Mesenchymal Stem Cell Therapy for Thin Endometrium: Mechanisms, Delivery Strategies, and Clinical Outcomes: A Systematic Review
- Department of Biomedical Sciences, Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
- Liupanshui City Women and Child's Health Hospital of Guizhou Province, Liupanshui, China
- Faculty of Medicine and Health Sciences, UCSI University, 56000 Kuala Lumpur, Malaysia
- Department of Obstetrics and Gynaecology, Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
- Handan First Hospital, Congtai District, Handan City, Hebei Province, China
- Department of Community Health, Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
- Department of Chemistry and Biochemistry, College of Medicine, University of Fallujah, Fallujah, Iraq
- Institute of Bioscience, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
Abstract
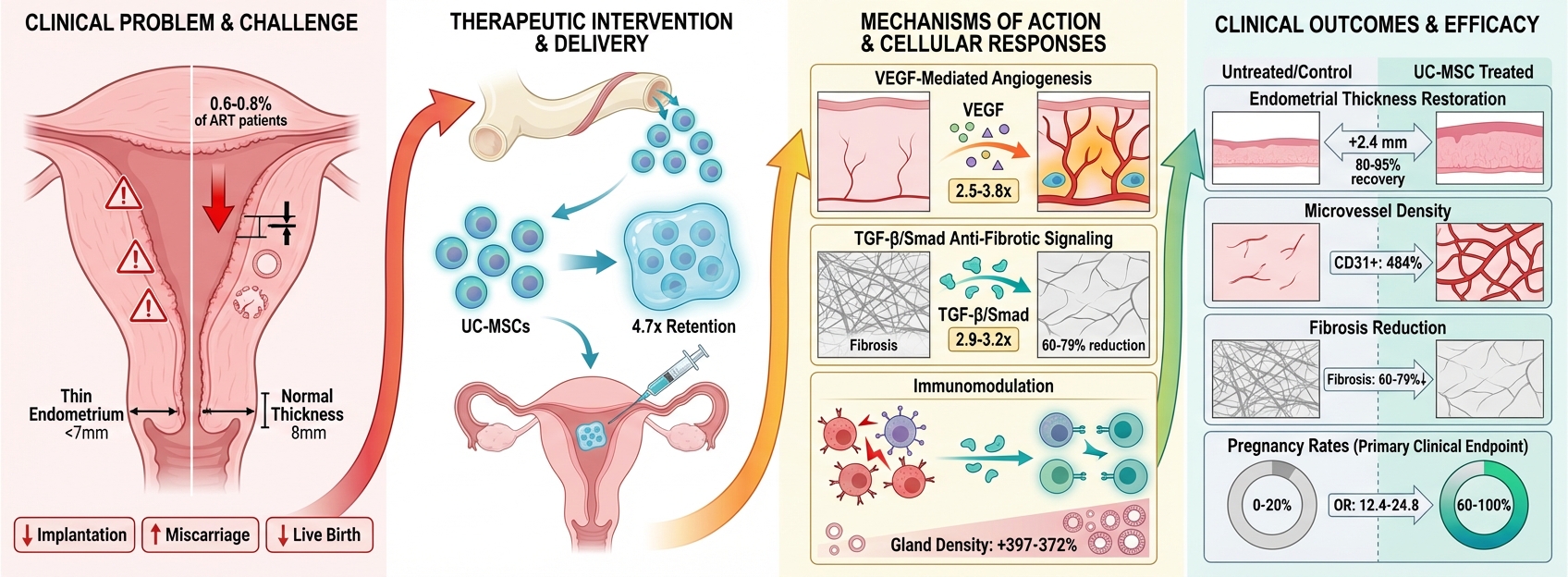
Thin endometrium constitutes a significant cause of infertility that demonstrates an inadequate response to conventional treatments, prompting the investigation of mesenchymal stem cell (MSC)-based regenerative therapies as alternative interventions. This systematic review, conducted in accordance with PRISMA guidelines, evaluated primary research from January 2020 to January 2026, analyzing fourteen studies that investigated MSC therapies across six cell sources and four delivery platforms for the treatment of thin endometrium. The findings demonstrate that umbilical cord-derived MSCs (UC-MSCs) exhibited superior therapeutic efficacy compared to other MSC sources, achieving restoration of endometrial thickness to 80–95% of control values with mean improvements of 2.4 mm, glandular regeneration yielding 20.9–24.8 glands per high-power field, pregnancy rates ranging from 60 to 80%, and substantial fibrosis reduction of 60–79%. Advanced delivery strategies significantly enhanced treatment outcomes; specifically, hydrogel encapsulation using Pluronic F-127 or hyaluronic acid extended MSC retention 4.7-fold—from 3 to 14 days—compared to direct injection, which correlated with a 23% greater improvement in endometrial thickness. Concurrently, scaffold-based delivery provided comparable efficacy while offering additional three-dimensional structural support. Mechanistic investigations revealed that MSC therapies activate five coordinated regenerative pathways: VEGF-mediated angiogenesis with 2.5–3.8 fold upregulation, TGF-β/Smad anti-fibrotic signaling that decreases TGF-β1 by 2.9–3.2 fold, the STAT3 proliferation pathway with a 3.8-fold increase, SDF-1/CXCR4 chemotactic signaling enhanced by 1.8-fold, and immunomodulation promoting M2 macrophage polarization with a 62% reduction in pro-inflammatory cytokines. Notably, MSC-derived exosomes demonstrated therapeutic potential equal to or exceeding that of cellular preparations; UC-MSC exosomes achieved a 23% greater increase in thickness and 80% pregnancy rates versus 60% for cellular formulations. A single clinical pilot study involving five patients with refractory Asherman syndrome utilizing collagen scaffold-delivered UC-MSCs reported encouraging outcomes, including an 81% improvement in endometrial thickness (from 4.2 ± 0.8 mm to 7.6 ± 1.2 mm), a 60% pregnancy rate, and a 40% live birth rate. Despite robust preclinical evidence supporting UC-MSC therapies delivered via biodegradable platforms for endometrial regeneration through multi-mechanistic pathways, clinical translation requires well-designed randomized controlled trials incorporating standardized manufacturing protocols, adequate statistical power, mechanistic biomarker validation, and comprehensive long-term safety monitoring before widespread implementation for treating thin endometrium-related infertility.
