Targeting Resistant Staphylococcus aureus with small Extracellular Vesicles from Human Adipose-Derived Stromal Vascular Fraction
- Department of Microbiology, Central Research Laboratory and Nitte University Centre for Stem Cell Research & Regenerative Medicine (NUCSReM), KS Hegde Medical Academy (KSHEMA), Nitte (Deemed to be University), Deralakatte, Mangaluru 575018, India
- Department of Plastic Surgery, KS Hegde Medical Academy (KSHEMA), Nitte(Deemed to be University), Deralakatte, Mangaluru 575018, India
- Nitte University Centre for Stem Cell Research & Regenerative Medicine (NUCSReM), KS Hegde Medical Academy (KSHEMA), Nitte (Deemed to be University), Deralakatte, Mangaluru 575018, India
- Central Research Laboratory, KS Hegde Medical Academy (KSHEMA), Nitte (Deemed to be University), Deralakatte, Mangaluru 575018, India
Abstract
Introduction: Antimicrobial resistance (AMR), particularly involving antibiotic-resistant pathogens such as methicillin-resistant Staphylococcus aureus (MRSA), poses a critical global health challenge. Driven primarily by the mecA gene, MRSA is prevalent in both healthcare and community settings and continues to cause severe infections and high mortality worldwide, despite the availability of antibiotics. The COVID-19 pandemic further highlighted the limitations and adverse effects of conventional synthetic antibiotics and cell-based therapies, emphasizing the urgent need for safer, innovative, and preferably natural, cell- and drug-free therapeutic approaches.
Objective: Given the well-established immunomodulatory properties of human adipose tissue, adipose-derived vesicles were selected as a biologically relevant, economical, non-invasive, and promising therapeutic candidate for this investigation. This study investigated the intrinsic antibacterial and antibiofilm properties of small extracellular vesicles (sEVs) derived from the stromal vascular fraction (SVF) of healthy human adipose tissue (hHAT) against MRSA, thereby addressing a critical research gap in current AMR treatment strategies.
Methods: SVF‑sEVs from hHAT were isolated using the established Priglinger method and characterized in accordance with MISEV 2023 guidelines. Their antibacterial activity was evaluated in vitro against both MRSA and methicillin-sensitive S. aureus (MSSA) strains—comprising American Type Culture Collection (ATCC) reference strains and clinical isolates (CS)—across multiple time points. Plain phosphate-buffered saline (PBS), devoid of antibiotics or antifungals, served as the negative control. Following the confirmation of antibacterial efficacy, the concentration-dependent inhibition of biofilm formation was assessed for MRSA, MSSA, and the rpoA reference strain, given the pivotal role of biofilms in AMR.
Results: Naturally occurring, cell-free, nanoscale neat SVF-sEVs (EVN; 100 µL, containing 2.15 × 10⁹ sEVs/100 µL) exhibited antibacterial activity against both MRSA and MSSA, alongside a concentration-dependent inhibition of biofilm formation, including in the rpoA reference strain. These effects were confirmed quantitatively via colony-forming unit (CFU) counts and qualitatively via zone of inhibition (ZOI) assays, revealing a significant modulation of bacterial viability and biofilm development. The ATCC MSSA strain demonstrated the largest inhibition zones, the CS-MRSA strain exhibited the greatest reduction in colony counts, and the ATCC MRSA strain showed significant antibiofilm effects.
Conclusions: SVF‑sEVs constitute a novel, biocompatible, and ethically sound therapeutic strategy against MRSA. They offer a promising adjunct or alternative to conventional antibiotics, with a potentially lower risk of driving antimicrobial resistance.
Introduction
The future of global health depends on the ability to manage and mitigate a diverse array of microorganisms, particularly antibiotic-resistant microorganisms (ARMs). These pathogens pose significant health threats by evading standard antimicrobial therapies, thereby complicating clinical management and causing widespread infections across diverse populations. Rapid population growth, limited healthcare access, and the misuse of antibiotics further exacerbate these challenges, compromising both public health systems and global economies.1
Staphylococcus aureus (S. aureus) is a Gram-positive bacterium that commonly colonizes human skin and nasal epithelia; however, it is also an opportunistic pathogen capable of causing severe pneumonia, sepsis, and mortality. The bacterium acquires resistance to beta-lactam and other broad-spectrum antibiotics primarily through the mecA gene, evolving into a multidrug-resistant pathogen. The most clinically significant variant, methicillin-resistant S. aureus (MRSA), is increasingly prevalent in both healthcare and community settings and is transmitted primarily via direct contact with infected individuals. Despite the availability of potent antimicrobial agents, MRSA remains a leading cause of infection-related mortality worldwide.2 In response to this escalating crisis, the World Health Organization (WHO) has projected that, without immediate intervention, antimicrobial resistance (AMR) could result in approximately 389,000 deaths annually in South Asia and an estimated 10 million deaths globally by 2050.3,4
The COVID-19 pandemic has further underscored the critical need for safer, innovative strategies to combat ARMs and safeguard public health. It has demonstrated that relying solely on synthetic antibiotics is insufficient, as these agents can induce adverse effects, long-term complications, and severe toxicities. Consequently, there is an urgent demand for novel, preferably natural, therapeutics—administered either as monotherapy or as an adjuvant to conventional antibiotics—capable of eradicating MRSA without compromising host immune function.
Extracellular vesicles (EVs) have emerged as promising therapeutic modalities. These nanosized, lipid membrane-bound particles—which include subtypes such as exosomes, microvesicles, and ectosomes—are secreted by virtually all cell types and encapsulate bioactive cargo, including proteins, nucleic acids (RNAs), and lipids. Within the context of infection biology, EVs replicate aspects of their parent cells' physiological roles by transferring microbial signals, serving as intercellular molecular messengers, and modulating host immune responses. Contingent upon their cellular origin and the intrinsic antimicrobial defenses of the target cells, EVs can either facilitate or impede pathogen survival. Among the various EV subtypes, small extracellular vesicles (sEVs)—typically ranging from 50 to 200 nm in diameter—and particularly those derived from primary tissue sources (ti-sEVs), have garnered significant interest due to their structural stability and robust therapeutic potential.5,6,7
Healthy human adipose tissue (hHAT) is now recognized as a dynamic immunometabolic organ rather than an inert lipid reservoir. Its stromal vascular fraction (SVF) comprises a heterogeneous population of endothelial, immune, and progenitor cells that collectively reflect the tissue's physiological profile. Small extracellular vesicles (sEVs) secreted by these cells mediate immunomodulatory communication throughout the body.8,9,10,11,12 Therefore, we hypothesized that by harnessing these properties, the intrinsic inhibitory effects of SVF-derived sEVs (SVF-sEVs) could improve clinical outcomes against MRSA, thereby addressing a major gap in current research. To the best of our knowledge, this hypothesis had not been previously investigated.
The significance of this study lies in the utilization of SVF-sEVs, which are naturally occurring, highly biocompatible, nanoscale, and entirely cell-free. They offer a novel strategy to combat MRSA, one of the most persistent bacterial pathogens globally, while circumventing the ethical and regulatory concerns associated with stem cell therapies. Although previous studies have reported the antibacterial effects of mesenchymal cell-derived EVs, our demonstration of their efficacy against MRSA biofilms using adipose tissue-derived SVF represents a novel contribution to the field. Furthermore, unlike many synthetic pharmacological agents, SVF-sEVs possess a lower propensity for inducing antimicrobial resistance and exhibit an enhanced safety profile compared to cell-based therapies, positioning them as highly promising therapeutic agents.
For this investigation, SVF-sEVs were isolated from hHAT utilizing the established Priglinger method and characterized in accordance with the MISEV2023 guidelines.13,14 Subsequently, the SVF-sEVs were evaluated in vitro for their antibacterial efficacy against MRSA and methicillin-sensitive S. aureus (MSSA) strains—sourced from both reference collections and clinical isolates—across multiple time points. Following the confirmation of their antibacterial activity, the concentration-dependent inhibitory effects of SVF-sEVs were assessed on MRSA and MSSA biofilms, as well as on the rpoA reference strain. This assessment was critical, given that antimicrobial resistance is intrinsically associated with the production of extracellular polymeric substances (EPS), a primary structural component of biofilms.15
Methods
Study Setting
All experiments were conducted at the Central Research Laboratory of a tertiary-care hospital affiliated with Nitte (Deemed to be University) in Mangalore, Karnataka, India. The facility is fully accredited in accordance with National Accreditation Board for Testing and Calibration Laboratories (NABL) and International Organization for Standardization (ISO) guidelines.
Ethical Considerations
The study was conducted in compliance with the Declaration of Helsinki (1964, as revised in 2013) and received approval from the Institutional Central Ethics Committee (Ref: NU/CEC/2021/230, dated January 13, 2022). The committee reviewed and approved the use of 150 mL of hHAT, ensuring the strict protection of donor rights and the appropriate handling of all biological specimens.
Type of Sampling
A purposive sampling strategy was employed to ensure the consistent quality and biological relevance of the source material for sEV isolation. A cohort of six donors was selected, based on established EV literature indicating that five or more biological replicates are sufficient to achieve reproducible vesicle yields and ensure biological consistency.
