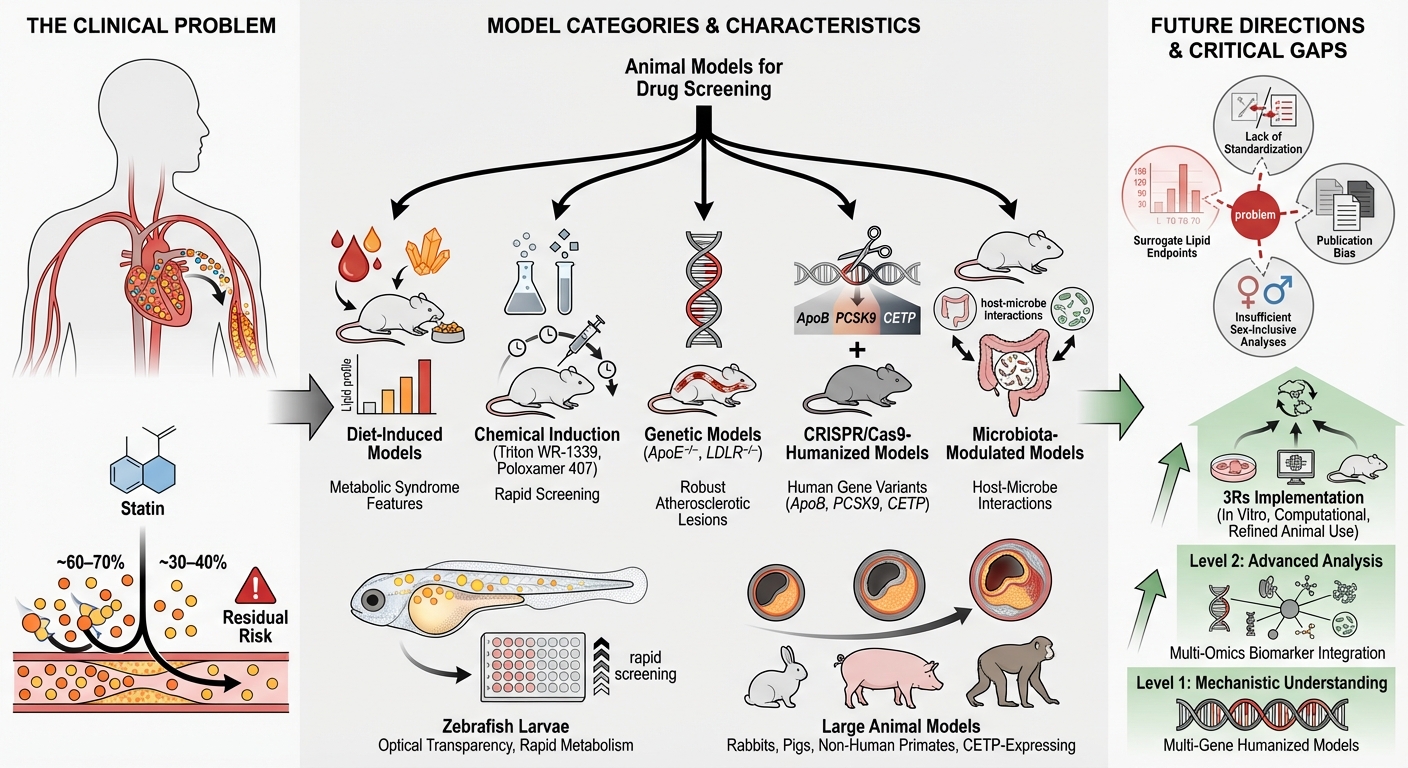
Integrative Insights into Animal Models for Antihyperlipidemic Drug Screening: Bridging Preclinical and Translational Gaps in Research
- Faculty of Pharmacy, Integral University, Kursi Road, Lucknow, India
Abstract
Hyperlipidemia is a principal modifiable risk factor for atherosclerosis and cardiovascular disease, underscoring the critical need for predictive preclinical models to evaluate lipid-lowering therapies. Animal models are indispensable for elucidating lipid metabolism, validating pharmacological targets, and screening novel antihyperlipidemic agents prior to clinical translation. This integrative review presents an updated, critical synthesis of the animal models utilized in antihyperlipidemic drug screening, encompassing diet-induced, chemically induced, genetically modified, microbiota-modulated, and large-animal systems. Each model is evaluated based on its physiological relevance, methodological characteristics, and translational reliability. Small animals—such as mice, rats, and hamsters—provide accessible, cost-effective, and genetically versatile platforms for mechanistic investigations and early-stage screening. Conversely, larger species, including rabbits, pigs, and non-human primates, more accurately replicate human lipid profiles, atherosclerotic pathology, and cardiovascular physiology. Recent advancements in microbiota modulation, CRISPR-based genetic engineering, and multi-omics technologies have significantly enhanced mechanistic understanding and improved predictive accuracy in these models. By integrating classical and next-generation methodologies, this review proposes a translational framework designed to support ethical, reproducible, and clinically relevant preclinical research in lipid pharmacology.
Introduction
Cardiovascular diseases (CVDs) remain the leading cause of mortality worldwide, accounting for approximately 17.9 million deaths annually—representing nearly 32% of all global deaths 1. Among the modifiable risk factors contributing to cardiovascular morbidity and mortality, hyperlipidemia occupies a central role. It is characterized by elevated plasma concentrations of total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), and triglycerides (TG), alongside reduced levels of high-density lipoprotein cholesterol (HDL-C). These lipid derangements significantly accelerate atherogenesis and its associated clinical complications 2,3. Hyperlipidemia drives endothelial dysfunction, arterial lipid deposition, and chronic vascular inflammation. Consequently, it remains a primary therapeutic target in the prevention and management of atherosclerotic cardiovascular disease.
Clinical Burden and Therapeutic Limitations
Hyperlipidemia is a core component of metabolic syndrome and frequently coexists with obesity, insulin resistance, and hypertension. Its global prevalence continues to rise, driven by sedentary lifestyles, high-fat dietary patterns, and an aging population 4. While statins remain the cornerstone of pharmacological therapy, their clinical utility is occasionally constrained by adverse effects, residual cardiovascular risk, interindividual response variability, and long-term adherence challenges 5. These limitations have stimulated the development of alternative lipid-lowering strategies, such as PCSK9 inhibitors, cholesterol absorption inhibitors, CETP inhibitors, and nutraceutical-based interventions. Concurrently, the advent of precision medicine has amplified the demand for preclinical models that more accurately recapitulate human lipid metabolism and disease heterogeneity 6. Historically, traditional drug discovery pipelines have been hindered by poor translation from animal studies to clinical outcomes. This translational gap primarily stems from species-specific divergences in lipoprotein metabolism, inflammatory responses, and plaque biology 7,8.
Role of Animal Models in Hyperlipidemia Research
Animal models provide a critical foundation for investigating lipid metabolism, identifying therapeutic targets, and evaluating the safety and efficacy of candidate antihyperlipidemic agents prior to human trials 8. These models are broadly classified into three categories. First, induced models utilize dietary, chemical, or hormonal interventions to generate hyperlipidemia. Second, genetically modified models—encompassing knockout, knock-in, or transgenic animals—are designed to mimic inherited dyslipidemias, such as familial hypercholesterolemia. Third, spontaneous models, exemplified by the Watanabe heritable hyperlipidemic (WHHL) rabbit, naturally develop hyperlipidemia and atherosclerosis over time. Each model presents distinct advantages for mechanistic and translational research. However, species-specific limitations persist, notably differences in cholesteryl ester transfer protein (CETP) activity, apolipoprotein expression, and LDL receptor regulation. These factors demand careful consideration when interpreting experimental outcomes and extrapolating preclinical findings to human physiology 8,9,10.