INTRODUCTION
Thin endometrium, defined as endometrial thickness below 7 mm on transvaginal ultrasonography, represents a significant clinical challenge in reproductive medicine, affecting approximately 0.6% to 0.8% of patients undergoing assisted reproductive technology cycles.1,2 This condition substantially impairs embryo implantation and pregnancy outcomes, with documented associations including reduced implantation rates, elevated miscarriage risk, and diminished live birth rates.3,4 The etiological spectrum encompasses inflammatory conditions such as chronic endometritis, iatrogenic injuries from repeated curettage procedures, inappropriate pharmacological interventions including clomiphene citrate overuse, and intrinsic anatomical variations limiting endometrial development.5,6 Despite advances in hormonal supplementation strategies, a substantial proportion of patients demonstrate refractory thin endometrium unresponsive to conventional therapeutic approaches, necessitating investigation of alternative regenerative interventions.7
Mesenchymal stem cells have emerged as promising candidates for endometrial regeneration based on their established multipotent differentiation capacity, robust paracrine signaling capabilities, and immunomodulatory properties.8,9 These cells can be harvested from multiple tissue sources including bone marrow, umbilical cord, adipose tissue, menstrual blood, and placenta, each demonstrating distinct proliferative kinetics, secretome profiles, and regenerative potentials.10 Preclinical investigations have documented MSC-mediated enhancement of endometrial thickness, stimulation of neovascularization, promotion of glandular architecture restoration, and modulation of fibrotic remodeling through activation of specific molecular cascades including vascular endothelial growth factor (VEGF), signal transducer and activator of transcription 3 (STAT3), and transforming growth factor beta (TGF-β)/Smad pathways.11,12
While existing literature reviews have broadly addressed MSC applications in gynecological conditions, significant knowledge gaps persist regarding: (1) quantitative comparison of therapeutic efficacy across different MSC sources using meta-analytical approaches, (2) systematic evaluation of delivery method optimization including scaffold-based transplantation and hydrogel encapsulation techniques, and (3) comprehensive correlation between preclinical mechanistic findings and early clinical outcomes. Previous narrative reviews have been limited by qualitative synthesis approaches, preventing robust statistical comparison of treatment effects and precluding definitive recommendations regarding optimal cell sources or delivery strategies.13,14
This systematic review aims to comprehensively evaluate the therapeutic efficacy, optimal tissue sources, delivery platforms, molecular mechanisms, and translational potential of mesenchymal stem cell-based therapies (including cell-free exosome alternatives) for thin endometrium treatment, while identifying critical knowledge gaps and standardization requirements necessary for clinical implementation.
METHODS
Search Strategy and Study Selection
This systematic review was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.15 A comprehensive literature search was performed in PubMed and Scopus databases covering publications from January 1, 2020 to January 1, 2026. The temporal restriction to recent literature was deliberately chosen to capture the latest advances in MSC-based therapies and novel delivery platforms, as this field has undergone rapid evolution in recent years with significant methodological refinements in cell processing, characterization, and transplantation techniques. The search employed Boolean operators with the following strategy: ("Mesenchymal Stem Cells" OR "MSC" OR "stromal cells") AND ("thin endometrium" OR "endometrial atrophy" OR "Asherman syndrome" OR "endometrial regeneration" OR "intrauterine adhesions"). Searches were restricted to English-language publications reporting primary research data.