Eligibility Criteria
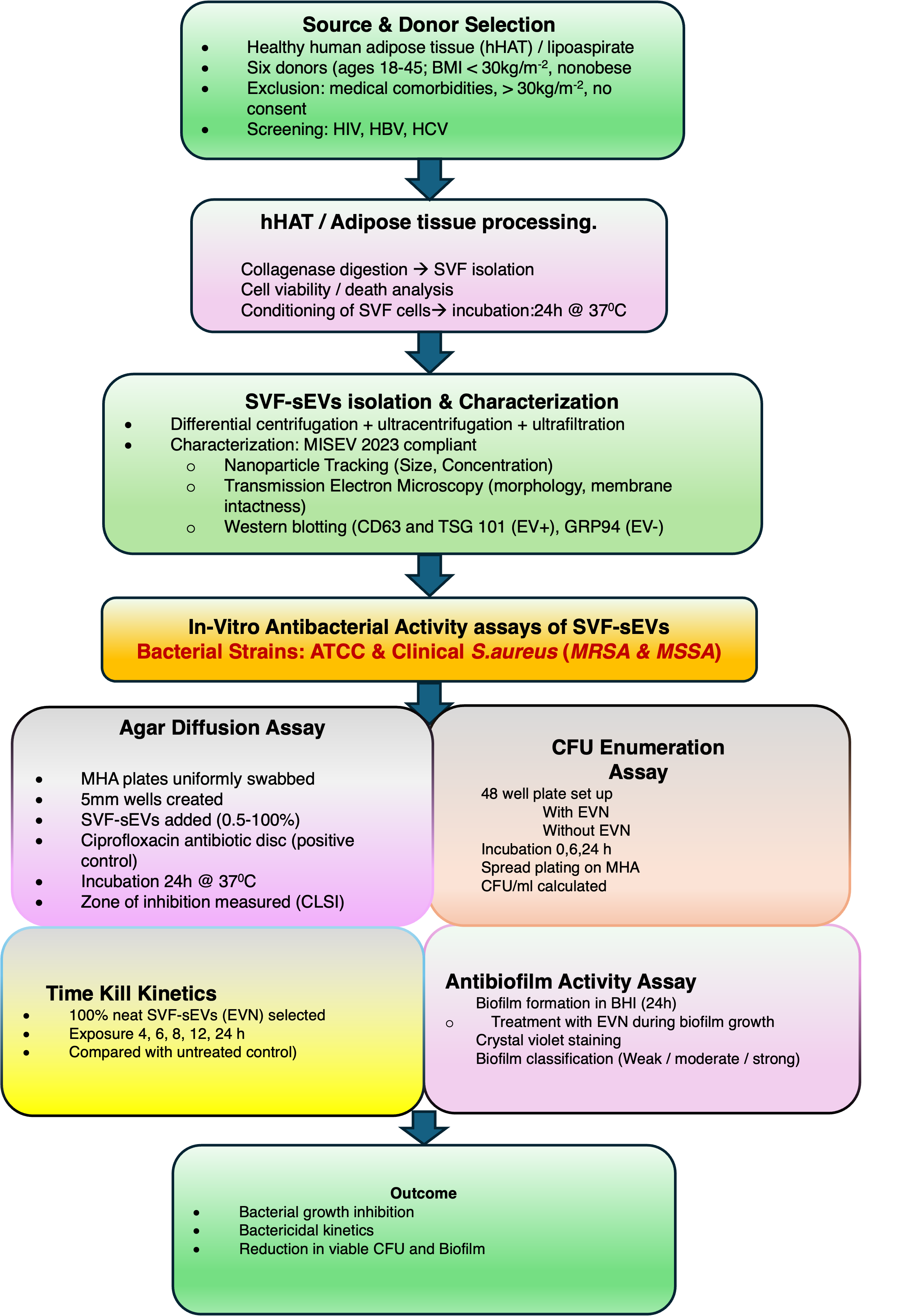
Adipose tissue samples were procured from healthy, non-obese adults (body mass index [BMI] < 30 kg/m) aged 18–45 years who were undergoing elective liposuction procedures. Individuals falling outside this age range, presenting with significant medical comorbidities, or possessing a BMI ≥ 30 kg/m were excluded from the study. All donors underwent standard screening to confirm the absence of infectious diseases, including HIV, hepatitis B, and hepatitis C. SVF from healthy donors was specifically utilized to minimize variability arising from metabolic or infectious conditions and to yield physiologically normal, immunomodulatory vesicles.
Research Participant Consent
Written informed consent was obtained from all participants prior to enrollment. Confidentiality was strictly maintained by anonymizing samples utilizing coded identifiers, and participants retained the right to withdraw their consent at any time without penalty. Tissue collection was performed by trained surgical personnel during routine clinical procedures to mitigate any associated risks and minimize patient discomfort.
Study Design and Reporting
This original, in vitro, observational study was designed to investigate the innate antibacterial activity of SVF-sEVs against MRSA.
hHAT-SVF-sEV Preparation Protocol and Antibacterial Property Data Collection
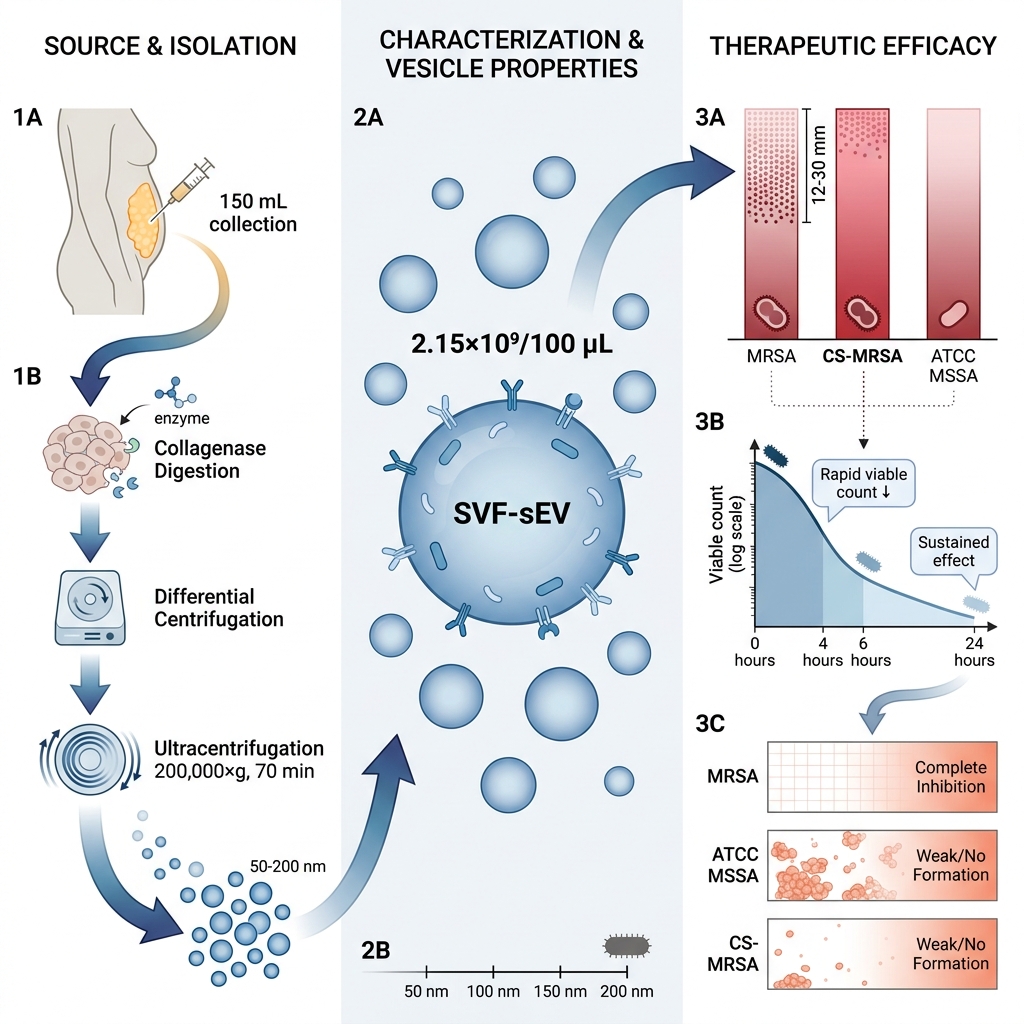
All samples were processed within 2 hours of collection to preserve vesicle integrity and prevent the degradation of bioactive molecules. All experiments were outcome-driven and performed under controlled in vitro laboratory conditions, without randomization or the involvement of live human or animal subjects. Isolated SVF-sEVs were characterized for vesicle morphology, protein profiles, and antibacterial properties utilizing nanoparticle tracking analysis (NTA), transmission electron microscopy (TEM), and optical density measurements at 600 nm (OD). This purposive design facilitated the evaluation of 100% neat (undiluted and intact) SVF-sEVs against MRSA under standardized conditions, as delineated in Figure 1.
Schematic overview demonstrating that host-derived small extracellular vesicles (sEVs) from the stromal vascular fraction (SVF) of healthy human adipose tissue (hHAT) exhibit antibacterial effects against antibiotic-resistant
The study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines to ensure methodological transparency, comprehensive reporting, and reproducibility. Detailed information regarding sample procurement, vesicle isolation, quality control procedures, data validation, and the complete STROBE checklist is provided in the Supplementary File.
hHAT Sample Collection
A total volume of 150 mL of residual hHAT—also referred to as subcutaneous adipose tissue or lipoaspirate, and typically considered biological waste—was collected from the 360° collection sites of six consenting research participants. The cohort comprised healthy, non-obese Indian males and females aged 18 to 45 years with a normal body mass index (BMI). The date of sample collection, total volume of lipoaspirate, number of freeze-thaw cycles, and specific storage conditions for downstream analyses were systematically documented.