Importance of Model Selection and Standardization
Appropriate model selection is paramount for the meaningful preclinical evaluation of antihyperlipidemic therapies. A mismatch between a compound's pharmacodynamic properties and the metabolic background of the selected model can yield misleading efficacy or safety conclusions. For instance, wild-type mice lack CETP activity, inherently limiting their suitability for evaluating HDL-targeted therapies. In contrast, rabbits and non-human primates express CETP, thereby more closely mirroring human lipoprotein dynamics 9,10,11. Beyond model selection, inadequate standardization of induction protocols, dietary compositions, outcome measures, and reporting practices continues to be a major source of irreproducibility in preclinical research. International guidelines and regulatory frameworks—including those established by the OECD, ICH, and the ARRIVE consortium—strongly emphasize the necessity of ethical conduct, methodological rigor, and transparent reporting in animal studies 12.
Emerging Trends in Model Development
Recent technological advancements have significantly expanded the scope and sophistication of animal models utilized in lipid research. CRISPR/Cas9-mediated genome editing has facilitated the generation of humanized models that express key human apolipoproteins, LDL receptors, or PCSK9 variants. These innovations have markedly enhanced the biological relevance of preclinical models for investigating inherited and complex lipid disorders 13. Furthermore, multi-omics approaches—encompassing transcriptomics, proteomics, metabolomics, and lipidomics—are increasingly being integrated into animal studies to deliver systems-level insights into lipid regulation and pharmacological responses. Non-invasive imaging modalities, advanced biomarker profiling, and computational modeling further elevate the precision of disease monitoring and therapeutic evaluation 14. The present review aims to provide a comprehensive and critical overview of the animal models employed in antihyperlipidemic drug screening. Emphasis is placed on their biological relevance, mechanistic utility, and translational potential, whilst also identifying persistent gaps and opportunities for refining the predictive validity of preclinical lipid research.
Methods: Literature Search and Study Selection
Search Strategy
A comprehensive and systematic literature search was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA 2020) guidelines to identify studies evaluating animal models utilized for antihyperlipidemic drug screening. Four electronic databases—PubMed/MEDLINE, Web of Science (Core Collection), Scopus, and Google Scholar—were queried to ensure a broad capture of experimental, mechanistic, and translational literature.
In PubMed/MEDLINE, the search strategy integrated Medical Subject Headings (MeSH) and free-text terms using Boolean operators, structured as follows: (“Hyperlipidemia”[MeSH] OR “Dyslipidemias”[MeSH] OR hyperlipidemia OR dyslipidemia OR hypercholesterolemia OR hypertriglyceridemia) AND (“Animal Models”[MeSH] OR “Disease Models, Animal”[MeSH] OR animal model* OR rodent* OR mouse OR mice OR rat OR rabbit OR hamster OR zebrafish OR primate*) AND (“Drug Screening” OR “Pharmacological Screening” OR “Preclinical Evaluation” OR antihyperlipidemic*). Detailed, database-specific search strategies are outlined in Supplementary Table S1. Boolean operators, truncation (*), and controlled vocabularies (e.g., MeSH) were tailored to meet the specific requirements of each database. For Google Scholar, simplified keyword combinations were applied, and the first 300 relevance-ranked records were screened to mitigate redundancy and exclude low-quality retrievals.
Eligibility Criteria
Across all databases, the search was restricted to English-language publications released between January 2000 and March 2025. Eligible studies comprised original research articles, as well as narrative and systematic reviews, that delineated animal models of hyperlipidemia, dyslipidemia, or atherosclerosis within the context of pharmacological screening or preclinical evaluation of antihyperlipidemic agents.
Studies were excluded if they met any of the following criteria:
-
Consisted exclusively of
in vitro or non-animal methodologies. -
Focused on disease models extraneous to lipid metabolism or atherosclerosis.
-
Lacked a pharmacological intervention or drug-screening component.
-
Were formatted as editorials, conference abstracts, commentaries, or opinion pieces.
"Non-relevant disease models" were defined a priori as those devoid of altered lipid metabolism or not explicitly intended for antihyperlipidemic drug evaluation.
Study Selection and Data Extraction
The initial database search yielded 2,453 records. Following the removal of 658 duplicate records utilizing reference management software (Zotero), 1,795 unique records advanced to title and abstract screening. During this phase, 1,303 records were excluded, categorized as in vitro or non-animal studies (n = 543), conference abstracts or editorials (n = 236), and non-relevant disease models (n = 524). Subsequently, 492 full-text articles underwent rigorous eligibility assessment.
At the full-text evaluation stage, an additional 205 articles were excluded, predominantly due to inadequate methodological detail, an absence of pertinent lipid-related outcomes, or publication in a language other than English (n = 72). Ultimately, 287 studies fulfilled all inclusion criteria and were incorporated into the qualitative synthesis.
Data extraction systematically captured the animal species and strain, model induction methodology (e.g., dietary, chemical, genetic, microbiota-modulated, or large-animal), pharmacological intervention, evaluated lipid parameters, and translational relevance. The selected studies were qualitatively synthesized by categorizing the models into diet-induced, chemically induced, genetic, microbiota-modulated, and large-animal paradigms. Any discrepancies arising during the screening or data extraction processes were resolved via consensus among the authors. The comprehensive study selection workflow is visually detailed in Figure 1 (PRISMA Flow Diagram).
