Advances in research on ferroptosis in jaw bone augmentation
- 22 Wenchang West Road, Higher Education Park, Wuhu City, Anhui Province, China
- Department of Stomatology, Yijishan Hospital, Wannan Medical College, Wuhu, Anhui 241000, China
Abstract
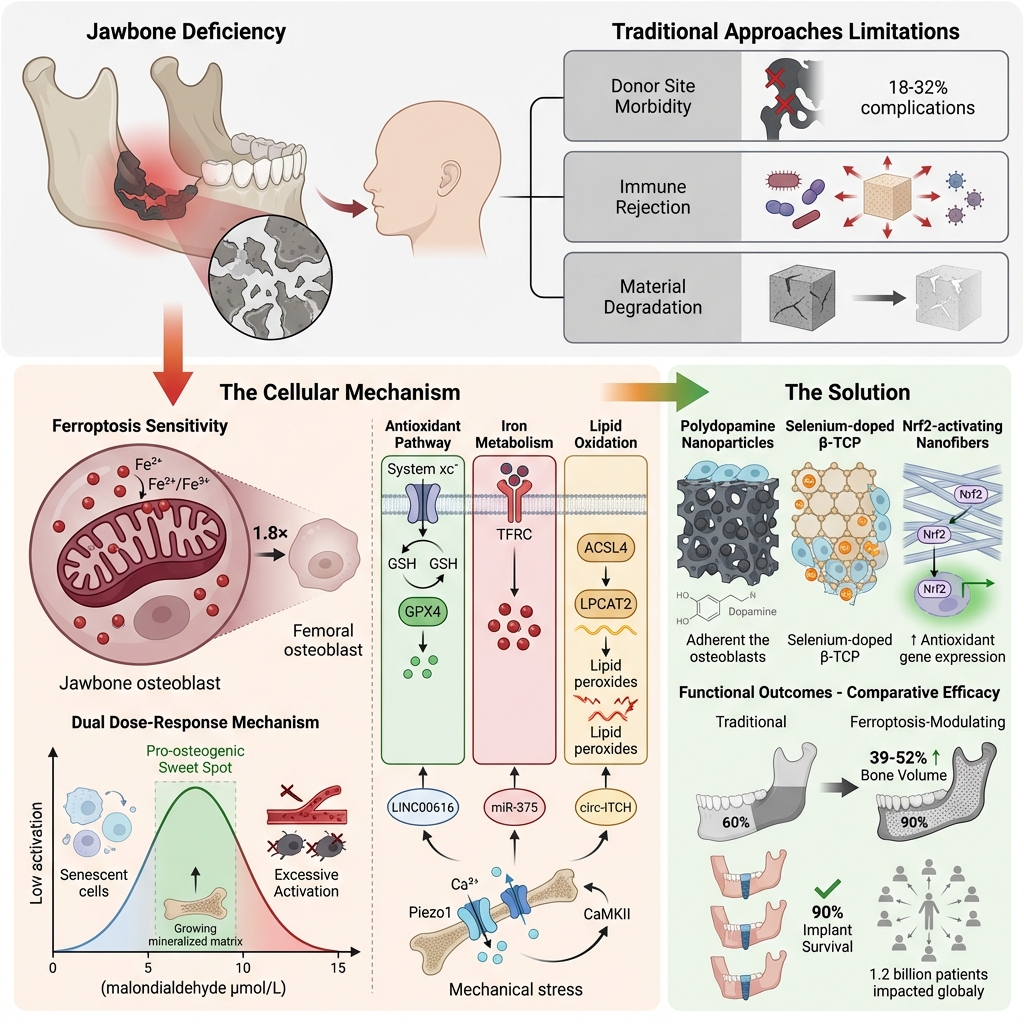
Ferroptosis, an iron-dependent form of regulated cell death (RCD) driven by lipid peroxidation, has emerged as a critical regulatory mechanism in jawbone augmentation, which addresses a major barrier to successful dental implants: insufficient jawbone volume. This review summarizes the molecular basis of ferroptosis and jaw-specific features that regulate it (high mitochondrial density, PUFA enrichment, and GPX4 dependency in jawbone osteoblasts), with a focus on core regulatory networks (namely, the system xc⁻/GSH/GPX4 axis, lipid metabolism pathways, Nrf2/p53 transcriptional regulation, and non-coding RNAs). We highlight that jawbone osteoblasts are more sensitive to ferroptosis than to other RCD pathways (apoptosis, necroptosis, pyroptosis) due to their intrinsic biological properties. Importantly, modulation of ferroptosis exhibits substantial translational value in bone-related diseases, including osteoporosis (through alleviating osteoblast loss) and bone defect repair (via optimizing regenerative niche formation). We analyze ferroptosis's context-dependent dual effects—moderate activation promotes regeneration by clearing senescent cells and modulating inflammation, while excessive activation impairs functional cells and vascularization—and position these effects as the central theme. Preclinical data validate ferroptosis-modulating biomaterials (e.g., PDA/β-TCP scaffolds, tFNA, selenium-doped substitutes) as effective strategies to overcome limitations of traditional techniques (donor site morbidity, microbial rejection, degradation-osteogenesis mismatch). Finally, we discuss clinical translation priorities and challenges, highlighting ferroptosis as a promising target for optimizing jawbone augmentation and thereby improving dental implant success.