SVF Isolation
To isolate the SVF, the aliquoted adipose tissue samples were washed with an equal volume of Dulbecco’s phosphate-buffered saline (DPBS) and subsequently centrifuged at 500 × g for 5 minutes at 37 °C. This washing procedure was repeated a minimum of three times until macroscopic blood and debris were removed. Next, an equal volume of 0.1% type II collagenase tissue digestion enzyme (TDE) was added to the washed tissue (up to 20 mL of wet tissue per conical tube), and the suspension was incubated at 37 °C with moderate agitation for 4.5 hours. Following incubation, enzymatic digestion was terminated by the addition of cold PBS, and the mixture was centrifuged at 1500 × g for 5 minutes. Finally, the resulting SVF pellet was resuspended in PBS, filtered through a 100 µm cell strainer, and subjected to a final centrifugation step.
SVF Cell Counting, Viability Assessment, and Conditioning
The isolated SVF pellet was resuspended in 8 mL of plain Dulbecco’s Modified Eagle Medium (DMEM), and initial cell quantification and viability assessments were performed utilizing a trypan blue exclusion assay. For standard cell culture propagation, a routine 1% antibiotic-antimycotic mixture (comprising penicillin and streptomycin) was supplemented. However, to ensure unbiased outcomes and prevent experimental interference, an alternate conditioning protocol entirely devoid of antibiotics and antifungals was strictly utilized for all samples designated for downstream microbiological evaluations.
SVF-sEV Isolation, Cryopreservation, and Thawing
The resulting cell suspension was aliquoted into T-75 tissue culture flasks at a seeding density of approximately 6 × 10 SVF cells per flask. The cultures were incubated at 37 °C in a humidified atmosphere containing 5% CO₂. Once the heterogeneous SVF cell populations reached >60% sub-confluence, the conditioned medium was harvested at 16 and 20 hours. This conditioned medium was then processed via sequential differential centrifugation: 300 × g for 10 minutes to remove cells and large debris; 2,000 × g for 20 minutes to eliminate apoptotic bodies; and 10,000 × g for 30 minutes to pellet larger extracellular vesicles. The resulting supernatant was subjected to ultracentrifugation at 200,000 × g for 70 minutes at 4 °C utilizing a Beckman Coulter Optima ultracentrifuge equipped with a Type 70 Ti rotor to precipitate the SVF-sEVs. The obtained pellets were washed once with PBS and re-ultracentrifuged at 100,000 × g for 70 minutes. To enhance vesicle purity and remove non-EV-associated proteins and lipids, the final SVF-sEV pellets were resuspended in PBS and subjected to ultrafiltration using a 10 kDa Amicon Ultra-15 centrifugal filter unit (Millipore, Merck KGaA, Ireland) at 4,000 × g for 30 minutes at 4 °C. The purified SVF-sEVs were resuspended in an equal volume of PBS and aliquoted into 1.5 mL click-lock microcentrifuge tubes, which were either stored at −80 °C for long-term cryopreservation or maintained at 4 °C for immediate downstream analyses.
SVF-sEV Physicochemical Assessment to Confirm Vesicle Identity
To confirm vesicle identity in strict accordance with the MISEV2023 guidelines, the isolated SVF-sEVs were characterized for size, morphology, and molecular markers utilizing complementary orthogonal techniques. The specific methodologies employed were as follows:
-
Nanoparticle Tracking Analysis (NTA): Particle size distribution and vesicle concentration were assessed utilizing a NanoSight LM10 instrument (Malvern Instruments, Malvern, UK) equipped with a 405 nm laser. The SVF-sEV samples were diluted 1:10,000 in DPBS prior to analysis. Instrument calibration was performed using National Institute of Standards and Technology (NIST)-traceable 200 nm polystyrene beads. The detection threshold and camera level were standardized at 7 and 14, respectively.
-
Transmission Electron Microscopy (TEM): Vesicle morphology and membrane integrity were evaluated using an FEI Tecnai T20 transmission electron microscope operating at 200 kV. The acquired images were subsequently processed utilizing Gatan Microscopy Suite software.
-
Fluorescence Microscopy (FM): Fluorescence microscopy was employed to visualize vesicle ultrastructure and to further assess the integrity of the lipid membrane and the characteristic cup-shaped morphology.
-
Western Blotting (WB): Biochemical validation of the SVF-sEVs was conducted via Western blotting, in accordance with MISEV2023 recommendations. This involved probing for the EV-enriched positive markers CD63 and TSG101, alongside the negative marker GRP94. The absence of GRP94 was utilized to confirm minimal contamination by intracellular components. Samples were concentrated, lysed, sonicated, and centrifuged, after which the total protein content was quantified utilizing a bicinchoninic acid (BCA) assay (Thermo Fisher Scientific, Waltham, MA, USA). Equivalent amounts of protein were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and subsequently detected utilizing horseradish peroxidase (HRP)-conjugated secondary antibodies (1:10,000). β-actin (1:1,000) served as an internal loading control. Primary antibodies directed against CD63, TSG101, and GRP94 were sourced from Invitrogen (Cat# SAB4301607, PA531260, and SAB2101094, respectively), while secondary antibodies were procured from Thermo Fisher Scientific (Cat# G21234 and G21040).16
Yield, Purity, Endotoxin, and Sterility Considerations
SVF-sEV yield was quantified via NTA as particles per frame and normalized to the initial mass of the adipose tissue processed. This normalization facilitated relative comparisons across preparations and demonstrated consistent vesicle recovery, thereby supporting workflow reproducibility and scalability. An Amicon ultrafiltration step was utilized to minimize co-isolated impurities, such as lipoproteins and protein aggregates. The purity of the resulting SVF-sEV preparations was subsequently verified via Western blot analysis. Formal endotoxin quantification utilizing a Limulus amebocyte lysate (LAL) assay was not performed; thus, potential endotoxin contributions cannot be entirely excluded and remain a key limitation of this study. However, all SVF-sEV isolations were performed aseptically utilizing sterile reagents and consumables, and no microbial contamination was observed during processing or functional assays. Sterility was assessed qualitatively by the absence of turbidity, visible microbial growth, or unexpected cytotoxicity in downstream experiments. Future studies incorporating formal endotoxin quantification and rigorous sterility testing will be requisite to support regulatory compliance and clinical translation. The prepared SVF-sEVs were subsequently utilized in triplicate microbiological experiments to assess their capacity to inhibit MRSA and MSSA. All experiments were conducted in compliance with Good Clinical and Laboratory Practice (GCLP) standards utilizing the bacterial strains detailed below.
Bacterial Strains
The antimicrobial activity of the SVF-sEVs was evaluated against Staphylococcus aureus. Reference strains of MRSA (ATCC 43300) and MSSA (ATCC 29213), alongside clinical MRSA and MSSA isolates, were procured from the Central Research Laboratory. Methicillin resistance was genetically confirmed via polymerase chain reaction (PCR) detection of the mecA gene.
SVF-sEV In Vitro Antibacterial Activity Assays
-
Agar Diffusion Assay:
Inoculum Preparation: Three to five isolated colonies were selected from the ATCC reference and clinical strain plates, inoculated into Mueller-Hinton (MH) broth, and incubated for 4 hours at 37 °C. The turbidity of the bacterial suspension was subsequently adjusted to 0.5 McFarland units (approximately 1.5 × 108 CFU/mL).Inoculation of Agar Plates: A sterile cotton swab was immersed in the standardized inoculum and utilized to uniformly inoculate the entire surface of a Mueller-Hinton agar (MHA) plate by swabbing in three intersecting directions. Following inoculation, a sterile, heated 5 mm cork borer was forcefully pressed into the agar to create wells; each plate contained three to six wells. Varying concentrations of SVF-sEVs (0.5%, 1%, 5%, 10%, 25%, 50%, 75%, and 100%) were then dispensed into the respective wells to ensure effective diffusion into theS. aureus lawns. A commercially available single-dose ciprofloxacin disk (5 µg) served as a positive control. The plates were incubated at 37 °C for 24 hours, after which the zones of inhibition were measured in strict accordance with Clinical and Laboratory Standards Institute (CLSI) guidelines.17 -
Time-Kill Assay: Following the confirmation that 100% neat SVF-sEVs (EVN) demonstrated the highest efficacy among the tested concentrations, their bactericidal impact against the
S. aureus strains was evaluated longitudinally from 4 to 24 hours, with the results compared against a blank control. -
Colony-Forming Unit (CFU) Enumeration via Spread Plate Method: This assay was conducted to quantitatively assess the inherent antibacterial efficacy of the purified SVF-sEVs. Assays were performed in 48-well microtiter plates under two experimental conditions: with and without the addition of SVF-sEVs. In the untreated control wells ("without SVF-sEVs"), a mixture of 90 µL of fresh Luria-Bertani (LB) broth and 10 µL of the respective bacterial inoculum was utilized. Conversely, in the treatment wells ("with SVF-sEVs"), 10 µL of EVN was added to 100 µL of the respective inoculated culture broth. The microtiter plates were incubated, and samples were drawn at 0, 6, and 24 hours. At each time point, a 10 µL (0.01 mL) aliquot from each well was spread onto fresh MHA plates using a sterile glass spreader and incubated overnight. The plates were subsequently examined for visible colony inhibition, and viable microbial counts were determined both before and after EVN treatment. The concentration of viable bacteria was calculated using the following formula: CFU/mL = (Number of colonies × Dilution factor) / Volume plated (mL). Given that direct plating was performed without prior dilution, the dilution factor was 1, simplifying the calculation to: Number of colonies × 100.