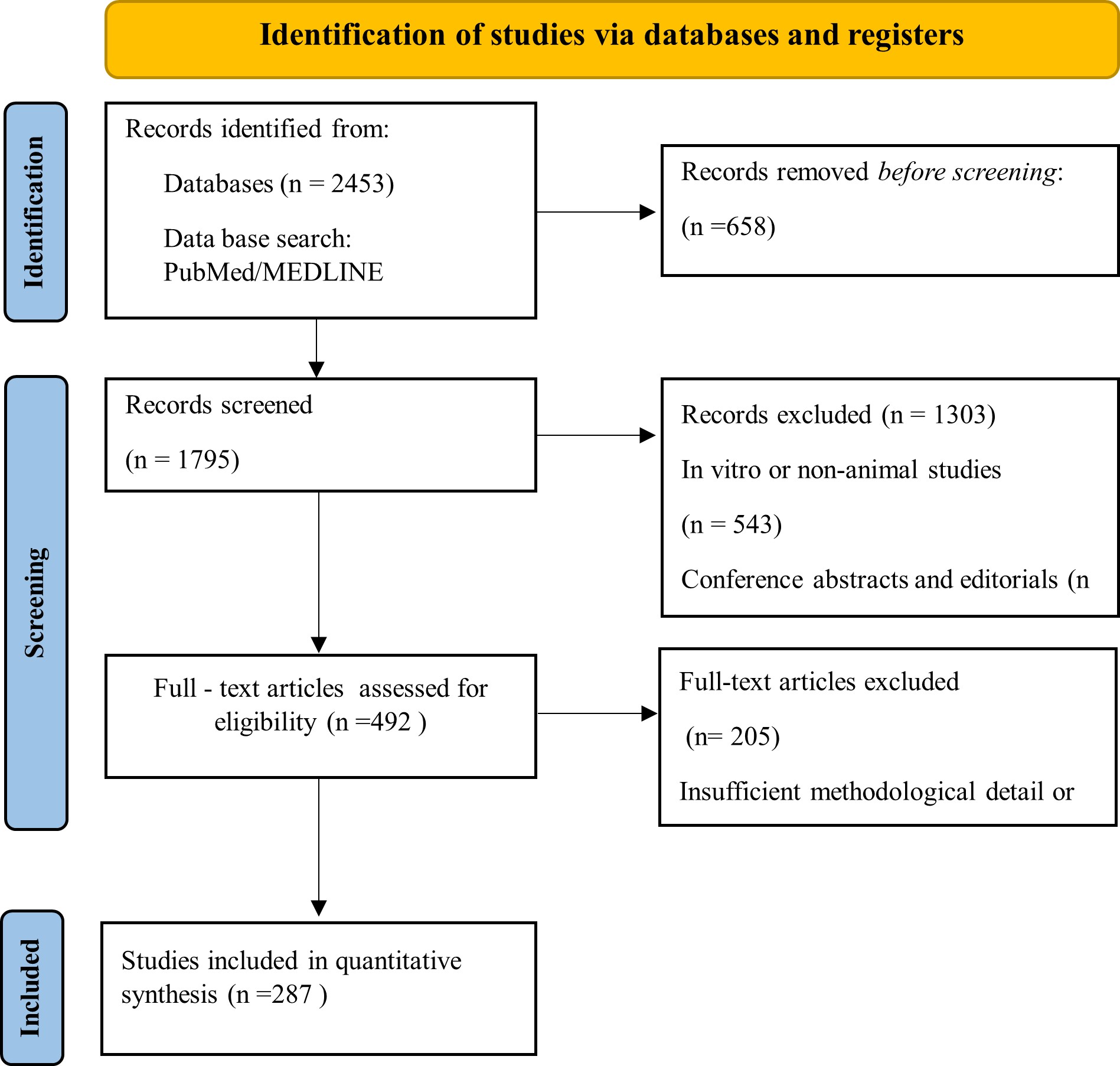
PRISMA 2020 flow diagram. This diagram details the systematic literature search, screening process, eligibility assessment, and final inclusion of studies evaluating animal models utilized in antihyperlipidemic drug screening.
Lipoprotein Function and Classification
Overview of Lipid Transport
Lipids—encompassing triglycerides (TG), cholesterol, phospholipids, and cholesteryl esters—are hydrophobic molecules essential for cellular architecture, membrane integrity, and energy metabolism. Due to their restricted solubility in aqueous environments, lipids necessitate specialized transport mechanisms to circulate within the bloodstream. This critical physiological function is fulfilled by lipoproteins 13,14. Lipoproteins are spherical macromolecular complexes comprising a hydrophobic lipid core—predominantly containing triglycerides and cholesteryl esters—enveloped by an amphipathic shell of phospholipids, unesterified cholesterol, and apolipoproteins 14,15,16. Beyond lipid transport, lipoproteins govern various physiological processes, including cellular lipid uptake, reverse cholesterol transport, steroidogenesis, and the maintenance of cellular membrane fluidity 16,17,18.
Structure and Components of Lipoproteins
Although lipoproteins exhibit heterogeneity in size and composition, they share a conserved structural architecture. The hydrophobic core houses neutral lipids, specifically triglycerides and cholesteryl esters, whereas the surface monolayer is enriched with phospholipids, unesterified cholesterol, and apolipoproteins. Apolipoproteins are integral to maintaining structural stability, facilitating receptor-mediated interactions, and modulating the activity of key metabolic enzymes. For instance, apolipoprotein B-100 (ApoB-100) is obligatory for the assembly and secretion of very-low-density lipoprotein (VLDL) and its subsequent binding to the LDL receptor. Apolipoprotein A-I (ApoA-I) serves as the primary structural protein of high-density lipoprotein (HDL) and acts as a vital activator for lecithin-cholesterol acyltransferase (LCAT). Furthermore, apolipoprotein C-II activates lipoprotein lipase (LPL), whereas apolipoprotein E (ApoE) mediates the hepatic clearance of remnant lipoproteins 16,18.
Classification of Lipoproteins
Lipoproteins are systematically classified based on their hydrated density, particle diameter, lipid-to-protein ratio, electrophoretic mobility, and specific apolipoprotein composition. According to these physicochemical properties, lipoproteins are categorized into chylomicrons, VLDL, intermediate-density lipoproteins (IDL), LDL, and HDL (Table 1) 2,5,15,16.
Classification of Lipoproteins.
| Lipoprotein | Major Lipid Component | Key Apolipoproteins | Primary Function | Atherogenic Potential |
|---|---|---|---|---|
| Chylomicrons | Triglycerides | ApoB-48, ApoC-II, ApoE | Transport dietary lipids from intestine | Low |
| VLDL | Triglycerides | ApoB-100, ApoC-II, ApoE | Transport endogenous triglycerides | Moderate |
| IDL | Triglycerides & cholesteryl esters | ApoB-100, ApoE | VLDL remnant metabolism | Moderate |
| LDL | Cholesteryl esters | ApoB-100 | Cholesterol delivery to peripheral tissues | High |
| HDL | Phospholipids, cholesterol | ApoA-I, ApoA-II | Reverse cholesterol transport | Protective |
Each lipoprotein class fulfills a specialized physiological role. Chylomicrons are responsible for transporting dietary triglycerides from the intestinal lumen to peripheral tissues. VLDL particles distribute endogenously synthesized triglycerides from the liver to peripheral sites. LDL functions as the principal carrier of cholesterol to peripheral cells, while HDL mediates reverse cholesterol transport by mobilizing excess peripheral cholesterol back to the liver for biliary excretion.
Functional Pathways of Lipoprotein Transport
Lipoprotein metabolism is meticulously regulated via three primary transport pathways.
Exogenous Pathway
The exogenous pathway commences with the intestinal absorption of dietary lipids. Enterocytes synthesize chylomicrons, which subsequently enter the systemic circulation via the lymphatic network. The triglycerides packaged within chylomicrons undergo hydrolysis by LPL, liberating free fatty acids for cellular energy utilization or adipocyte storage. The resultant chylomicron remnants are then rapidly cleared from the circulation by the liver through ApoE-mediated receptor endocytosis 13,14.
Endogenous Pathway
The endogenous pathway is driven by the hepatic assembly and secretion of VLDL particles, which transport endogenous triglycerides to peripheral tissues. Progressive triglyceride hydrolysis remodels VLDL into IDL, and ultimately into LDL. LDL particles deliver cholesterol to peripheral tissues and hepatocytes primarily via LDL receptor-mediated endocytosis. Perturbations in LDL receptor functionality, ApoB structural integrity, or PCSK9 regulation precipitate impaired LDL clearance, culminating in hypercholesterolemia 15,16.
Reverse Cholesterol Transport
Reverse cholesterol transport is predominantly orchestrated by HDL particles. HDL effluxes excess cholesterol from peripheral tissues, including lipid-laden macrophages residing within the arterial intima. Within the HDL particle, LCAT esterifies free cholesterol, while cholesteryl ester transfer protein (CETP) facilitates the equimolar exchange of cholesteryl esters and triglycerides between HDL and ApoB-containing lipoproteins. Ultimately, the transferred cholesterol is cleared by the liver and excreted in the form of bile acids 17,18.