Introduction
Jawbone augmentation is a core procedure in oral and maxillofacial surgery, aimed at enhancing jawbone volume for dental implant placement, orthognathic surgery, and prosthetic rehabilitation. With global population aging and rising tooth loss rates—the World Health Organization estimates that 1.2 billion people worldwide suffer from partial edentulism1—the clinical demand for this procedure is increasing annually, at a rate of 15% per year in China alone, according to the 2025 China Stomatological Association White Paper2. Despite advancements in traditional techniques (autologous or allogeneic bone grafts, synthetic biomaterials such as β-tricalcium phosphate [β-TCP], hydroxyapatite [HA]), three jaw-specific challenges persist:
-
Donor site morbidity: Autologous bone grafts (e.g., mandibular ramus, iliac crest) are associated with complications at 18–32% prevalence, including nerve injury, persistent pain, and aesthetic defects3,4;
-
Microbially induced immune rejection: The oral cavity harbors over 700 bacterial species, with P. gingivalis-derived lipopolysaccharide (LPS) able to trigger excessive inflammation and thereby reducing graft survival5,6,7;
-
Material degradation–osteogenesis mismatch: Resorption of β-TCP in the maxillary sinus proceeds faster than new bone formation, leading to graft volume loss in some cases8,9,10.
Notably, the jawbone serves as a biomechanically active interface between the oral mucosa and the skeletal system, being continuously exposed to masticatory stress11 and microbial insults. These stimuli dysregulate ferroptosis in jaw-specific cells (jawbone osteoblasts, periodontal ligament stem cells [PDLSCs], osteocytes), impairing bone regeneration—an understudied mechanism compared to that of systemic bone biology (e.g., femoral or tibial bone)12,13,14. For non-specialist readers, ferroptosis is defined by three core molecular features: 1) GPX4, which serves as the central inhibitor, being a selenium-dependent enzyme that scavenges lipid peroxides to prevent cell death; 2) SLC7A11-mediated redox balance, wherein the cystine/glutamate antiporter provides cystine for glutathione (GSH) synthesis, a cofactor for GPX415,16,17,18; 3) lipid ROS as the key effector, wherein iron-catalyzed lipid peroxidation (via the Fenton reaction) generates reactive oxygen species (ROS) that disrupt cell membranes19,20. Jawbone osteoblasts exhibit distinct intrinsic traits—higher mitochondrial density, PUFA-enriched membranes, and strict GPX4 dependency—that render them more sensitive to ferroptosis than to other forms of regulated cell death (RCD) pathways11,15,21. Ferroptosis, first defined by Dixon et al. in 201220, is distinct from apoptosis (nuclear condensation) and necrosis (organelle swelling) based on its morphological features (mitochondrial shrinkage, intact cell membrane)20,22. Crucially, ferroptosis exerts a context-dependent dual role in jawbone augmentation—moderate activation promotes regeneration, while excessive activation causes damage—driven by jaw-specific regulatory mechanisms23. This review, based on a systematic literature search, focuses on this dual role and its underlying mechanisms, providing a theoretical basis for optimizing clinical strategies to treat jawbone augmentation, osteoporosis, and bone defects.
Literature Search Methodology
To ensure the comprehensiveness, transparency, and reproducibility of this review, the literature search and selection process followed standardized guidelines for narrative reviews:
Databases and Search Strategy
The following databases were searched: PubMed, Web of Science Core Collection, Embase, and CNKI (China National Knowledge Infrastructure) to cover both English- and Chinese-language literature.
Search string: A Boolean combination of keywords: ("ferroptosis" OR "iron-dependent cell death") AND ("jawbone" OR "alveolar bone" OR "maxillofacial bone" OR "dental implants" OR "periodontitis" OR "jawbone defect" OR "oral and maxillofacial surgery") AND ("osteoblasts" OR "bone marrow mesenchymal stem cells" OR "periodontal ligament stem cells" OR "biomaterials" OR "bone regeneration").
Time frame: Literature published from January 2012 (the year in which ferroptosis was formally defined) to October 2025, and priority was given to studies published during the past 5 years (2020–2025) to highlight recent advances.
Inclusion and Exclusion Criteria
Inclusion criteria: 1) Original research (in vitro, in vivo, or clinical) or comprehensive reviews focusing on ferroptosis in jawbone-related tissues (jawbone, alveolar bone, periodontal ligament stem cells [PDLSCs], peri-implant tissues); 2) Studies investigating the molecular mechanisms, regulatory factors, or the biomaterial-based modulation of ferroptosis for jawbone augmentation or repair; 3) Articles in English or Chinese, with available full-text.
Exclusion criteria: 1) Studies solely focusing on systemic bone tissues (e.g., femur, tibia) lacking oral relevance; 2) Case reports, letters to the editor, or conference abstracts lacking full data; 3) Studies with an unclear experimental design or unvalidated results.