-
Antibiofilm Activity Assay: The biofilm formation capacity of both ATCC and clinical MRSA and MSSA strains was evaluated utilizing a standard microtiter plate method.18 The
S. aureus strains were inoculated into brain heart infusion (BHI) broth and incubated for 4 hours to achieve a turbidity equivalent to 0.5 McFarland units (1.5 × 108 CFU/mL). For the baseline biofilm formation assay, 180 µL of fresh BHI broth was dispensed into the first four wells of a 96-well microtiter plate, followed by the addition of 20 µL of the standardized inoculum for each strain. A fifth well, containing only plain BHI broth, served as the negative control. Each treatment group was evaluated in triplicate, and the plate was incubated at 37 °C for 24 hours. Post-incubation, the wells were gently rinsed three times with 1× PBS, inverted, and tapped to remove planktonic cells and residual media, thereby minimizing background staining. Adherent biofilms were fixed by adding 200 µL of 95% ethanol, followed by staining with 200 µL of a 1% crystal violet (CV) solution per well. The plate was incubated at room temperature for 15 minutes, washed three times with 1× PBS, and blotted with paper towels to remove excess dye. Finally, 200 µL of 30% glacial acetic acid was added to solubilize the bound CV. After a 15-minute incubation at room temperature, the plate was inverted to dry. Uninoculated broth was utilized as a control to verify sterility and account for the non-specific binding of media components.
Statistical Analysis
Data derived from the agar diffusion assays were analyzed using GraphPad Prism version 9.0.2 (GraphPad Software, San Diego, CA, USA). Data normality was assessed using the Shapiro–Wilk test, and statistical significance was established at p < 0.05, corresponding to a 95% confidence interval. Comparisons between MRSA and MSSA strains treated with SVF-sEVs were conducted utilizing unpaired, two-tailed Student’s t-tests or a one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test, as appropriate. Descriptive statistics were generated utilizing SPSS version 26.0 (IBM Corp., Armonk, NY, USA). Data from the time-kill kinetic assays are presented as the mean ± standard deviation (SD) derived from a minimum of three independent experiments. Statistical differences across time points and between treatment conditions were evaluated utilizing a two-way repeated-measures ANOVA. For the CFU enumeration assays, CFU/mL values were log₁₀-transformed to stabilize variance and subsequently assessed for normality using the Shapiro–Wilk test. In instances where normality assumptions were satisfied, intergroup comparisons were performed using a one-way ANOVA with Tukey’s post hoc test; conversely, non-parametric data were analyzed utilizing the Kruskal–Wallis test followed by Dunn’s post hoc analysis. A post hoc power assessment indicated sufficient statistical power to detect large antimicrobial effect sizes; however, given the exploratory nature of this investigation, the analyses were interpreted as hypothesis-generating. Although multiple SVF-sEV concentrations were evaluated experimentally, only data corresponding to the 100% concentration are presented. No correction for multiple comparisons was applied, as these specific analyses were restricted to MRSA and MSSA strains evaluated at a single concentration.
Baseline biofilm formation and subsequent antibiofilm activity were assessed utilizing a semi-quantitative ordinal scale (categorized as no biofilm, weak, or moderate). For image-based analyses, qualitative scores were converted into ordinal numerical values (0–2) and evaluated using the Kruskal–Wallis test to ascertain overall intergroup differences. Because the data were categorical and the sample sizes per group were relatively small, post hoc pairwise comparisons were omitted, and the results are presented descriptively. These analyses are intended to illustrate comparative biofilm behavior and should be strictly interpreted as exploratory in nature.
Results
Identification of Isolated SVF Cells
Assessing the viability of SVF cells based on membrane integrity is essential for the production of therapeutic-grade SVF-sEVs. Utilizing a trypan blue exclusion assay, we quantified the cell count, viability, and overall cellular composition of the heterogeneous SVF population, confirming it was comparable to its parent tissue source. This analysis demonstrated 80% to 90% cell viability and revealed the presence of a diverse immune cell population, which accounted for approximately 80% of the hHAT cellular composition (Figure 2i, ii).

Viability and cellular composition of the isolated hHAT-SVF. Trypan blue exclusion assays indicate 80–90% cell viability within a diverse cell population representing approximately 80% of the hHAT cellular composition. Abbreviations: hHAT, healthy human adipose tissue; SVF, stromal vascular fraction.
SVF-sEV Phenotypic Characterization
Within the PBS suspension of the prepared SVF-sEVs, dispersed nanoparticles were visualized in the raw images acquired via nanoparticle tracking analysis (NTA) (Figure 3). Transmission electron microscopy (TEM) identified nanoscale vesicles morphologically consistent with SVF-sEVs; intact, spherical structures encapsulating protein cargo were clearly evident (indicated by red arrows, Figure 4i–iii). Furthermore, fluorescence microscopy confirmed the presence of the intact, cup-shaped ultrastructure characteristic of sEVs (Figure 5).

Nanoparticle tracking analysis (NTA) of SVF-sEVs. A raw video frame displays SVF-sEVs in a phosphate-buffered saline (PBS) suspension, illustrating discrete, non-overlapping nanoparticle trajectories without evidence of fusion or aggregation. This pattern indicates intact vesicle membranes and a relatively homogeneous particle population characterized by uniform Brownian motion and the absence of clumping, which is consistent with efficient isolation and good colloidal stability of the sEV preparation. Abbreviations: NTA, nanoparticle tracking analysis; PBS, phosphate-buffered saline; sEVs, small extracellular vesicles; SVF, stromal vascular fraction.
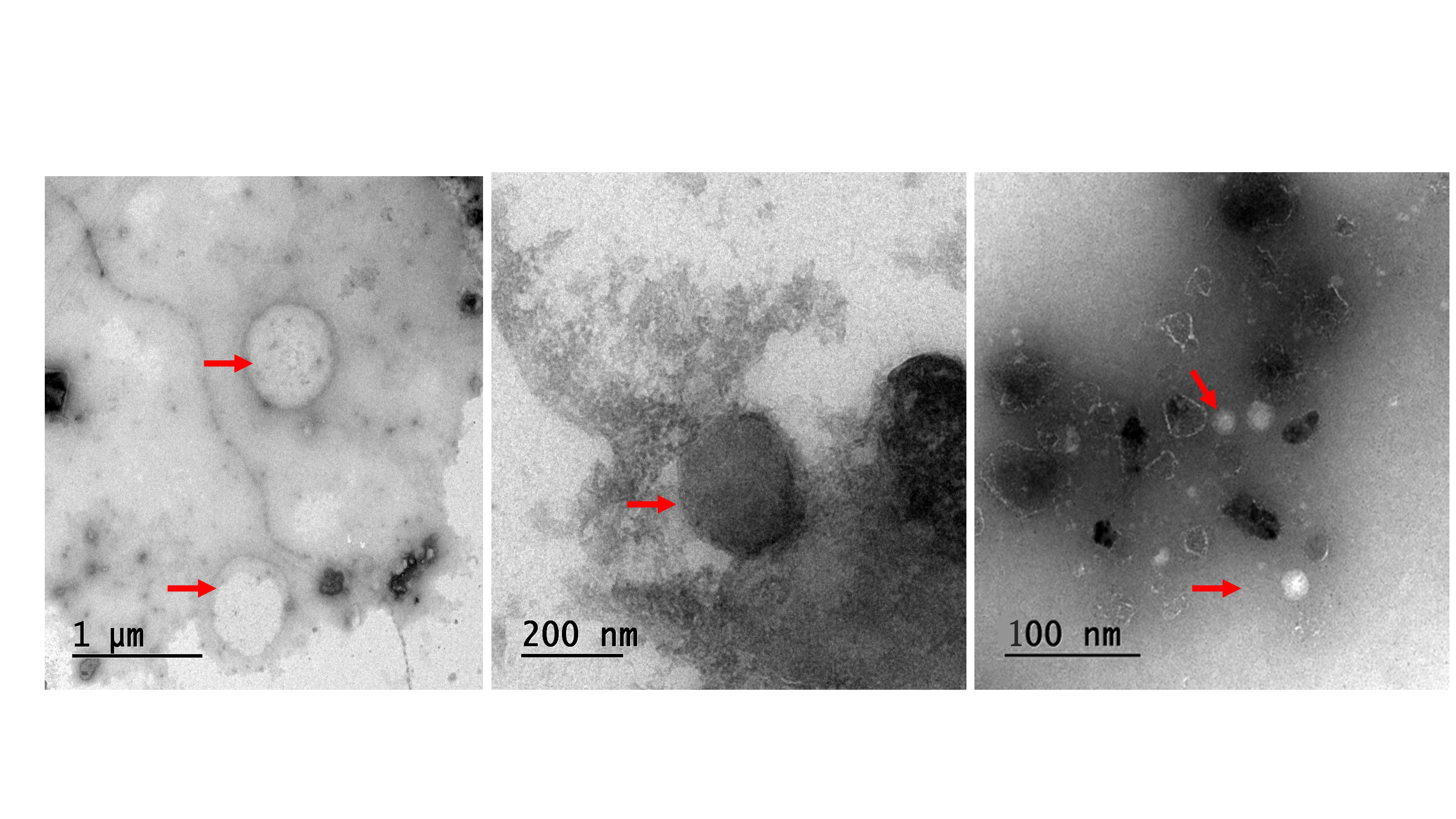
Morphological characterization of SVF-sEVs utilizing transmission electron microscopy (TEM). Representative images (i, ii, and iii) display intact, spherical structures resembling nanovesicles (indicated by red arrows), identified as SVF-sEVs. Abbreviations: sEVs, small extracellular vesicles; SVF, stromal vascular fraction; TEM, transmission electron microscopy.