Key Enzymes Regulating Lipoprotein Metabolism
Several key enzymes exert critical control over lipoprotein remodeling and systemic lipid homeostasis (Table 2). LPL catalyzes the hydrolysis of triglycerides sequestered in chylomicrons and VLDL, thereby facilitating lipid clearance. Hepatic lipase modifies HDL and IDL particles, significantly influencing hepatic cholesterol turnover. Furthermore, LCAT is indispensable for HDL maturation, whereas CETP governs the dynamic distribution of cholesterol across distinct lipoprotein fractions. Intracellularly, acyl-CoA:cholesterol acyltransferase (ACAT) dictates cholesterol esterification, a process central to macrophage foam cell formation 16,18. Pathological alterations in the activities of these enzymes can predispose individuals to lipid accumulation, atherogenesis, and systemic metabolic dysfunction (Table 2).
Enzymes Regulating Lipoprotein Metabolism.
| Enzyme | Primary Function | Lipoprotein Affected | Clinical Relevance |
|---|---|---|---|
| Lipoprotein lipase (LPL) | Hydrolyzes triglycerides | Chylomicrons, VLDL | TG clearance |
| Hepatic lipase | Lipoprotein remodeling | HDL, IDL | HDL metabolism |
| LCAT | Cholesterol esterification | HDL | HDL maturation |
| CETP | Lipid exchange | HDL ↔ LDL/VLDL | Translational relevance |
| ACAT | Cholesterol esterification | Macrophages | Foam cell formation |
Clinical and Experimental Implications
Dysregulated lipoprotein metabolism forms the fundamental pathological basis for numerous cardiometabolic disorders. Elevated concentrations of circulating LDL and oxidatively modified LDL actively promote endothelial dysfunction and macrophage foam cell generation, acting as the primary instigators of atherosclerosis. Moreover, increased levels of VLDL and small, dense LDL particles are hallmark features of insulin-resistant states and diabetic dyslipidemia. Conversely, diminished HDL concentrations compromise reverse cholesterol transport and attenuate its inherent atheroprotective properties 7,9.
These distinct, species-specific variations in lipoprotein metabolism must meticulously guide animal model selection during experimental design. Models inherently lacking CETP activity, such as wild-type mice, inadequately recapitulate human HDL dynamics. In contrast, CETP-expressing species—including rabbits, hamsters, and non-human primates—offer substantially improved translational relevance for the preclinical evaluation of HDL-targeted therapeutic interventions 15,19,20.
Atherosclerosis – Pathogenesis and Relevance to Hyperlipidemia
Atherosclerosis is a chronic, progressive inflammatory disease of the arterial wall, characterized by lipid accumulation, immune cell infiltration, and fibrous tissue formation. It constitutes the primary pathological basis for ischemic heart disease, ischemic stroke, and peripheral artery disease, which collectively account for a substantial proportion of global cardiovascular mortality. Dyslipidemia—specifically, elevated levels of LDL-C and triglycerides coupled with reduced HDL-C—serves as a principal driver of atherogenesis. Hyperlipidemia contributes fundamentally to disease initiation and progression by promoting oxidative stress, endothelial dysfunction, foam cell formation, and deleterious vascular remodeling 15,16,17.
Pathophysiological Stages of Atherogenesis
The development of atherosclerosis proceeds through a continuum of overlapping pathophysiological stages intricately linked to underlying lipid abnormalities (Table 3) 15,16,17,18.
Key Stages in the Pathogenesis of Atherosclerosis and Associated Lipid Abnormalities.
| Stage | Pathological Event | Associated Lipid Abnormality | Experimental Relevance |
|---|---|---|---|
| Endothelial dysfunction | Increased permeability | Elevated LDL | Early disease modeling |
| Fatty streak formation | Foam cell accumulation | Oxidized LDL | Common in rodents |
| Plaque progression | SMC proliferation | Persistent dyslipidemia | Intermediate models |
| Plaque destabilization | Fibrous cap thinning | Inflammatory lipids | Large animals |
| Plaque rupture | Thrombosis | Lipoprotein(a), LDL | Rare in rodents |
Endothelial Dysfunction
Elevated circulating LDL particles infiltrate the arterial intima, where they are subjected to oxidative modification. The resultant oxidized LDL (oxLDL) triggers potent pro-inflammatory and pro-thrombotic signaling cascades, culminating in endothelial activation. Activated endothelial cells upregulate the expression of adhesion molecules, such as VCAM-1 and ICAM-1, thereby facilitating the recruitment and firm adhesion of circulating leukocytes 21,22.
Monocyte Recruitment and Foam Cell Formation
Monocytes adhere to the activated endothelium, transmigrate into the intimal space, and differentiate into mature macrophages. These macrophages avidly internalize oxLDL via scavenger receptors, including CD36 and SR-A1, driving their transformation into lipid-laden foam cells. The progressive accumulation of these foam cells yields fatty streaks, which represent the earliest macroscopically visible atherosclerotic lesions 23.
Smooth Muscle Cell Proliferation and Fibrous Cap Formation
Vascular smooth muscle cells (VSMCs) migrate from the tunica media into the intima, where they undergo phenotypic switching and subsequent proliferation. These cells synthesize substantial quantities of extracellular matrix components, predominantly collagen and elastin, contributing to the formation of a fibrous cap. This fibrous cap serves to stabilize the atheromatous plaque by sequestering the highly thrombogenic, lipid-rich necrotic core from the luminal bloodstream 24.
Necrotic Core Formation and Plaque Rupture
Unrelenting localized inflammation, coupled with foam cell apoptosis and secondary necrosis, leads to the progressive expansion of the necrotic core. Concurrently, macrophage-derived matrix metalloproteinases (MMPs) enzymatically degrade the extracellular matrix of the fibrous cap, significantly compromising plaque stability. Plaque rupture eventually exposes the highly thrombogenic necrotic core to circulating coagulation factors, triggering acute thrombosis that can precipitate severe clinical events, including myocardial infarction or ischemic stroke 25.
Immunological Mechanisms in Atherosclerosis
Atherosclerosis is increasingly characterized as a complex immunometabolic disorder governed by both innate and adaptive immune responses. In the innate arm, macrophages, dendritic cells, and neutrophils heavily contribute to localized cytokine production and the propagation of the necrotic core. Pro-inflammatory cytokines, notably IL-1β, TNF-α, and MCP-1, serve to amplify lesion progression. Adaptive immune responses are equally critical; for instance, Th1-polarized T cells secrete interferon-γ (IFN-γ), which potently activates macrophages while simultaneously inhibiting collagen synthesis by VSMCs, thereby fostering plaque instability. Consequently, targeting inflammatory pathways has emerged as a vital complementary strategy in cardiovascular therapeutics, a paradigm exemplified by the CANTOS trial, which demonstrated a significant reduction in cardiovascular events following IL-1β inhibition, independent of lipid-lowering efficacy 17,26,27.
Role of Lipoproteins in Atherogenesis
Distinct lipoprotein classes contribute differentially to plaque pathogenesis. LDL serves as the primary atherogenic particle, with its oxidative modification acting as a requisite initiating event in lesion formation. Conversely, HDL exerts profound atheroprotective effects, primarily mediated through reverse cholesterol transport and intrinsic anti-inflammatory properties. Lipoprotein(a) [Lp(a)], which contains the highly homologous apolipoprotein(a), possesses dual pro-atherogenic and pro-thrombotic properties and is frequently elevated in patients with familial hyperlipidemia. Importantly, species-specific variations in baseline lipoprotein profiles significantly dictate susceptibility to atherosclerosis, thereby determining the translational relevance of specific animal models employed in drug screening 28.