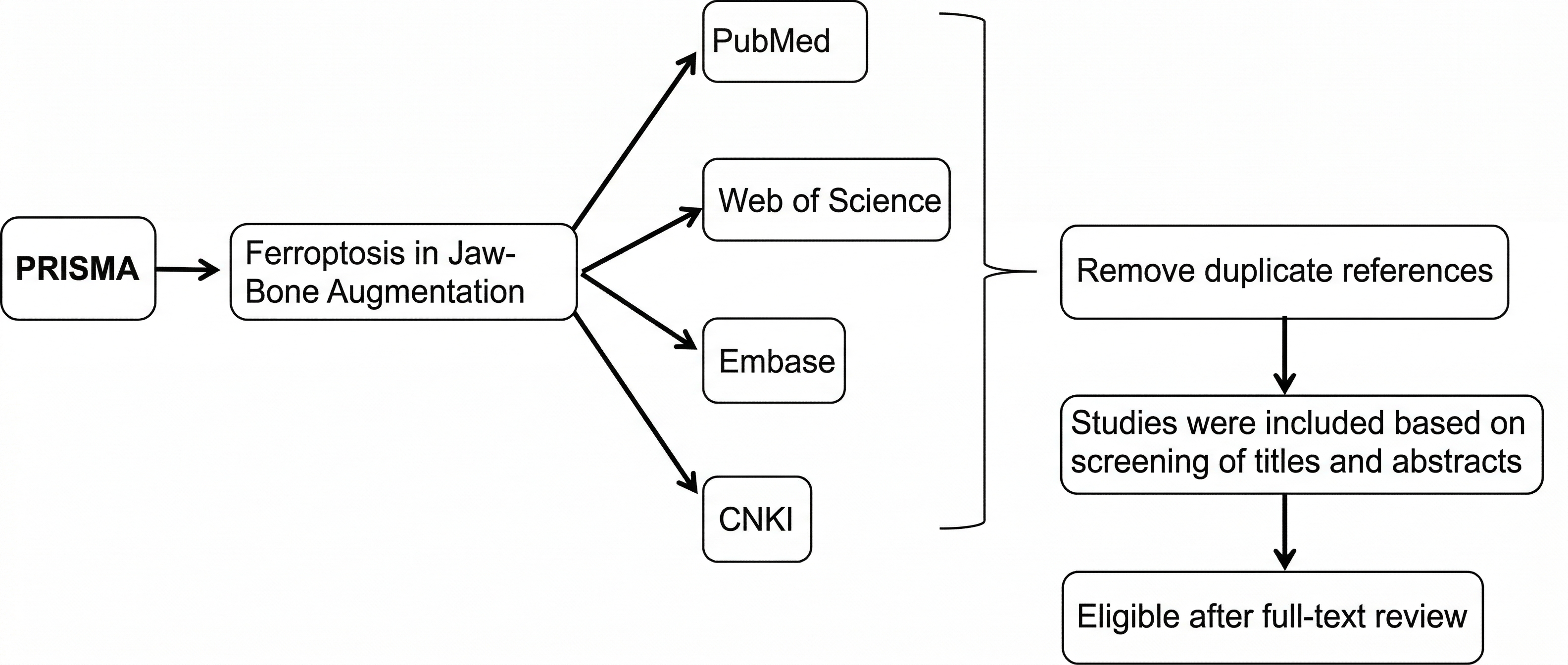
PRISMA flow diagram for the literature search and study selection of ferroptosis in jaw-bone augmentation.
Basic Molecular Mechanisms of Ferroptosis (Brief Overview)
Ferroptosis is driven by iron-dependent lipid peroxidation, distinct from other RCDs (apoptosis, pyroptosis, necroptosis)19,24. Its core molecular network revolves around three interconnected axes, which form the basis for its dual role in jawbone augmentation:
Core Regulatory Axes
The System xc⁻-GSH-GPX4 Axis: System xc⁻ (composed of SLC7A11 and SLC3A2) mediates cystine uptake, which is converted to cysteine for GSH synthesis25. GPX4 (selenium-dependent) uses GSH to scavenge lipid peroxides, representing the final enzymatic step in preventing ferroptosis26,27. Downregulation of SLC7A11 or GPX4 is a key trigger for excessive ferroptosis28.
The Iron Metabolism Axis: Intracellular iron homeostasis is maintained by the uptake (TFRC), storage (ferritin), and export (ferroportin) mechanisms29. Iron overload (e.g., TFRC upregulation, ferritin degradation) increases the labile iron pool (LIP), amplifying the Fenton reaction (Fe²⁺ + H₂O₂ → Fe³⁺ + ·OH + OH⁻) and thereby promoting lipid ROS production30,31.
The Lipid Metabolism Axis: Polyunsaturated fatty acids (PUFAs) are the primary substrates for lipid peroxidation. Enzymes such as ACSL4 (PUFA activation), LPCAT2 (PUFA incorporation into phospholipids), and LOX (PUFA oxidation) promote ferroptosis, while ACSL3 (saturated/monounsaturated fatty acid activation) counteracts it15,32.
Key Regulators
Suppressors include GPX4, SLC7A11, Nrf2 (a transcriptional activator of antioxidant genes)33, and FSP1 (a GPX4-independent ferroptosis inhibitor)34. Promoters include ACSL4, TFRC, p53 (under pathological conditions), 15-LOX, and PLA2 (PUFA release).
This simplified framework provides the foundation for understanding the ferroptosis's dual effects in jawbone augmentation, which are further shaped by jaw-specific regulatory features (detailed in Section 5).
The Dual Role of Ferroptosis in Jawbone Augmentation (Central Narrative)
Ferroptosis exerts context-dependent effects on jawbone regeneration, closely tied to activation intensity, cell type (senescent vs. functional cells), and microenvironment (infection, mechanical stress). This dual role is the core of ferroptosis-based jawbone augmentation strategies.