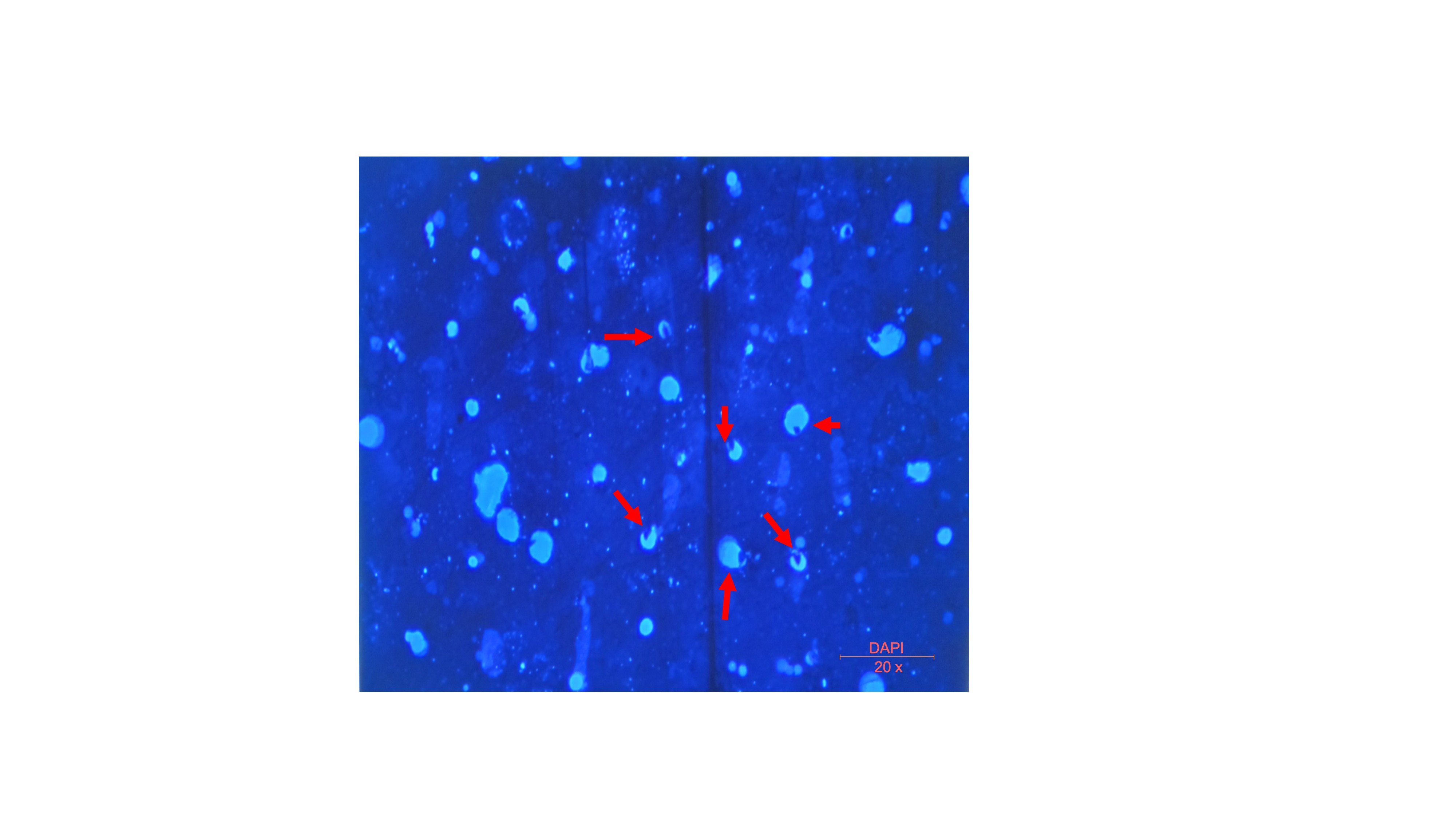
Fluorescence microscopy of SVF-sEVs. The red arrow indicates an intact, cup-shaped ultrastructure characteristic of sEVs. Abbreviations: sEVs, small extracellular vesicles; SVF, stromal vascular fraction.
Confirmation of MRSA Strains
A 533-base pair (bp) mecA gene amplicon was detected in both the ATCC MRSA reference strain and the clinical MRSA isolates. In contrast, the MSSA strains exhibited no amplification, thereby confirming their methicillin susceptibility (Figure 6).
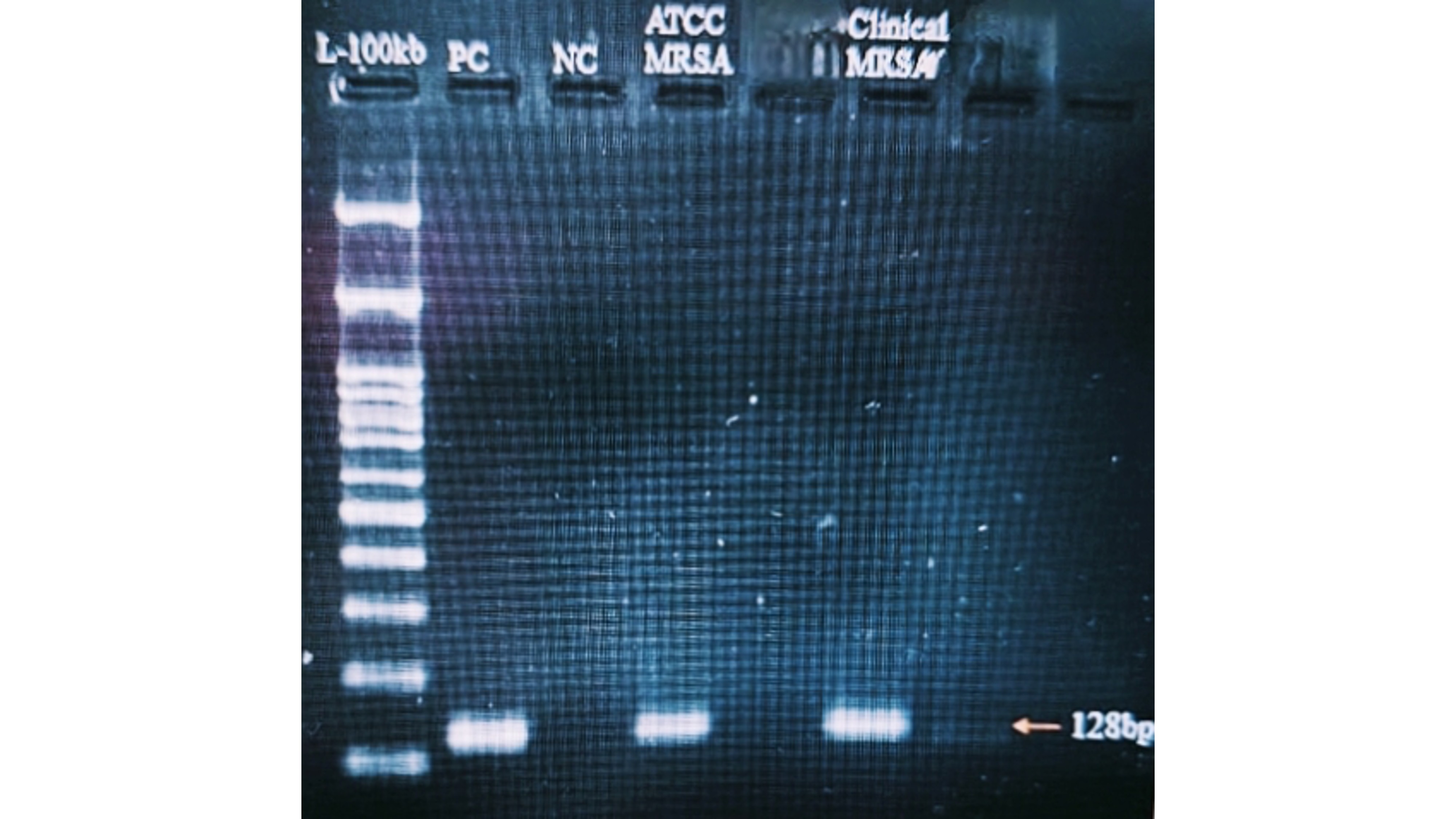
Detection of the
SVF-sEV In Vitro Antibacterial Activity
-
Agar Diffusion Assay—MRSA Inhibition by SVF-sEVs: All tested
S. aureus strains exhibited dose-dependent susceptibility to SVF-sEVs, with maximal inhibition observed at a 100% concentration. Neat SVF-sEVs generated zones of inhibition ranging from 12 mm to 30 mm across the tested strains (Figure 7i, ii). The ATCC MSSA and clinical MRSA isolates demonstrated the most pronounced inhibition. In contrast, diluted SVF-sEVs (≤25%) produced substantially smaller or negligible zones, indicating diminished efficacy. Ciprofloxacin disks (positive control) consistently yielded zones of inhibition comparable to those of the 100% SVF-sEVs, while PBS (negative control) exhibited no inhibition. -
Time-Kill Assay—Bactericidal Activity Over Time: The bactericidal effects of the SVF-sEVs were monitored longitudinally. Treatment with 100% neat SVF-sEVs induced a rapid reduction in viable
S. aureus counts within 4 hours, followed by a substantial decrease in the overall bacterial load at 24 hours (Figure 8). This time-dependent killing kinetic demonstrates the potent and sustained antibacterial activity of the SVF-sEVs. -
Colony-Forming Unit (CFU) Assay: Exposure to 100% SVF-sEVs resulted in substantial reductions in CFU counts in both MRSA and MSSA cultures at 6 and 24 hours compared with untreated controls. The ATCC MRSA and clinical MRSA/MSSA isolates exhibited enhanced susceptibility, demonstrating pronounced CFU declines by 24 hours. Consistent bacteriostatic and bactericidal effects were observed across all experimental replicates (Figure 9i, ii). Log₁₀-transformed CFU/mL analyses revealed statistically significant decreases following SVF-sEV treatment compared with the baseline (evaluated via a one-way ANOVA with Tukey’s
post hoc test or a Kruskal–Wallis test with Dunn’spost hoc test, as applicable), thereby supporting a robust antimicrobial effect. Given the exploratory design of this study, these results should be regarded as preliminary yet biologically relevant. -
Antibiofilm Activity of SVF-sEVs: SVF-sEV treatment resulted in a notable suppression of biofilm formation in both the ATCC reference and clinical
S. aureus strains. Untreated controls maintained moderate biofilm formation, whereas SVF-sEV-treated wells exhibited a reduction in biofilm intensity to weak or undetectable levels, with complete inhibition observed in the MRSA cultures. These antibiofilm effects were consistent across the tested strains; residual weak biofilm was detected solely in the CS-MSSA isolate at 24 hours (Figure 10). Based on OD₅₇₀ measurements and predefined classification criteria, the treated wells shifted from the strong or moderate categories to the weak or non-biofilm categories. The negative controls remained unstained, whereas the positive controls retained strong staining, indicative of mature biofilms. Table 1 summarizes the baseline biofilm formation (BF) and subsequent antibiofilm activity (ABF) derived from these analyses.