Relevance in Animal Models
The accurate biological reproduction of atherosclerotic pathology is a prerequisite for the rigorous evaluation of antihyperlipidemic agents. When subjected to high-fat diets, ApoE⁻/⁻ and LDLR⁻/⁻ murine models reliably develop atherosclerotic lesions localized to the aorta and aortic root; however, the architectural complexity of these plaques remains somewhat limited relative to human disease 28. In contrast, WHHL rabbits and cholesterol-fed New Zealand White rabbits consistently develop foam cell-rich vascular lesions that closely parallel early-stage human plaques 10. Larger models, including pigs and non-human primates, are capable of exhibiting advanced plaques characterized by distinct necrotic cores, calcification, and mature fibrous caps, thereby offering superior physiological parallels to human atherosclerosis 28,29.
Limitations in Modelling Atherosclerosis
Despite their undeniable utility, animal models harbor inherent translational limitations. Spontaneous plaque rupture and subsequent occlusive thrombosis remain exceedingly rare events in most non-human species. Fundamental discrepancies in lipoprotein metabolism, immunologic repertoire, and relatively abbreviated lifespans further constrain the fidelity of long-term disease modeling. Specifically, the complete absence of CETP expression in rodents fundamentally restricts their utility in reliably replicating human HDL biology. Consequently, rational model selection must be meticulously guided by the specific pathophysiological features and therapeutic mechanisms under active investigation to ensure maximal translational relevance 29.
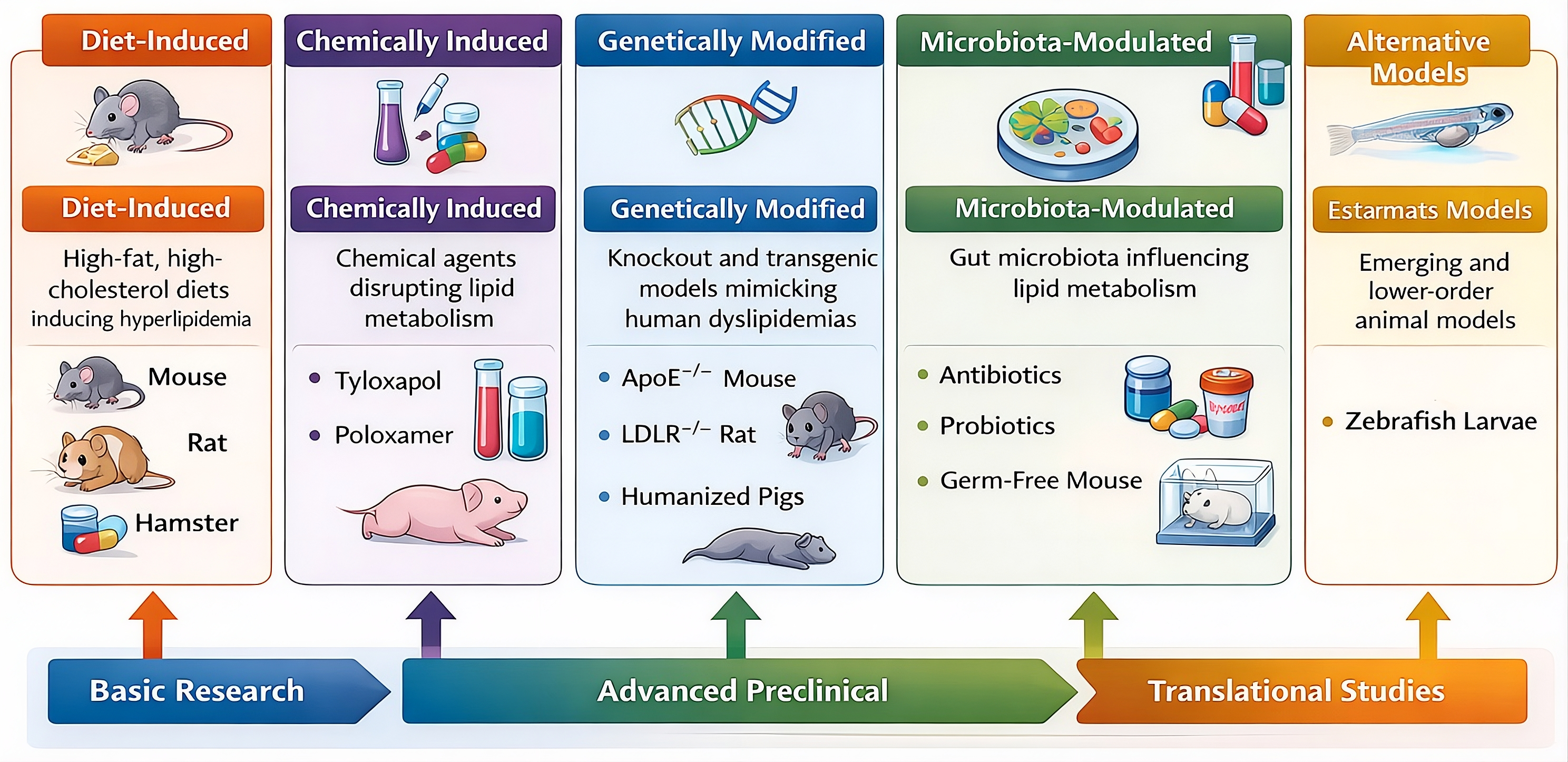
Classification of animal models utilized for antihyperlipidemic drug screening. The models are broadly categorized based on their induction methodologies and physiological characteristics, encompassing diet-induced, chemically induced, genetically modified, microbiota-modulated, and emerging alternative platforms.
Animal Models for Screening Antihyperlipidemic Agents
Animal models constitute the cornerstone of preclinical research for the discovery and evaluation of antihyperlipidemic agents. These models enable the rigorous assessment of lipid-lowering efficacy, mechanistic pathways, safety profiles, and pharmacodynamic responses prior to clinical translation 8,30. The selection of an appropriate model depends fundamentally on the specific research objective, disease complexity, and the metabolic pathway targeted by the therapeutic intervention. Based on the method of induction and genetic background, animal models of hyperlipidemia can be broadly categorized into diet-induced, chemically induced, genetically modified, microbiota-modulated, and emerging alternative models (Figure 2).
Diet-Induced Hyperlipidemia Models
Diet-induced models are among the most widely utilized systems for studying hyperlipidemia and its associated metabolic disorders. These models typically employ high-fat diets (HFD), high-cholesterol diets (HCD), or combinatorial diets enriched with saturated fats, sucrose, or fructose to induce dyslipidemia. Rodents fed an HFD or HCD exhibit elevated plasma triglycerides, total cholesterol, and LDL-C, alongside reduced HDL-C. These metabolic alterations are frequently accompanied by insulin resistance, hepatic steatosis, and low-grade systemic inflammation, closely mimicking the features of human metabolic syndrome 31. However, diet composition varies considerably across studies with respect to fat source, cholesterol content, and duration of feeding. Such heterogeneity introduces significant variability and complicates cross-study comparisons. Furthermore, rodents require supraphysiological dietary cholesterol to induce hypercholesterolemia, inherently limiting their ability to fully replicate human lipid metabolism 32.