Beneficial Effects of Moderate Ferroptosis Activation
Moderate ferroptosis creates a “regenerative niche” for BMSC proliferation and osteogenesis by targeting harmful cells and modulating inflammation, thus addressing key barriers in aged or infected jawbone defects.
Senescent Cell Clearance
Senescent cells secrete a senescence-associated secretory phenotype of factors, such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α), which inhibit the osteogenic differentiation of bone marrow mesenchymal stem cells and impair angiogenesis. The underlying mechanism involves a markedly higher iron accumulation in senescent jawbone osteoblasts compared to their non-senescent counterparts, coupled with reduced GPX4 activity, thereby rendering them selectively vulnerable to ferroptosis 35. Preclinical evidence from a rat model of aged jawbone defects demonstrates that low-dose erastin can reduce the senescent cell population and thereby increase new bone volume 36. Notably, this clearance is cell-type-specific—functional BMSCs and endothelial cells are spared due to intact GPX4 expression.
Inflammation Modulation
Oral infections (e.g., P. gingivalis-induced periodontitis) trigger excessive infiltration by M1 macrophages, which secrete pro-inflammatory cytokines (TNF-α, IL-1β) that impair graft integration. The mechanism is as follows: Moderate ferroptosis in osteoblasts releases damage-associated molecular patterns (DAMPs, e.g., HMGB1) that induce M1 macrophage ferroptosis (via the CaSR/AKT/β-catenin pathway) and promote M2 polarization 37. M2 macrophages secrete TGF-β and BMP2, thus enhancing BMSC osteogenesis and vascularization. Preclinical case studies: in P. gingivalis-infected jawbone defects, co-treatment with low-dose Fer-1 (1 μmol/L) and erastin (0.2 μmol/L) reduces M1 macrophages, increases M2 macrophages, and improves graft survival 38,39.
Biomarker of Regeneration Efficacy
Moderate ferroptosis correlates with favorable clinical outcomes, serving as a potential prognostic marker. A clinical study in 50 patients undergoing maxillary sinus floor lift augmentation showed that serum MDA (lipid peroxidation marker) levels of 5–8 μmol/L (moderate ferroptosis) were associated with 90% implant survival at 1 year, versus 65% for MDA <3 μmol/L (insufficient ferroptosis) or >12 μmol/L (excessive ferroptosis) 40. Preclinical validation: aldehyde dehydrogenase 2 (ALDH2) activates Nrf2 to restrict ferroptosis within the moderate range, reducing periodontitis-related inflammation and promoting PDLSC osteogenic differentiation 41.
Detrimental Effects of Excessive Ferroptosis Activation
Excessive ferroptosis, triggered by jaw-specific stressors (microbial infection, implant overload, and biomaterial mismatch), damages functional cells and disrupts regeneration, thereby exacerbating the limitations of traditional augmentation techniques.
Loss of Functional Osteoblasts/BMSCs
Osteoblasts and BMSCs constitute the primary cell types responsible for jawbone regeneration. With microbial induction, P. gingivalis LPS upregulates TFRC and ACSL4 via the TLR4/NF-κB pathway, thereby inducing ferroptosis in jawbone BMSCs, which reduces ALP activity and mineralization 30,42. With mechanical induction, implant overload activates the YAP-TEAD complex to inhibit GPX4 transcription, thereby inducing ferroptosis in peri-implant osteoblasts and reducing bone-implant contact 11. This cell loss directly impairs new bone formation.
Impairment of Vascularization
Endothelial cells play a critical role in providing nutrient delivery to and graft integration, but they remain highly vulnerable to ferroptosis owing to their high PUFA content in their membranes. The underlying mechanism involves accumulation of excessive lipid peroxides, which disrupt endothelial cell membranes, thereby reducing tube formation and VEGF expression 43. In animal models of rat maxillary sinus lift, excessive ferroptosis (induced by 5 μmol/L erastin) reduces vascular density and new bone formation 43, representing a key contributor to “volume collapse” in traditional β-TCP grafts.
Clinical Correlates to Poor Outcomes
In patients with periodontitis, those presenting with severe disease exhibit reduced serum glutathione peroxidase 4 (GPX4) activity and undergo increased alveolar bone resorption 12,44. In individuals with diabetes and jaw defects, they show elevated serum levels of malondialdehyde (MDA), with a corresponding increase in the failure rate of maxillary sinus augmentation procedures, unlike what is typically seen in non-diabetic patients 45,46. Similarly, in elderly patients with osteoporosis, the jawbone demonstrates increased susceptibility to ferroptosis, which correlates with decreased bone mineral density 45. Preclinical interventions confirm that inhibiting excessive ferroptosis (e.g., via Nrf2 activation, or GPX4 stabilization) reverses these adverse outcomes 47,48.
Jaw-Specific Regulatory Mechanisms (Why the Dual Role Exists)
The dual role of ferroptosis in jawbone augmentation is not random but shaped by unique anatomical, physiological, and microenvironmental features of the jawbone. These jaw-specific traits amplify ferroptosis susceptibility under stress while enabling controlled activation for regeneration:
Mitochondrial and Metabolic Specificity
Jawbone osteoblasts exhibit a 1.8-fold higher mitochondrial density than do femoral osteoblasts11,21, which adapts them to the high energy demands of masticatory stress. This increases polyunsaturated fatty acid (PUFA) content in mitochondrial membranes, making them preferred substrates for lipid peroxidation; this explains why jawbone cells are more sensitive to ferroptosis triggers15,49.