Semi-quantitative ordinal scale analysis of SVF-sEVs effect on biofilm formation (BF) and antibiofilm activity (ABF) against tested
| Assay | ATCC MRSA | ATCC MSSA | CS-MRSA | CS-MSSA | -Ve control (NC) | +ve control (PC) |
|---|---|---|---|---|---|---|
| BF | moderate (2) | moderate (2) | weak (1) | moderate (2) | no biofilm (0) | (2) |
| ABF | no biofilm (0) | no biofilm (0) | no biofilm (0) | weak(1) | no biofilm (0) | (0) |
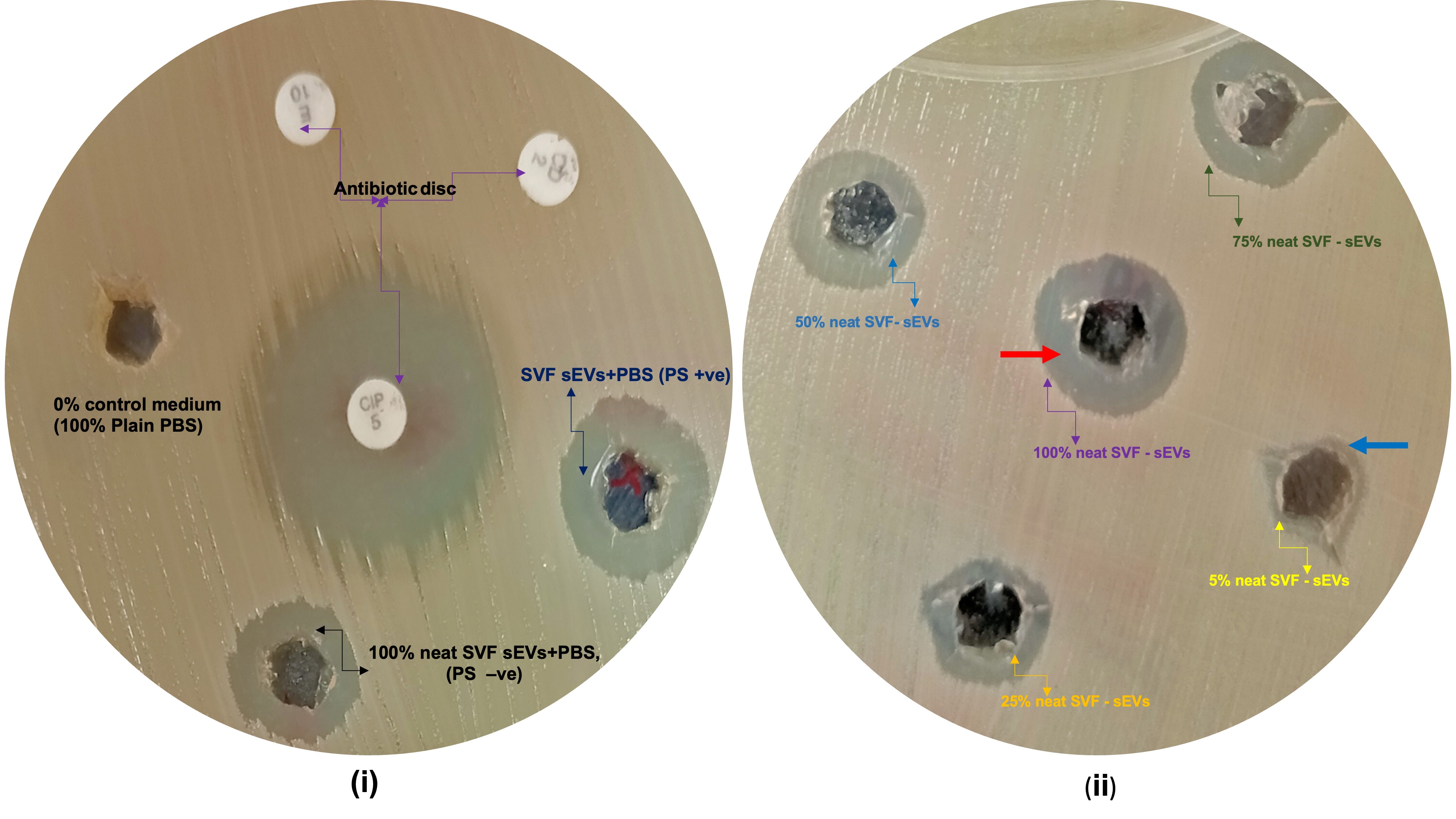
Agar diffusion assay evaluating the antibacterial activity of SVF-sEVs against
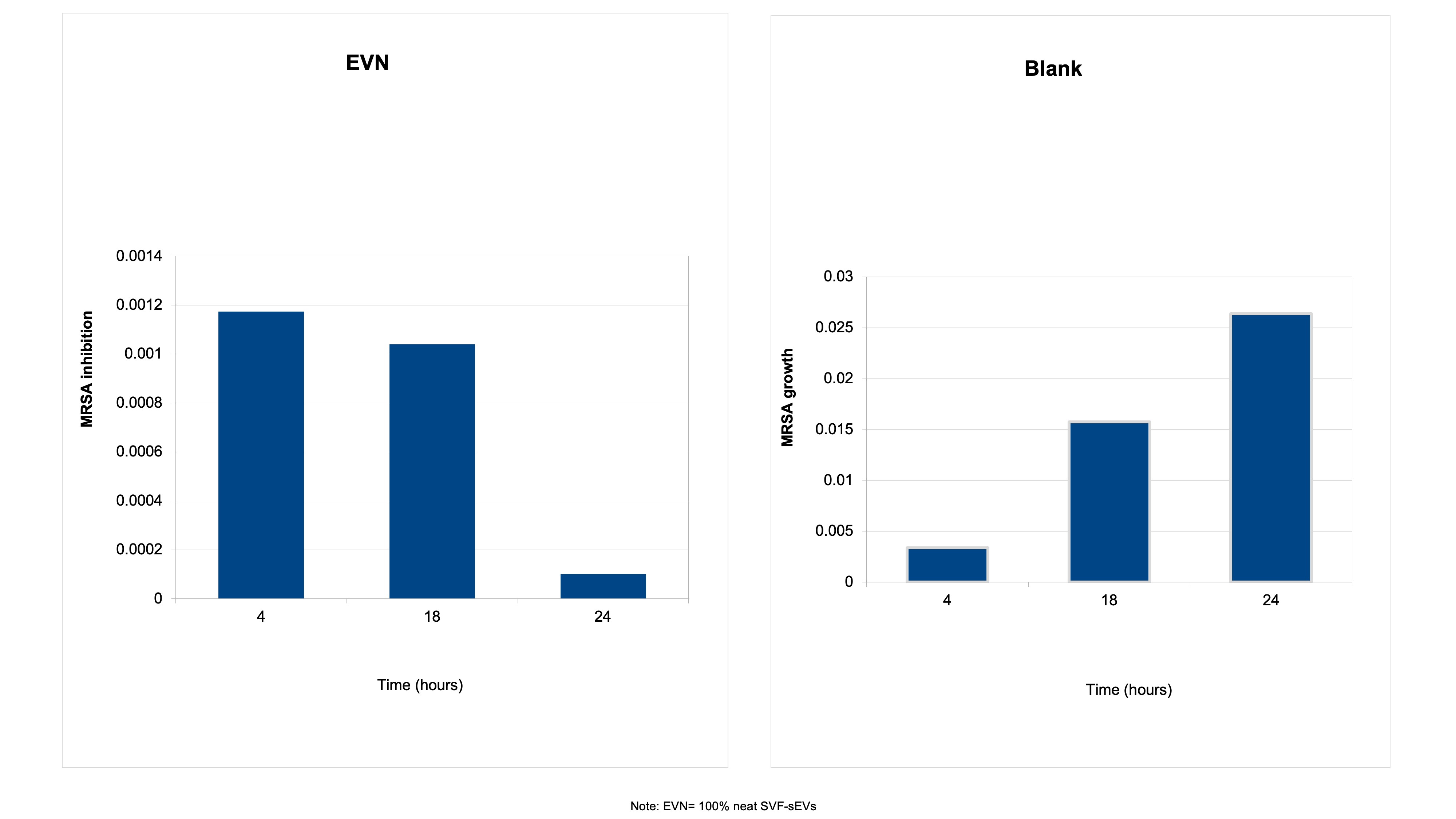
Time-kill kinetics of neat SVF-sEVs (EVN) against the ATCC MRSA strain. The antibacterial activity of EVN against
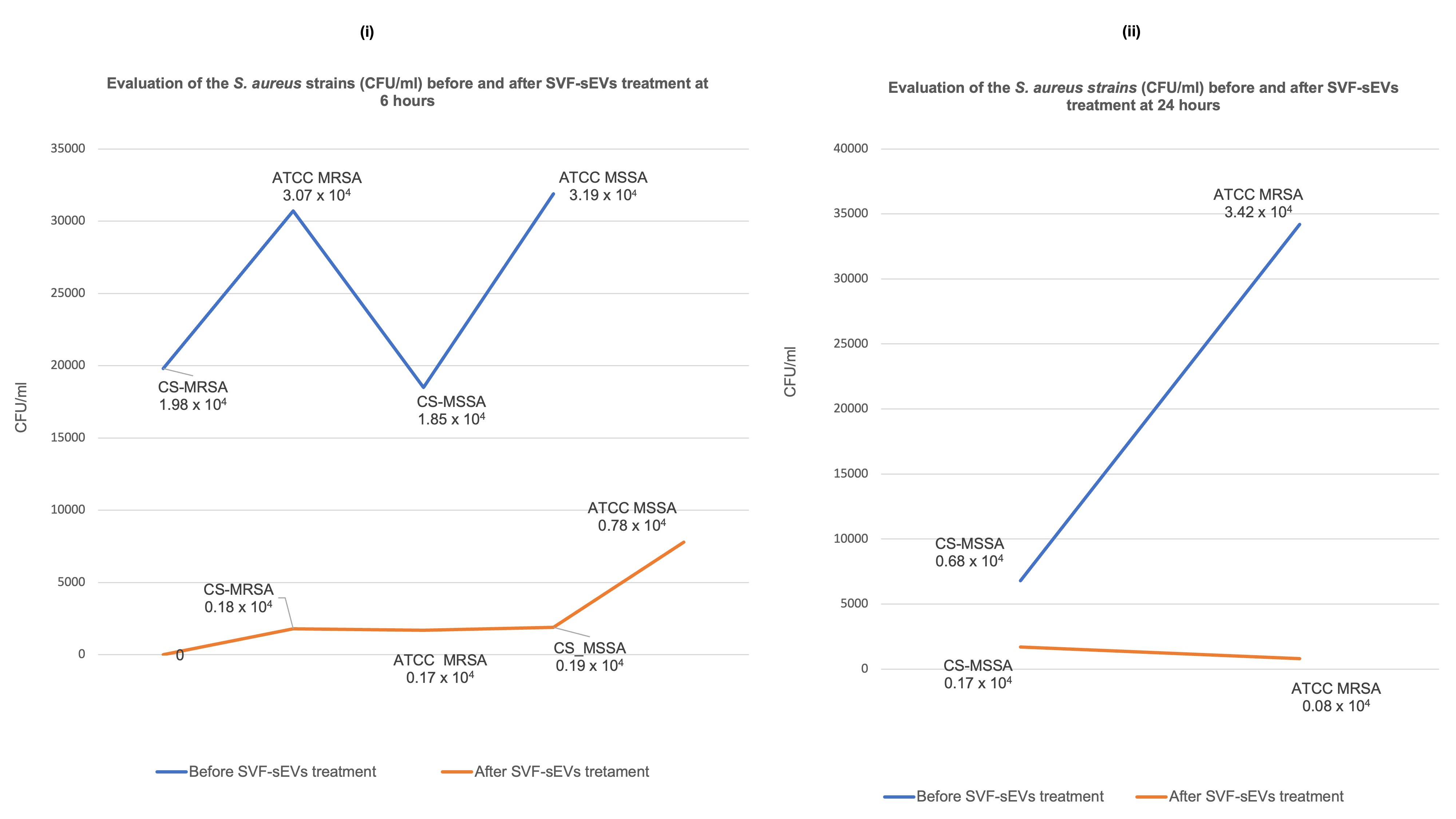
Quantitative colony-forming unit (CFU) assay assessing the viability of
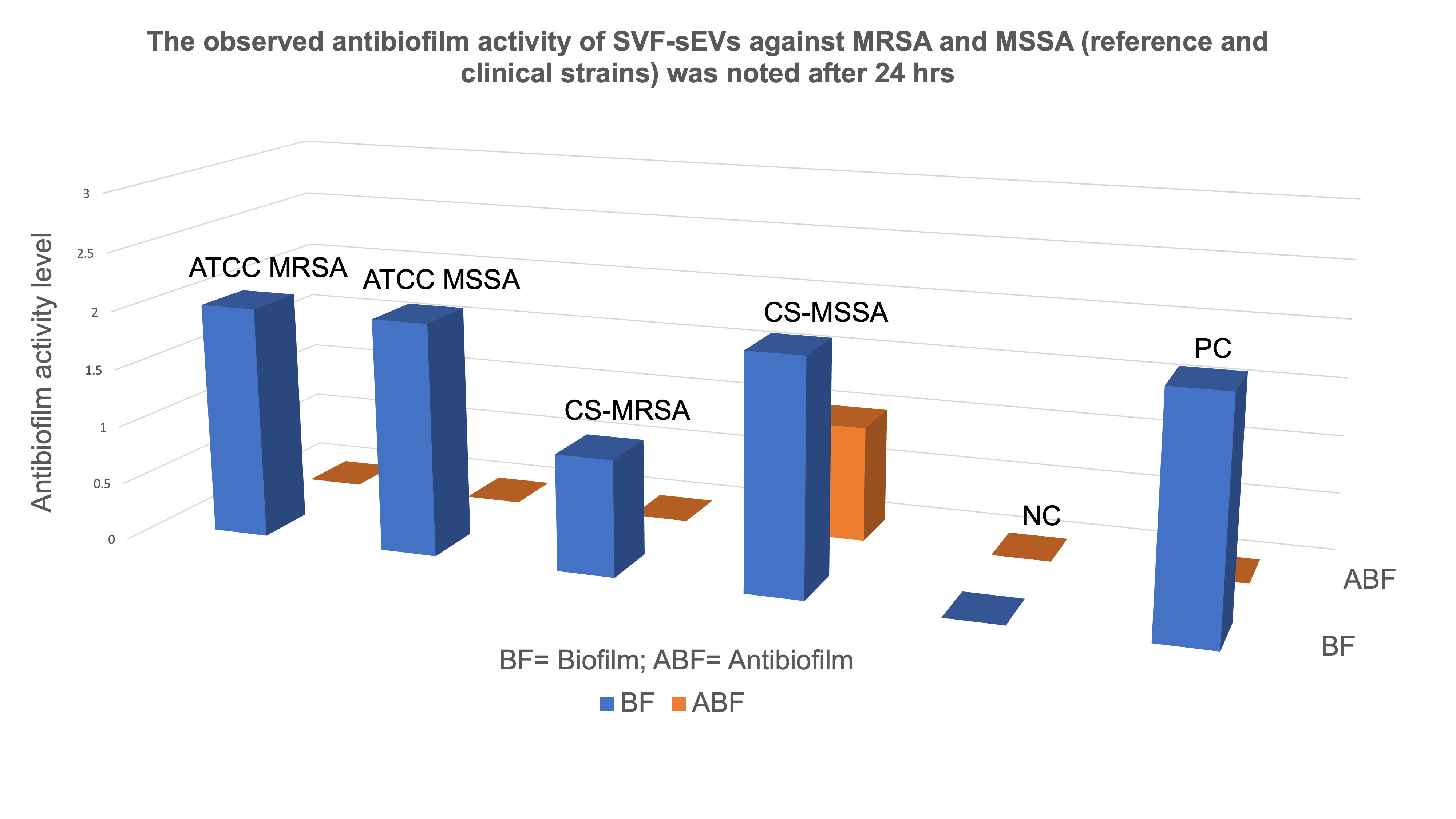
Antibiofilm activity of SVF-sEVs against
For quantitative classification, biofilm producers were categorized using an ordinal scale (corresponding to codes 0 through 3) based on the cut-off optical density (ODc). The negative control (NC) consisted solely of plain media, while the positive control (PC) comprised media inoculated with ATCC MRSA. The classification criteria were formally defined as follows:
-
0 (Non-biofilm producer): OD ≤ ODc
-
1 (Weak producer): ODc < OD ≤ 2 × ODc
-
2 (Moderate producer): 2 × ODc < OD ≤ 4 × ODc