Chemically Induced Hyperlipidemia Models
Chemical induction models utilize pharmacological agents to disrupt systemic lipid homeostasis. Triton WR-1339 (tyloxapol) is commonly employed to inhibit lipoprotein lipase activity, resulting in an acute elevation of plasma triglycerides and cholesterol. Similarly, poloxamer 407 induces hyperlipidemia through the dual inhibition of lipoprotein clearance and the upregulation of hepatic lipid synthesis 33. These models are particularly advantageous for the rapid, short-term screening of lipid-lowering compounds and for mechanistic studies involving triglyceride metabolism. However, chemically induced hyperlipidemia fails to replicate the chronic inflammatory and complex vascular features characteristic of human dyslipidemia. Consequently, their translational relevance for evaluating long-term cardiovascular outcomes remains highly limited 34,35.
Genetically Modified Animal Models
Genetically modified models provide invaluable insights into inherited dyslipidemias and the specific molecular pathways regulating lipid metabolism. Apolipoprotein E-deficient (ApoE⁻/⁻) and LDL receptor-deficient (LDLR⁻/⁻) mice are extensively utilized in atherosclerosis research due to their profound propensity to develop hypercholesterolemia and vascular lesions 36. ApoE⁻/⁻ mice develop spontaneous atherosclerosis even when maintained on standard chow diets, whereas LDLR⁻/⁻ mice require dietary cholesterol supplementation to accelerate lesion formation. Despite their robust utility, these models differ substantially from human lipid metabolism, particularly regarding lipoprotein distribution and the absence of CETP activity 37. Recent advances in CRISPR/Cas9 technology have enabled the generation of humanized models expressing human ApoB, PCSK9, or CETP, thereby significantly improving their translational relevance. Nonetheless, the high cost and technical complexity associated with generating and maintaining such models continue to limit their widespread adoption 38.
Microbiota-Modulated Models
Emerging evidence highlights the critical role of the gut microbiota in systemic lipid metabolism and cardiovascular risk. Interventions utilizing probiotics, prebiotics, and synbiotics have been shown to modulate lipid profiles via bile acid metabolism, short-chain fatty acid production, and the regulation of host gene expression 39,40. Animal models incorporating microbiota modulation, including antibiotic-treated or germ-free rodents, are increasingly used to elucidate complex host–microbe interactions in hyperlipidemia. While highly promising, such studies often exhibit significant methodological variability in microbial strain selection, dosing regimens, and intervention duration, which complicates reproducibility and translational interpretation 41. Therefore, critical appraisal of these studies is essential, particularly when sample sizes are restricted or appropriate control groups are lacking.
Alternative and Emerging Models
Zebrafish (Danio rerio) have gained considerable attention as alternative models for lipid research due to their optical transparency, genetic manipulability, and rapid lipid metabolism. Zebrafish larvae fed high-cholesterol diets exhibit pronounced lipid accumulation and vascular lipid deposition, providing an efficient platform for the high-throughput screening of prospective antihyperlipidemic compounds. However, fundamental evolutionary differences in cardiovascular anatomy, lipoprotein composition, and immunological responses severely limit their applicability for modeling advanced, complex atherosclerosis. Consequently, zebrafish models are optimally positioned for early-stage discovery and screening rather than for definitive translational studies 42.
Comparative Utility of Small and Large Animal Models
Animal models exhibit considerable variation regarding their physiological concordance with humans, associated ethical considerations, financial costs, and overall suitability for specific research objectives. Consequently, a rigorous comparative evaluation of small versus large animal models is imperative to ensure rational model selection 43,44.
Small Animal Models
Small animals—predominantly mice, rats, and hamsters—are extensively utilized due to their cost-effectiveness, ease of handling, rapid reproductive cycles, and the vast availability of genetic manipulation tools. These models are particularly advantageous for mechanistic investigations, target validation, and early-stage pharmacological screening 43. However, they possess substantial limitations. Notably, most rodents inherently lack CETP activity, exhibit disparate baseline lipoprotein distributions compared to humans, and rarely manifest spontaneous plaque rupture or occlusive thrombosis. Furthermore, a pervasive sex bias remains in preclinical research, as numerous studies are conducted exclusively on male cohorts, thereby significantly restricting the clinical generalizability of the findings (Figure 3).
Large Animal Models
Large animal models, including rabbits, pigs, and non-human primates, provide a superior physiological resemblance to human lipid metabolism, vascular architecture, and atherosclerotic disease progression. Rabbits naturally express CETP and readily develop cholesterol-enriched lipoproteins alongside foam cell-dense vascular lesions. Pigs and non-human primates are capable of developing complex, advanced atherosclerotic plaques characterized by mature fibrous caps and necrotic cores 44. Despite these distinct translational advantages, the utility of large animal models is frequently constrained by prohibitive costs, extended study durations, heightened ethical scrutiny, and restricted biological availability. Additionally, stringent regulatory requirements and substantial infrastructural demands further limit their routine implementation in lipid research (Figure 3) 38,44.
Strategic Integration of Models
Employing a tiered, strategic approach that integrates both small and large animal models substantially enhances the probability of translational success. Small animals are optimally suited for initial hypothesis generation, fundamental mechanistic exploration, and preliminary efficacy screening. Conversely, large animals are requisite for late-stage translational validation, comprehensive safety assessments, and precise dose optimization. A judicious, sequentially aligned combination of both model archetypes—tailored specifically to the given research phase (e.g., early discovery versus advanced preclinical validation)—ensures scientific robustness, ethical compliance, and maximal translational relevance (Figure 3) 45.
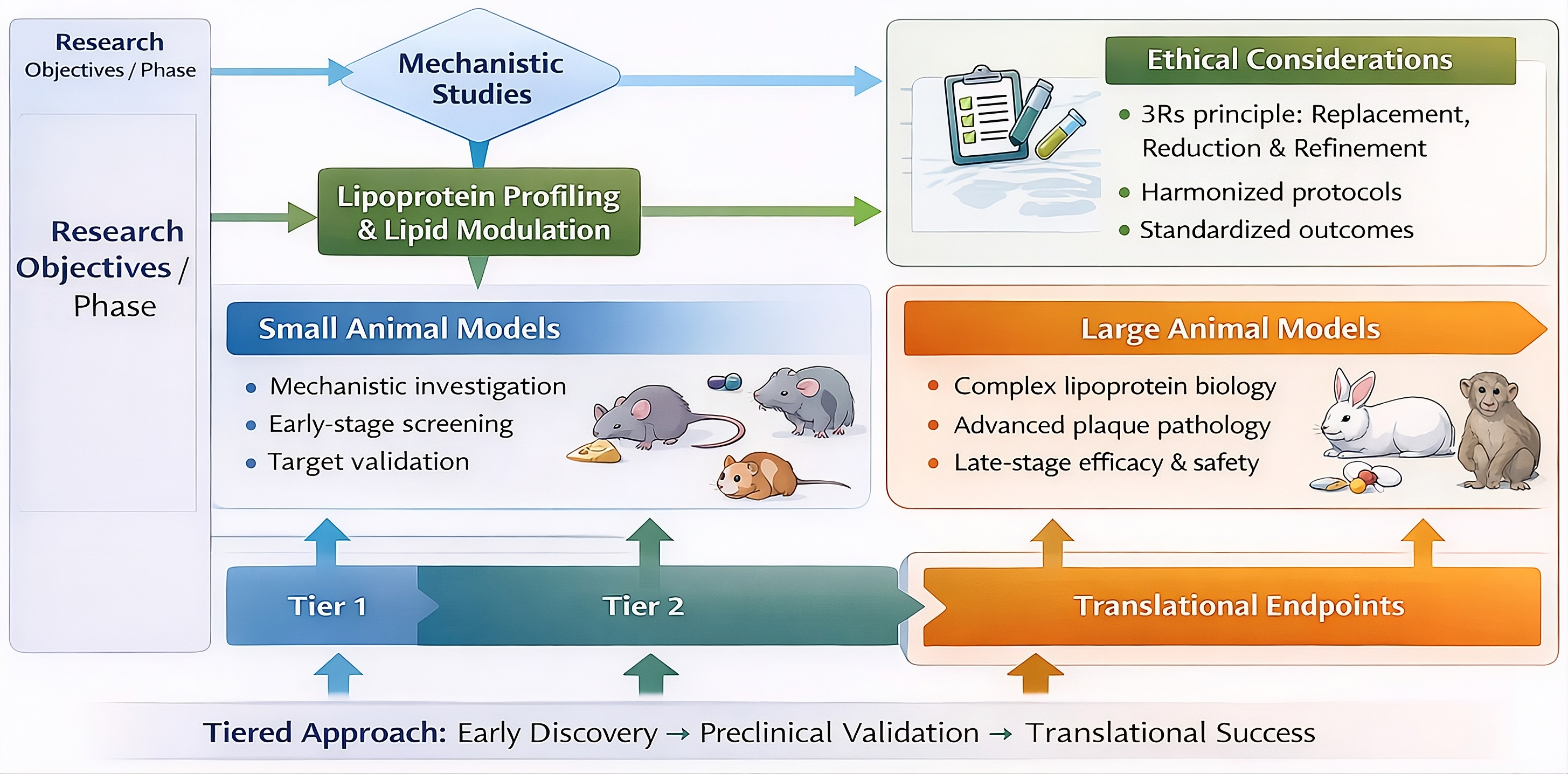
Translational framework linking animal model selection to research objectives. This tiered, decision-guided approach illustrates the strategic integration of small animal models for early-stage mechanistic investigation and target validation, followed by the deployment of large animal models for advanced preclinical efficacy, safety assessment, and translation to clinical endpoints.