Enhanced ROS Production: Masticatory stress activates mitochondrial complex III, thereby increasing mtROS11. mtROS reduces Fe³⁺ to Fe²⁺, amplifying the Fenton reaction—this “ROS burst” can induce moderate ferroptosis for senescent cell clearance but triggers excessive ferroptosis under prolonged stress50.
Oral Microenvironment Specificity
The oral cavity’s unique microbial ecosystem directly modulates ferroptosis:
P. gingivalis-mediated regulation: P. gingivalis LPS activates TLR4/NF-κB to upregulate ACSL4 and downregulate GPX412, while its gingipains cleave GPX431—both mechanisms synergistically promote excessive ferroptosis. Conversely, short-term LPS exposure activates Nrf2 as an adaptive response, limiting ferroptosis to moderate levels12,51.
Quorum sensing by Fusobacterium nucleatum: F. nucleatum secretes autoinducer-2 (AI-2), which upregulates TFRC via the LuxS/AI-2 pathway; this increases iron uptake and fine-tunes ferroptosis activation52,53.
Mechanical Stress Specificity
The jawbone endures continuous masticatory stress and implant-induced occlusal force, which modulates ferroptosis in a dose-dependent manner:
Moderate stress activates the Piezo1/Ca²⁺/CaMKII pathway, thereby upregulating Nrf2, and maintaining GPX4 expression while limiting ferroptosis to beneficial levels11,54.
Excessive stress activates the YAP-TEAD complex, which binds to the GPX4 promoter, reducing GPX4 levels and inducing excessive ferroptosis11—a key mechanism of implant failure.
Tissue-Specific GPX4 Regulation
GPX4 serves as the core ferroptosis inhibitor, with jaw-specific expression and regulation:
High basal expression: Jawbone osteoblasts express higher levels of GPX4 than femoral osteoblasts, providing inherent protection against spontaneous ferroptosis21. GPX4 knockout leads to 80% mortality of jawbone osteoblasts within 72 hours26.
Selenium dependency: Jawbone tissues exhibit higher selenium demand55, as selenium is a GPX4 cofactor. Selenium deficiency (common in elderly edentulous patients) reduces GPX4 activity and increases ferroptosis55—thereby explaining age-related jawbone loss.
Emerging Regulators: Non-Coding RNAs (ncRNAs)
In addition to protein-coding regulators, ncRNAs play critical roles in fine-tuning ferroptosis’ dual role in jawbone augmentation—acting as “molecular switches” to maintain ferroptosis within beneficial levels. These ncRNAs exhibit jaw-specific expression patterns and modulate ferroptosis via ceRNA networks or direct protein binding:
lncRNAs
The lncRNA LINC00616 is highly expressed in periodontitis-associated jawbone tissues 30, and it sponges miR-370 to upregulate TFRC, promoting excessive ferroptosis in PDLSCs. Knockdown of LINC00616 in rat periodontitis models reduces alveolar bone loss and restores PDLSC osteogenesis 34. The lncRNA Linc01133 acts as a ceRNA to sequester miR-30c, upregulating BGLAP (osteocalcin) and inhibiting ferroptosis via SLC7A11 upregulation, thereby enhancing hPDLSC osteogenic differentiation 56.
circRNAs
circ-ITCH is downregulated in diabetic jawbone defects, and it recruits TAF15 to stabilize Nrf2 mRNA, activating the Nrf2/GPX4 pathway. circ-ITCH-containing BMSC exosomes alleviate high glucose-induced HUVEC ferroptosis, boosting angiogenesis and accelerating diabetic jawbone regeneration 57. circ-Snhg11 sponges miR-144-3p to activate SLC7A11/GPX4, inhibiting excessive ferroptosis and promoting jawbone regeneration and angiogenesis 58.
miRNAs
miR-375 is upregulated in implant overload-induced jawbone defects 59; it targets the Nrf2 3’-UTR to reduce Nrf2 expression, downregulating GPX4/SLC7A11 and inducing excessive ferroptosis. AntagomiR-375 treatment increases bone-implant contact 59.