-
3 (Strong producer): OD > 4 × ODc
Discussion
This study aimed to evaluate the antibacterial efficacy of SVF-sEVs against both clinical isolates and standard reference strains of MSSA and MRSA. Although previous studies have reported the antibacterial activity of mesenchymal cell-derived EVs, our demonstration of their efficacy against MRSA biofilms utilizing adipose SVF-derived sEVs represents a novel contribution to the field. This exploratory, proof-of-concept study was explicitly designed to evaluate the antimicrobial activity of SVF-sEVs utilizing standardized in vitro assays.
Antibacterial assays against the MRSA strains were conducted via optical density measurements at 600 nm (OD), recorded with a Synergy™ HTX microplate reader (BioTek Instruments, Winooski, VT, USA). Data were analyzed utilizing GraphPad Prism version 9.0 (GraphPad Software, San Diego, CA, USA). Both a clinical isolate of S. aureus and the methicillin-resistant reference strain ATCC 43300, sourced from the Central Research Laboratory's microbiology repository, were selected to assess the antibacterial activity and potential clinical significance of the SVF-sEVs. The findings demonstrated that a concentration of 2.15 × 10 EVs/100 µL of EVN (100% neat SVF-sEVs) exhibited significant antibacterial activity, albeit with variable efficacy across the tested strains.
