed death-ligand 1; PI3K/Akt: Phosphoinositide 3-kinase/Protein kinase B; RGD: Arginyl-Glycyl-Aspartic acid; ROS: Reactive oxygen species; RVG: Rabies virus glycoprotein; SCI: Spinal cord injury; SOD: Superoxide dismutase; TGF-β: Transforming growth factor beta; TIMP: Tissue inhibitor of metalloproteinases; TNF-α: Tumor necrosis factor alpha; VEGF: Vascular endothelial growth factor; VEGFR2: Vascular endothelial growth factor receptor 2
Acknowledgments
The authors would like to thank the OXYZ Health & Wellness Academy for their administrative and technical support during the preparation of this manuscript.
Author’s Contributions
All authors contributed equally to the conception and design of the study. All authors read and approved the final manuscript.
Funding
This research was fully supported by OXYZ Health & Wellness Academy (Research and Development Department, Malaysia) as part of its ongoing scientific initiative on mesenchymal stem cell–based regenerative medicine and peptide integration studies.
Availability of Data and Materials
No new data was generated for this review article. Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Declaration of Generative AI and AI-Assisted Technologies in the Writing Process
The authors declare that they have not used generative AI (a type of artificial intelligence technology that can produce various types of content including text, imagery, audio and synthetic data).
Competing Interests
The authors declare that they have no competing interests.