Mechanistic Role of Ferroptosis-Related ncRNAs and Key Molecules in Jaw Bone Growth
| Key Molecule/ncRNA | Molecular Function | Role in Dual Effects | Corresponding References (Author et al., Year; Ref. No.) |
|---|---|---|---|
| GPX4 | Core ferroptosis inhibitor; scavenges lipid peroxides | Limits excessive ferroptosis; maintains beneficial activation | Ingold I et al., 2018; |
| Nrf2 Pathway | Master antioxidant regulator; upregulates GPX4/SLC7A11 | Suppresses excessive ferroptosis; enables moderate activation | Qiao S et al., 2023; |
| p53 Pathway | Dual regulator; represses SLC7A11 (pathological); upregulates GPX4 (physiological) | Triggers excessive ferroptosis (aging/infection); limits it (healing) | Qu X et al., 2021; |
| SLC7A11 | Cystine uptake for GSH synthesis; inhibits ferroptosis | Prevents excessive ferroptosis | Qiao S et al., 2023; |
| TFRC | Iron uptake; promotes ferroptosis | Induces excessive ferroptosis (overexpression); modulates moderate activation (physiological) | Wang H et al., 2022; |
| lncRNA LINC00616 | Sponges miR-370; upregulates TFRC | Promotes excessive ferroptosis (periodontitis) | Wang H et al., 2022; |
| circ-ITCH | Stabilizes Nrf2 mRNA; activates Nrf2/GPX4 | Inhibits excessive ferroptosis (diabetic defects) | Chen J et al., 2023; |
| miR-375 | Targets Nrf2; downregulates GPX4/SLC7A11 | Promotes excessive ferroptosis (implant overload) | Guo X et al., 2025; |
Biomaterial-targeted ferroptosis modulation for jawbone augmentation
Biomaterial design for jawbone augmentation must account for jaw-specific stressors and ferroptosis’ dual role—tailoring formulations to inhibit excessive ferroptosis while preserving beneficial activation. Below are validated ferroptosis-modulating biomaterials with preclinical efficacy:
Polydopamine Nanoparticle (PDA NP)-Modified Scaffolds
Polydopamine nanoparticles (PDA NPs; 50–80 nm diameter), which match jaw defect scaffold pores (100–200 μm), integrate antioxidant, iron-chelating, and antimicrobial properties, addressing microbial-induced excessive ferroptosis. These PDA NPs (50–80 nm in diameter) are an ideal material for scaffold modification due to their unique chemical properties. The catechol groups in their structure scavenge reactive oxygen species (ROS) and chelate excess iron ions, inhibiting lipid peroxidation chain reactions upstream. They upregulate intracellular GPX4 protein expression, enhancing cellular antioxidant defenses. In a diabetic jawbone defect model, a curcumin-loaded PDA system alleviated oxidative stress in the hyperglycemic environment and promoted osteogenic differentiation of jawbone-derived mesenchymal stem cells through the Wnt/β-catenin signaling pathway, as evidenced by elevated alkaline phosphatase (ALP) activity and mineralized nodule formation43,55,63,64,65.
Tetrahedral Framework Nucleic Acids (tFNA)
Tetrahedral framework nucleic acids (tFNA; 10–15 nm DNA nanoparticles) are tailored for diabetic jawbone defects, where mitochondrial dysfunction exacerbates excessive ferroptosis. These nanostructures, formed by DNA self-assembly, exhibit excellent cell membrane penetration. They promote nuclear translocation of Nrf2, activating the antioxidant response element (ARE) and upregulating enzymes such as heme oxygenase-1 (HO-1) and NAD(P)H:quinone oxidoreductase 1 (NQO1). In oxidative stress models, tFNA treatment preserves mitochondrial membrane potential stability and elevates ATP production. Modification with M2 macrophage-derived exosomes enhances cellular uptake efficiency, with no significant immune response observed in in vitro or in vivo experiments66,67.
Selenium-Doped Bone Substitutes (PDT-TCP-SE)
These address selenium deficiency in elderly jawbone defects, a key contributor to GPX4 dysfunction and excessive ferroptosis. Incorporation of selenium, an essential component of the glutathione peroxidase 4 (GPX4) enzyme, enhances a material's antioxidant properties. Selenium-doped β-tricalcium phosphate (β-TCP) enables sustained selenium ion release during degradation. This directly activates GPX4 and synergizes antioxidant effects through the Sirt1/Nrf2 pathway. Its compressive strength is comparable to that of cancellous bone. In a rat calvarial defect model, it promoted greater new bone volume than regular β-TCP, with elevated vascular density55.
Epimedium Exosome-Loaded GelMA Hydrogel
Epimedium-derived exosomes contain microRNA-21-5p (miR-21-5p), which inhibits ferroptosis by targeting the Phosphatase and Tensin Homolog (PTEN), a phosphatase that suppresses the Phosphatidylinositol 3-kinase (PI3K)/Akt pathway.
Mechanism: E-exosomes increase Akt phosphorylation, activating Nuclear Factor Erythroid 2-Related Factor 2 (Nrf2) and upregulating Glutathione Peroxidase 4 (GPX4)68.
Preclinical Efficacy: E-exosome-loaded gelatin methacryloyl (GelMA) hydrogels reduced alveolar bone loss and enhanced osteogenic differentiation of periodontal ligament stem cells in a rat periodontitis model69.
pH-Responsive MgO₂ Scaffolds
Magnesium peroxide-based scaffolds dynamically regulate iron release and ferroptosis in the acidic oral microenvironment. pH-Dependent Release: Under acidic conditions, MgO₂ decomposes to release Mg²⁺ and H₂O₂. Mg²⁺ promotes osteogenesis in bone marrow mesenchymal stem cells by activating the Wnt3a/glycogen synthase kinase-3β (GSK-3β)/β-catenin pathway, while a low dose of H₂O₂ induces moderate ferroptosis to clear senescent cells 68. Preclinical Efficacy: The pH-responsive MgO₂ scaffold increased the bone volume fraction in a maxillary sinus augmentation model 68, a finding consistent with the results reported by Liu et al. regarding an osteoimmunomodulatory biopatch 70.