Comparative Predictive Value and Cost–Benefit Considerations of Emerging versus Traditional Models
Emerging experimental platforms, such as zebrafish and microbiota-modulated models, offer complementary advantages to traditional rodent, rabbit, and large-animal systems; however, they differ substantially in their predictive value for late-stage clinical translation. Zebrafish models facilitate high-throughput, cost-effective screening and enable the rapid phenotypic assessment of lipid metabolism and vascular lipid deposition, rendering them particularly valuable for early-phase discovery and mechanistic exploration. Nevertheless, fundamental interspecies differences in cardiovascular anatomy, lipoprotein composition, and hemodynamics inherently limit their capacity to predict complex human clinical outcomes, such as spontaneous plaque rupture or long-term cardiovascular risk.
Microbiota-modulated models effectively capture host–microbe–drug interactions, which are increasingly recognized as critical determinants of systemic lipid metabolism and therapeutic responsiveness. While these models enhance the biological relevance of metabolic modulation studies, their translational predictability remains constrained by significant inter-laboratory variability, diet-dependent confounding effects, and overarching challenges in protocol standardization.
In contrast, traditional rodent and rabbit models—despite incurring higher costs and a greater ethical burden—provide more robust pharmacokinetic and pharmacodynamic relationships, aligning more closely with stringent regulatory expectations. Large-animal models, particularly porcine and non-human primate systems, offer the highest degree of translational fidelity, albeit at a substantially greater financial, infrastructural, and ethical cost. Collectively, these comparative considerations underscore that emerging models are optimally positioned as early-phase, hypothesis-generating tools, whereas traditional models remain indispensable for confirmatory efficacy testing and definitive translational validation within a structured, decision-guided drug development framework.
Critical Appraisal of Evidence from Animal Models
While animal models have substantially advanced our understanding of lipid metabolism and atherosclerosis, their predictive value for clinical outcomes varies considerably. Consequently, a critical appraisal of methodological rigor, biological relevance, and translational reliability is essential when interpreting preclinical findings (Table 4) 46.
Key Parameters Used in Evaluating Antihyperlipidemic Agents.
| Parameter Category | Specific Endpoints | Translational Importance |
|---|---|---|
| Biochemical | TC, LDL-C, HDL-C, TG | Primary efficacy |
| Histological | Plaque size, lipid deposition | Disease severity |
| Molecular | Inflammatory markers, gene expression | Mechanistic insight |
| Functional | Endothelial function | Cardiovascular relevance |
| Safety | Liver enzymes, body weight | Toxicity assessment |
Quality Grading of Evidence
The strength of evidence derived from animal studies was qualitatively graded based on experimental reproducibility, consistency across species, mechanistic plausibility, and alignment with human pathophysiology. Models demonstrating conserved lipid pathways, endogenous CETP activity, and plaque characteristics comparable to human atheromas were considered to provide strong evidence (++ evidence). Conversely, models primarily suited for mechanistic or exploratory studies were graded as providing moderate evidence (+ evidence). Rodent models, for instance, offer robust mechanistic insights but demonstrate limited translational accuracy for evaluating HDL-targeted therapies. In contrast, rabbit and non-human primate models provide a stronger predictive value for LDL-centric interventions, although their utility is frequently constrained by ethical and logistical considerations 47.
Methodological Limitations in Primary Studies
Many of the primary studies included in this review report positive lipid-lowering outcomes without adequately addressing underlying experimental limitations. Common methodological issues include small sample sizes, an absence of sex-based analyses, abbreviated intervention durations, and insufficient reporting of randomization or blinding procedures. These factors collectively inflate the risk of bias and can lead to the overestimation of therapeutic efficacy 48. Furthermore, in microbiota-modulation studies, profound heterogeneity in microbial strain selection, dosing regimens, and treatment duration further complicates data interpretation. The pervasive absence of standardized outcome measures across independent laboratories remains a major contributor to the irreproducibility often observed in lipid research.
Publication Bias and Reporting Gaps
Publication bias represents a significant yet frequently underappreciated limitation within preclinical lipid research. Studies that demonstrate positive or novel findings are disproportionately more likely to be published, whereas neutral or negative results often remain underreported. This inherent bias can profoundly distort the perceived efficacy of emerging antihyperlipidemic strategies, particularly those evaluated in genetically modified or novel dietary models 49. Selective outcome reporting, coupled with incomplete methodological descriptions, further diminishes study reproducibility. Greater adherence to standardized reporting frameworks, such as the ARRIVE guidelines, alongside the pre-registration of animal studies, could substantially improve the transparency and scientific credibility of the field.
Ethical Considerations and the 3Rs Principle
Ethical responsibility remains paramount in animal-based research, particularly concerning chronic metabolic disease models that necessitate prolonged dietary manipulation and invasive experimental procedures. The fundamental principles of Replacement, Reduction, and Refinement (3Rs) establish an essential ethical and scientific framework for rigorous model selection and study design 50,51.
Replacement Strategies
Whenever feasible, alternative methodologies—such as in vitro lipid assays, organ-on-a-chip platforms, computational modeling, and zebrafish larval systems—should be prioritized to replace higher-order animals during early-stage pharmacological screening. Integrating these alternative approaches substantially diminishes animal utilization while simultaneously facilitating rapid and efficient hypothesis testing 51.
Reduction of Animal Use
Rigorous statistical planning, the implementation of longitudinal study designs, and the utilization of shared control cohorts can significantly curtail the number of animals required without compromising statistical power or data integrity. Furthermore, the integration of non-invasive imaging modalities and the serial measurement of circulating biomarkers actively minimize the need for repeated animal sacrifice 52.