The zone of inhibition assays demonstrated that EVN possess inherent antibacterial properties, as evidenced by a concentration-dependent increase in zone diameter. Specifically, EVN produced inhibition zones comparable to those of standard antibiotic disks (25–30 mm) against both the ATCC MSSA and clinical MRSA strains. These observations align with previous reports indicating that sEVs from mesenchymal sources can mediate antimicrobial effects, potentially via the transfer of bioactive molecules such as antimicrobial peptides, microRNAs (miRNAs), or metabolic enzymes.19 The complete absence of inhibitory activity in the PBS negative control confirms that the observed effects are specific to the SVF-sEVs rather than the suspension vehicle.
The time-kill assays further substantiated the antibacterial action of EVN, revealing a marked, time-dependent reduction in viable bacterial cells, particularly in the ATCC MSSA strain, within the first 4 to 6 hours post-treatment. This rapid onset of action suggests a direct antimicrobial mechanism, potentially mediated through bacterial membrane disruption or interference with essential cellular processes. This effect persisted for up to 24 hours, indicating the sustained activity of the SVF-sEVs. In the CFU assays, bacterial colony counts were consistently reduced across all strains at 24 hours compared with the 6-hour mark. The most pronounced reductions were observed in the clinical isolates (CS-MRSA and CS-MSSA), followed by the ATCC MRSA strain. Statistical testing was performed on log₁₀-transformed CFU data utilizing appropriate omnibus and post hoc methods. Collectively, these results support the hypothesis that 100% neat SVF-sEVs (EVN) can significantly inhibit the proliferation of S. aureus, irrespective of its specific resistance phenotype.
Biofilm formation is a critical virulence factor in S. aureus, particularly in MRSA infections, contributing to persistent and recurrent disease states due to enhanced antibiotic tolerance.20 Biofilm inhibition assays demonstrated that EVN can effectively disrupt preformed biofilms in both reference and clinical strains. This is a notable finding, given that few existing therapeutic agents exhibit consistent efficacy against mature biofilms. The moderate response observed in the CS-MSSA isolate may reflect intrinsic differences in biofilm matrix composition, metabolic status, or strain-specific vesicle uptake kinetics. Formal multiplicity correction was not strictly required, as only a single concentration (100% SVF-sEVs) and a highly limited set of biologically related bacterial strains were included in the final analysis. Overall, the data indicate that EVN exhibit multimodal antibacterial properties, affecting both planktonic cells and established biofilms in a time- and strain-dependent manner. Such variability may stem from underlying genetic and phenotypic differences, including variations in membrane composition, resistance gene expression profiles, and dynamic interactions with host-derived vesicular components. Further in vivo studies are warranted to comprehensively assess the therapeutic potential of this novel strategy.
Furthermore, SVF-sEVs offer several distinct practical advantages. As naturally derived vesicles with inherent antibacterial properties, they are highly biocompatible with the human immune system and function as cell-free, drug-free therapeutic agents. They are also significantly less likely than synthetic antibiotics to drive bacterial resistance, positioning them as sustainable, host-derived alternatives in the context of global antimicrobial resistance (AMR). Additionally, the relative abundance of adipose tissue supports the large-scale production of high-quality sEVs. Their minimally invasive procurement from immune and stromal cell populations—which are already recognized for their therapeutic utility—further enhances their translational appeal.
Study Limitations
Despite these encouraging findings, several important limitations must be acknowledged:
-
Exploratory Design and Sample Size: A primary constraint is the exploratory,
in vitro design combined with a small sample size, which limits statistical power and precludes definitive conclusions regarding precise effect sizes and dose-response relationships. Accordingly, only basic statistical methods appropriate for the available data were applied, and the current results are presented strictly as hypothesis-generating. Larger studies with greater biological replication and comprehensive dose-response testing are requisite to validate and refine these initial observations. -
Mechanistic and Translational Gaps: The molecular mechanisms underlying the antibacterial effects of SVF-sEVs were not investigated, nor were
in vivo validation studies performed; both remain essential steps toward the development of dosage-based nanotherapies and personalized treatment regimens. Key translational parameters, such as donor variability, long-term vesicle stability, and the immunomodulatory behavior of SVF-sEVs under physiological conditions, were not evaluated, though they are critical for assessing true clinical feasibility. -
Dosing and Proteomic Profiling: SVF-sEVs were evaluated at only a single concentration (100%), and quorum sensing (QS) gene expression relative to antibiofilm activity was not examined. This limits mechanistic interpretation and the ability to define precise therapeutic windows. Future studies must measure and report the particle concentration corresponding to each EVN treatment level to enhance translational relevance. Additionally, this study lacked proteomic or microRNA (miRNA) profiling of the SVF-sEVs, as well as comparative analyses with sEVs derived from non-antibacterial sources, thereby restricting insights into specific bioactive cargoes and signaling pathways.
-
Future Therapeutic Directions: Inhibition experiments (e.g., utilizing proteases, RNases, or other targeted inhibitors) to identify the specific active components responsible for the antibacterial activity were omitted but should be incorporated into subsequent studies. From a therapeutic perspective, combination regimens were not assessed. The co-administration of SVF-sEVs with conventional antibiotics represents a highly promising future direction, as it may synergistically enhance antibacterial efficacy while permitting dose reductions of traditional synthetic agents, thereby potentially mitigating the further emergence of AMR.
-
Statistical Power: A
post hoc power assessment indicated that the study lacked sufficient statistical power to detect small to moderate between-group differences, largely due to the qualitative scoring system employed for certain assays and the use of single observations per condition. Consequently, statistical analyses were intended to illuminate descriptive trends rather than provide definitive inferential conclusions. -
Safety and Scalability: The absence of formal endotoxin quantification and rigorous sterility testing is a key limitation of the present study and must be explicitly addressed in future work focused on clinical translation and scalable manufacturing.
Conclusion
While the majority of existing sEV-related antimicrobial studies focus on plant- or bacteria-derived vesicles, this investigation demonstrates the novel application of human-derived SVF-sEVs as intrinsic nanoscale antimicrobial and antibiofilm agents. This approach leverages host-derived vesicles possessing inherent antimicrobial properties to target both MRSA and MSSA, encompassing reference strains as well as clinical isolates. Consequently, our findings address an urgent clinical demand for innovative, highly biocompatible antibacterial agents that circumvent the reliance on cell-based therapies or conventional pharmacological strategies for the management of MRSA. The in vitro results presented herein provide a robust foundation for subsequent in vivo studies designed to thoroughly evaluate the safety, efficacy, and underlying molecular mechanisms of SVF-sEVs under physiological conditions. Ultimately, advancing this line of research has the potential to facilitate the development of next-generation, host-derived nano-antimicrobial therapeutics for the management of recalcitrant and difficult-to-treat infections.
Abbreviations
ABF: antibiofilm activity; AMR: Antimicrobial resistance; ANOVA: analysis of variance; ARM: Antibiotic-resistant microorganisms; ATCC: American Type Culture Collection; BCA: bicinchoninic acid; BF: biofilm formation; BHI: brain heart infusion; BMI: Body Mass Index; cCM: concentrated conditioned medium; CFU: Colony-forming unit; CLSI: Clinical and Laboratory Standards Institute; CS: clinical isolates; CV: crystal violet; DMEM: Dulbecco’s modified Eagle’s medium; DPBS: Dulbecco’s phosphate-buffered saline; EPS: extracellular polymeric substances; EVN: neat SVF-sEVs; EVs: Extracellular vesicles; FBS: Fetal bovine serum; FM: Fluorescent microscopy; GCLP: Good Clinical and Laboratory Practice; hHAT: healthy human adipose tissue; HRP: horseradish peroxidase; ISO: International Organization for Standardization; LAL: Limulus amebocyte lysate; LB: Luria-Bertani; MH: Mueller-Hinton; MHA: Mueller-Hinton agar; miRNAs: microRNAs; MRSA: Methicillin-Resistant Staphylococcus aureus; MSSA: Methicillin-Sensitive Staphylococcus aureus; NABL: National Accreditation Board for Testing and Calibration Laboratories; NC: negative control; NIST: National Institute of Standards and Technology; NTA: Nanoparticle tracking analysis; ODc: cut-off optical density; PBS: Phosphate buffer saline; PC: positive control; PCR: polymerase chain reaction; QS: quorum sensing; SD: standard deviation; SDS-PAGE: sodium dodecyl sulfate-polyacrylamide gel electrophoresis; sEVs: Small Extracellular Vesicles; STROBE: Strengthening the Reporting of Observational Studies in Epidemiology; SVF: Stromal Vascular Fraction; TDE: tissue digestion enzyme; TEM: Transmission electron microscopy; ti-sEVs: primary tissue sources derived small extracellular vesicles; WB: Western blotting; WHO: World Health Organization; ZOI: zone of inhibition
Acknowledgements
The authors would like to acknowledge the support of Nitte (Deemed to be University) in carrying out this study
Author’s contributions
The original draft was prepared by Dr VMHS. Formal analysis was conducted by Dr VMHS, Dr NSS, and Dr AVS. Writing, review, and editing were performed by Dr VMHS, Dr MKB, Dr PP, Dr NSS, and Dr AVS. All authors read and approved the final draft.
Availability of data and materials
Data and materials used and/or analyzed during the current study may be obtained from the corresponding author upon reasonable request. Some are available in the supplementary data.
Ethics approval and consent to participate
This study was performed in line with the principles of the Declaration of Helsinki (2004). Approval was granted by the Ethics Committee of NITTE University (Date: Jan 13 2022 /No. NU/CEC/2021/230).
Consent for publication
Not applicable
Declaration of generative AI and AI-assisted technologies in the writing process
The authors declare that no AI tools were used in the writing process prior to submission.
Competing interest
All authors have no competing interests.