Applications of ferroptosis modulation in bone tissue engineering
| Functional Direction | Material Name | Core Function | Preclinical Data (Animal Model, Outcome) | Corresponding References (Author, Year) |
|---|---|---|---|---|
| Enhancing cell adhesion/proliferation | Selenium-Doped Bone Substitutes (PDT-TCP-SE) | Supports cell adhesion; injectable/adhesive; activates Sirt1/Nrf2/GPX4 | Osteoporotic rat jaw defects: BV/TV +52%, GPX4 activity +60% | Huang L et al. 2024 |
| Epimedium Exosome-Loaded GelMA Hydrogel | Recruits PDLSCs; enhances adhesion via PI3K/Akt pathway | Rat periodontitis: Alveolar bone loss -42%, PDLSC osteogenesis +50% | Hu W et al. 2025 | |
| Alleviating ferroptosis injury | PDA NP-Modified PLGA/β-TCP Scaffolds | Scavenges >85% LPS-induced ROS; inhibits GPX4 ubiquitination | CD-1 mouse calvarial defect (4 mm, 6-8 w): BV/TV ↑39.2% (p<0.01), osseointegration rate 81% | Xu S et al. 2023 |
| tFNA-Modified HA Scaffolds | Activates Nrf2/GPX4; improves mitochondrial metabolism | Diabetic rat jaw defects: BV/TV +45%, ferroptosis -60% | Li Y et al. 2024 | |
| Caffeic Acid Nanocoating (Desferrioxamine) | Chelates Fe²⁺; activates SLC7A11/GSH/GPX4 | Rat implant models: Osseointegration +35%, lipid peroxides -55% | Yang Y et al. 2024 | |
| Enhancing BMSC osteogenesis | Ferrostatin-1 (Fer-1)-Loaded PLGA Scaffolds | Upregulates Runx2 (+2.3-fold); increases MC3T3-E1 mineralized nodules (+62%) | Rat calvarial defects: BV/TV +32%, ALP activity +45% | Valanezhad A et al. 2021 |
| pH-Responsive MgO₂ Scaffolds | Dynamically regulates iron release; activates Wnt3a pathway | Beagle dog MSL: BV/TV +40%, vascular density +35% | Bai L et al. 2025 | |
| MgO₂/PLGA Nanocomposite Scaffolds | Releases Mg²⁺; inhibits tumor cell ferroptosis (for tumor-associated defects) | Osteosarcoma rat models: Tumor growth -60%, new bone formation +45% | Li C et al. 2024 |
Future Perspectives for Clinical Translation
To translate ferroptosis-based strategies to clinical practice, three priorities emerge, focusing on jaw-specificity, precision, and translatability:
Elucidate Jaw-Specific Ferroptosis Mechanisms via Multi-Omics
Single-Cell Multi-Omics: Utilize single-cell RNA sequencing and spatial transcriptomics to identify ferroptosis-associated cell subpopulations within human jawbone defects. For instance, scRNA-seq analysis of 10 human maxillary sinus augmentation samples revealed three novel ferroptosis-related cell clusters77.
Microbiota-Ferroptosis Integration: Employ 16S rRNA sequencing and metabolomics to explore the mechanisms by which oral microbiota-derived metabolites regulate ferroptosis. A recent study found that butyrate, derived from Porphyromonas gingivalis induces ferroptosis by activating HDAC8, a target not yet fully elucidated78.
Jaw-Femur Comparative Proteomics: Employ proteomics to identify jaw-specific ferroptosis markers. A 2024 study identified differential expression of 51 proteins between jaw and femur osteoblasts, of which 12 are ferroptosis regulators79.
Develop Precision Biomarkers for Clinical Monitoring
The clinical utility of ferroptosis biomarkers requires validation to support personalized augmentation strategies:
Serum Biomarkers: Validate the predictive value of ACSL4 and serum hepcidin for augmentation outcomes. A multicenter study found that serum ACSL4 predicted maxillary sinus augmentation failure with 82% sensitivity40.
Tissue Biomarkers: Develop minimally invasive sampling methods to detect GPX4 activity and MDA levels in the local microenvironment. Gingival crevicular fluid (GCF) GPX4 is associated with an increased risk of peri-implantitis80.
Imaging Biomarkers: Utilize Raman spectroscopy to detect lipid peroxide accumulation in jawbone defects. Clinical studies indicate that Raman spectroscopy quantifies ferroptosis intensity with 90% accuracy81.
Advance Jaw-Tailored Localized Delivery Systems
Photothermal-Responsive Nanocarriers: Design near-infrared (NIR)-responsive liposomes for on-demand drug release specifically at the jaw defect site. Preclinical studies show that NIR-triggered Fer-1 release increases the survival rate of jawbone mesenchymal stem cells78.
Microbe-Responsive Scaffolds: Engineer scaffolds that release ferroptosis inhibitors in response to P. gingivalis LPS. This approach could reduce the required inhibitor dosage, minimizing systemic toxicity82.
3D-Printed Personalized Scaffolds: Utilize patient-specific CT data to 3D-print scaffolds with pore sizes (100-200 μm) and mechanical properties tailored to the jaw defect. 3D-printed biomimetic bioactive glass scaffolds developed by Kolan et al. achieved a 90% implant survival rate in a preliminary human study82.
Discussion
The clinical translation of ferroptosis-targeted therapies for jaw augmentation faces three major challenges: safety validation, regulatory approval, and cost control. Strategies to address these include: conducting long-term toxicity studies to verify material safety (e.g., polydopamine nanoparticles, which have shown good biocompatibility in long-term animal models 65,79); leveraging successful regulatory pathways, such as the FDA's classification of selenium-doped hydroxyapatite scaffolds as "combination" products (device + drug), which provides a template to simplify the approval process for similar products; and employing advanced technologies like microfluidics for scaled-up production of key components (e.g., tFNA, PDA NPs), which can significantly reduce manufacturing costs 79,83,84.