Refinement of Experimental Procedures
Refinement strategies are explicitly designed to minimize animal suffering and optimize overall welfare. These strategies encompass the provision of enriched housing conditions, the use of highly refined dietary formulations, the establishment of stringent humane endpoints, and the administration of robust analgesic protocols. Importantly, methodological refinement not only ensures ethical compliance but also bolsters data reliability by mitigating stress-induced metabolic and physiological variability 53. Collectively, strict ethical adherence and scientific rigor operate interdependently, ultimately reinforcing the translational validity of preclinical findings.
Translational Gaps, Challenges, and Future Directions
Despite decades of extensive preclinical research, the successful translation of antihyperlipidemic therapies from animal models to clinical efficacy remains inconsistent. Several translational gaps persist, underscoring the critical need for more predictive and integrative modeling strategies 54.
Failure of Promising Therapeutics
Numerous pharmacological agents that demonstrated robust efficacy in animal models have ultimately failed to achieve favorable clinical outcomes. CETP inhibitors represent a prominent example of this paradigm; substantial HDL elevation observed in preclinical and early-phase clinical studies did not consistently translate into a reduction in cardiovascular risk and, in certain instances, paradoxically increased adverse events 55. These clinical failures highlight the profound limitations of relying exclusively on biochemical surrogate endpoints—such as isolated alterations in LDL-C or HDL-C—without adequately evaluating plaque stability, systemic inflammation, or the long-term risk of clinical cardiovascular events.
Over-Reliance on Surrogate Endpoints
Preclinical investigations frequently prioritize the biochemical reduction of circulating lipids as their primary efficacy endpoint. However, the lowering of lipid levels does not invariably correlate with atherosclerotic plaque regression or a proportional decrease in clinical cardiovascular events. Integrating non-invasive vascular imaging, robust inflammatory biomarkers, and functional assessments of plaque stability into preclinical protocols may substantially improve translational accuracy 56.
Need for Standardization and Decision-Guided Model Selection
The pervasive lack of standardized experimental protocols governing diet composition, intervention duration, and specific endpoint measurements remains a major obstacle to ensuring cross-study comparability. Consequently, implementing a decision-tree framework that closely aligns specific research questions with the most appropriate animal models is essential. For example, mechanistic studies are optimally conducted using genetically modified rodents; lipoprotein profiling necessitates CETP-expressing species; and the comprehensive evaluation of plaque stability requires large-animal models. Such structured, evidence-based model selection fundamentally enhances both reproducibility and predictive value.
Future Directions
Future research in antihyperlipidemic pharmacology should prioritize the following key areas:
-
The development and utilization of humanized and multi-gene animal models.
-
The integration of multi-omics-based biomarkers to comprehensively profile metabolic responses.
-
The implementation of rigorously sex-inclusive experimental designs.
-
Strict ethical compliance with the 3Rs principles.
-
The adoption of multimodal endpoints that extend significantly beyond basic lipid quantification.
Collectively, these strategic approaches will support a more reliable, predictive, and ethically responsible translational pipeline (Table 5).
Comparative Translational Value of Common Animal Models.
| Model Category | Translational Value | Major Strength | Key Limitation | Ethical Consideration† |
|---|---|---|---|---|
| Rodents | Moderate | Genetic manipulability | Lack CETP | Low burden |
| Rabbits | High | CETP expression | Cost | Moderate |
| Pigs | Very High | Plaque similarity to humans | Infrastructure demands | High |
| Primates | Highest | Human-like lipid metabolism | Ethical constraints, cost | Very High |
| Zebrafish | Low–Moderate | High-throughput screening | Limited cardiovascular anatomy | Minimal |
Conclusion
Animal models remain indispensable tools for advancing antihyperlipidemic drug discovery and understanding the complex biology of lipid metabolism and atherosclerosis. This integrative review highlights the strengths and limitations of commonly used animal models, ranging from diet-induced and chemically induced systems to genetically modified, microbiota-modulated, and large-animal models. Each category offers distinct advantages depending on the specific research objective, disease stage, and therapeutic mechanism under investigation.
While small animal models provide cost-effective and mechanistically informative platforms for early-stage screening, their limited ability to replicate human lipoprotein profiles and plaque complexity restricts direct clinical extrapolation. In contrast, large animal models demonstrate superior translational relevance but are constrained by ethical, logistical, and financial considerations. Importantly, repeated translational failures, including those observed with CETP inhibitors, underscore the limitations of relying solely on lipid-centric surrogate endpoints.
Future progress in antihyperlipidemic research will depend on rational, question-driven model selection, improved methodological standardization, incorporation of sex-inclusive designs, and integration of multi-omics and imaging-based endpoints. Adherence to ethical principles, particularly the 3Rs, must remain central to preclinical study design. Collectively, these strategies will enhance reproducibility, predictive accuracy, and the successful translation of preclinical findings into clinically meaningful cardiovascular therapies.
Abbreviations
3Rs: Replacement, Reduction, and Refinement; ACAT: Acyl-CoA cholesterol acyltransferase; ApoA-I: Apolipoprotein A-I; ApoB-100: Apolipoprotein B-100; ApoE: Apolipoprotein E; ARRIVE: Animal Research: Reporting of In Vivo Experiments; CETP: Cholesteryl ester transfer protein; CRISPR: Clustered Regularly Interspaced Short Palindromic Repeats; CVDs: Cardiovascular diseases; HCD: High-cholesterol diets; HDL: High-density lipoprotein; HDL-C: High-density lipoprotein cholesterol; HFD: High-fat diets; ICAM-1: Intercellular Adhesion Molecule 1; ICH: International Council for Harmonisation; IDL: Intermediate-density lipoproteins; IL-1β: Interleukin-1 beta; LCAT: Lecithin-cholesterol acyltransferase; LDL: Low-density lipoprotein; LDL-C: Low-density lipoprotein cholesterol; LDLR: Low-density lipoprotein receptor; LPL: Lipoprotein lipase; MCP-1: Monocyte chemoattractant protein-1; MeSH: Medical Subject Headings; OECD: Organisation for Economic Co-operation and Development; PCSK9: Proprotein convertase subtilisin/kexin type 9; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; TC: Total cholesterol; TG: Triglycerides; TNF-α: Tumor necrosis factor-alpha; VCAM-1: Vascular cell adhesion molecule 1; VLDL: Very-low-density lipoprotein; and WHHL: Watanabe heritable hyperlipidemic.
Acknowledgments
The authors would like to thank the Hon. Chancellor, Pro-Chancellor, Vice-Chancellor, Head of the Department of Pharmacy, and the Dean, Faculty of Pharmacy, Integral University, for providing the necessary facilities to complete this work. The University assigned a unique manuscript communication number (IU/R&D/2024-MCN0002443).
Author’s contributions
JAA designed the structure of the manuscript and drafted the initial version. MK performed the literature review. SMH edited and critically reviewed the manuscript. FA formatted the manuscript according to journal guidelines. JA, SA, and NP contributed to data curation. All authors read and approved the final manuscript.
Funding
None.
Availability of data and materials
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Declaration of generative AI and AI-assisted technologies in the writing process
The authors declare that generative AI and AI-assisted technologies were not used in the writing of this manuscript prior to submission, except for minor language editing and grammatical corrections.
Competing interests
The authors declare that they have no competing interests.