However, several key scientific questions remain to be thoroughly investigated along the path toward clinical application. First, uncovering the biological basis underlying the exceptional sensitivity of jawbone cells to ferroptosis is essential. Future research should focus on whether the unique anatomical microenvironment—particularly its high mitochondrial density and persistent microbial exposure—specifically regulates ferroptosis by driving epigenetic modifications of key genes such as GPX4 85. Second, identifying a precise balance is required: that is, how to activate "moderate" ferroptosis to eliminate senescent cells while avoiding damage to normal osteoblasts. Preclinical data suggest that a combined strategy using low-dose inducers and inhibitors may be a feasible solution 48. Third, the mismatch between the degradation rates of biomaterials and that of new bone formation must be addressed. Developing "ferroptosis-responsive" smart, degradable scaffolds represents a promising approach to prevent post-operative volume collapse 68. Finally, well-designed clinical trials are crucial. Initial studies could focus on patients with mild to moderate jawbone defects. Preliminary findings demonstrating favorable safety and implant survival rates 66 provide an important foundation for subsequent large-scale clinical investigations.
Conclusion
Ferroptosis, driven by iron-dependent lipid peroxidation, serves as a regulator in jawbone augmentation, exhibiting context-dependent effects: moderate activation removes senescent cells and modulates inflammation, whereas excessive activation damages functional cells and impairs regeneration. Jawbone-specific characteristics uniquely regulate ferroptosis through the system xc⁻–GSH–GPX4 axis, mitochondrial ROS, and non-coding RNAs. Clinical studies have confirmed that ferroptosis-modulating biomaterials enhance jawbone regeneration in animal models, although high-quality human clinical trials are still lacking. By elucidating jawbone-specific ferroptotic mechanisms via multi-omics, validating clinical biomarkers, and developing localized delivery systems, traditional augmentation strategies can be optimized, ultimately improving the success rate of dental implants and improving the quality of life of edentulous patients.
Abbreviations
ACSL3: Acyl-CoA synthetase long-chain family member 3; ACSL4: Acyl-CoA synthetase long-chain family member 4; AI-2: Autoinducer-2; ALDH2: Aldehyde dehydrogenase 2; ALP: Alkaline phosphatase; ARE: Antioxidant response element; ATP: Adenosine triphosphate; BMSC: Bone marrow mesenchymal stem cell; BMP2: Bone morphogenetic protein 2; CaMKII: Ca²⁺/calmodulin-dependent protein kinase II; ceRNA: Competing endogenous RNA; circRNA: Circular RNA; CT: Computed tomography; DAMP: Damage-associated molecular pattern; DNA: Deoxyribonucleic acid; FDA: Food and Drug Administration; FSP1: Ferroptosis suppressor protein 1; GCF: Gingival crevicular fluid; GelMA: Gelatin methacryloyl; GPX4: Glutathione peroxidase 4; GSH: Glutathione; GSK-3β: Glycogen synthase kinase-3 beta; HA: Hydroxyapatite; HDAC8: Histone deacetylase 8; H₂O₂: Hydrogen peroxide; HMGB1: High mobility group box 1; HO-1: Heme oxygenase-1; HUVEC: Human umbilical vein endothelial cell; IL-1β: Interleukin-1 beta; IL-6: Interleukin-6; LIP: Labile iron pool; lncRNA: Long non-coding RNA; LOX: Lipoxygenase; LPCAT2: Lysophosphatidylcholine acyltransferase 2; LPS: Lipopolysaccharide; MDA: Malondialdehyde; miRNA: MicroRNA; mtROS: Mitochondrial reactive oxygen species; ncRNA: Non-coding RNA; NIR: Near-infrared; NQO1: NAD(P)H:quinone oxidoreductase 1; Nrf2: Nuclear factor erythroid 2-related factor 2; PDA NP: Polydopamine nanoparticle; PDLSC: Periodontal ligament stem cell; PI3K: Phosphatidylinositol 3-kinase; PLA2: Phospholipase A2; PLGA: Poly(lactic-co-glycolic acid); PUFA: Polyunsaturated fatty acid; PTEN: Phosphatase and tensin homolog; RCD: Regulated cell death; RNA: Ribonucleic acid; ROS: Reactive oxygen species; scRNA-seq: Single-cell RNA sequencing; SLC3A2: Solute carrier family 3 member 2; SLC7A11: Solute carrier family 7 member 11; TGF-β: Transforming growth factor-beta; tFNA: Tetrahedral framework nucleic acid; TFRC: Transferrin receptor; TLR4: Toll-like receptor 4; TNF-α: Tumor necrosis factor-alpha; VEGF: Vascular endothelial growth factor; β-TCP: Beta-tricalcium phosphate.
Acknowledgments
This work was supported by the Anhui Provincial Health and Wellness Research Project (AHWJ2023A10146) and the Scientific Research Foundation for Introducing Talents of Yijishan Hospital, Wannan Medical College (YR202117).
Author’s contributions
All authors equally contributed to this work. All authors read and approved the final manuscript.
Funding
None.
Availability of data and materials
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Wannan Medical College, in accordance with relevant guidelines.
Consent for publication
Not applicable.
Declaration of generative AI and AI-assisted technologies in the writing process
The authors declare that they have not used generative AI (a type of artificial intelligence technology that can produce various types of content including text, imagery, audio and synthetic data.
Competing interests
The authors declare that they have no competing interests.