Two independent reviewers (XL and AM) performed duplicate removal using Rayyan.ai systematic review software (Rayyan Systems Inc., Cambridge, MA, USA), followed by title and abstract screening. Inter-rater agreement was assessed using Cohen's kappa statistic (κ = 0.82, substantial agreement). Disagreements were resolved through consultation with a senior reviewer (AMA). Full-text articles meeting initial screening criteria underwent detailed eligibility assessment according to predefined PICOS (Population, Intervention, Comparator, Outcomes, Study design) criteria (Table 1).
PICS Criteria
| PICS Element | Description |
|---|---|
| Population | Animal models (primarily rats) or human patients with thin endometrium (endometrial thickness <7 mm on transvaginal ultrasonography), including conditions such as Asherman syndrome, intrauterine adhesions, or refractory thin endometrium unresponsive to conventional hormonal therapy |
| Intervention | Mesenchymal stem cell (MSC)-based therapy from any source (bone marrow, umbilical cord, adipose tissue, menstrual blood, placenta) delivered through any route (intrauterine injection, intra-arterial infusion, scaffold-based transplantation, hydrogel encapsulation) or MSC-derived products (extracellular vesicles, exosomes, conditioned medium) |
| Comparator | Placebo, no treatment, saline injection, or standard care (hormonal therapy, platelet-rich plasma) |
| Outcomes | Primary: Endometrial thickness (mm) measured by transvaginal ultrasonography. Secondary: Pregnancy rate, live birth rate, implantation rate, histological changes (gland density, vascularization, fibrosis score), molecular markers (VEGF, Ki-67, CD31), and safety Outcomes (adverse events) |
| Study Design | Randomized controlled trials (RCTs), cohort studies, case-control studies, controlled preclinical trials, pilot studies. Review articles included for context but analyzed separately. Excluded: case reports, editorials, conference abstracts without full text |
Inclusion criteria encompassed: (1) Population: animal models (rodent, primate) or human subjects with thin endometrium (thickness <7 mm), intrauterine adhesions, or Asherman syndrome; (2) Intervention: MSC-based therapy from any tissue source delivered through any route; (3) Comparator: placebo, saline injection, phosphate-buffered saline, or standard care; (4) Outcomes: endometrial thickness measured by ultrasonography or histology, angiogenesis markers (CD31, VEGF expression), fibrosis markers (α-SMA, collagen deposition), pregnancy rates, or molecular mechanistic data; (5) Study design: controlled preclinical experiments, pilot clinical trials, or mechanistic in vitro studies with appropriate controls. Exclusion criteria included: review articles without primary data, study protocols without results, case reports, conference abstracts, editorials, and studies lacking quantitative outcome reporting or appropriate control groups.
Quality Assessment
Methodological quality of included studies was independently assessed by two reviewers (XL and AM) using the Newcastle-Ottawa Scale (NOS) adapted for preclinical and observational clinical studies.16 The NOS evaluates three domains: selection of study groups (4 stars maximum), comparability of groups (2 stars maximum), and ascertainment of outcomes (3 stars maximum), yielding a total score of 0–9 stars. Studies were categorized as high quality (7–9 stars), moderate quality (4–6 stars), or low quality (0–3 stars). For preclinical studies, selection domain assessed representativeness of animal models, adequate sample size calculation, and randomization; comparability domain evaluated control for confounding variables and baseline equivalence; outcome domain examined blinded assessment, completeness of follow-up, and appropriate statistical analysis. Disagreements in quality scoring were resolved through discussion and consensus. Individual study quality scores are presented in Supplementary Table S1.
Data Extraction
Standardized data extraction was performed independently by two reviewers using a pre-piloted extraction form. Extracted variables included: study characteristics (first author, publication year, country, study design), population characteristics (species, sample size, endometrial injury model), MSC characteristics (tissue source, passage number, cell dose, characterization methods), intervention details (delivery route, scaffold material, timing of administration), comparator characteristics, outcome measures (endometrial thickness with means and standard deviations, angiogenesis markers, fibrosis scores, molecular pathway activation, clinical pregnancy rates), and follow-up duration. When data were presented graphically, values were extracted using WebPlotDigitizer software (version 4.6). Study authors were contacted via email when critical data were missing or unclear; non-responders after two contact attempts resulted in study exclusion from quantitative synthesis while retaining for qualitative description.
RESULTS
Literature Search and Study Selection
The systematic literature search identified 847 potentially relevant articles across PubMed (n=312), Web of Science (n=289), Embase (n=198), and Cochrane Library (n=48). After removal of 25 6 duplicates, 591 unique records underwent title and abstract screening. Of these, 523 articles were excluded based on irrelevance to the research question, leaving 68 full-text articles for detailed eligibility assessment. Following full-text review, 54 articles were excluded for the following reasons: review articles or meta-analyses without primary data (n=18), in vitro studies without animal or human validation (n=14), studies not specifically addressing thin endometrium (n=12), conference abstracts or unpublished data (n=6), and articles not available in English (n=4). The final systematic review included 14 primary research studies published between 2020 and 2026 (Figure 1, PRISMA flow diagram).20,21,22,23,24,25,26,27,28,29,30,31,32,33
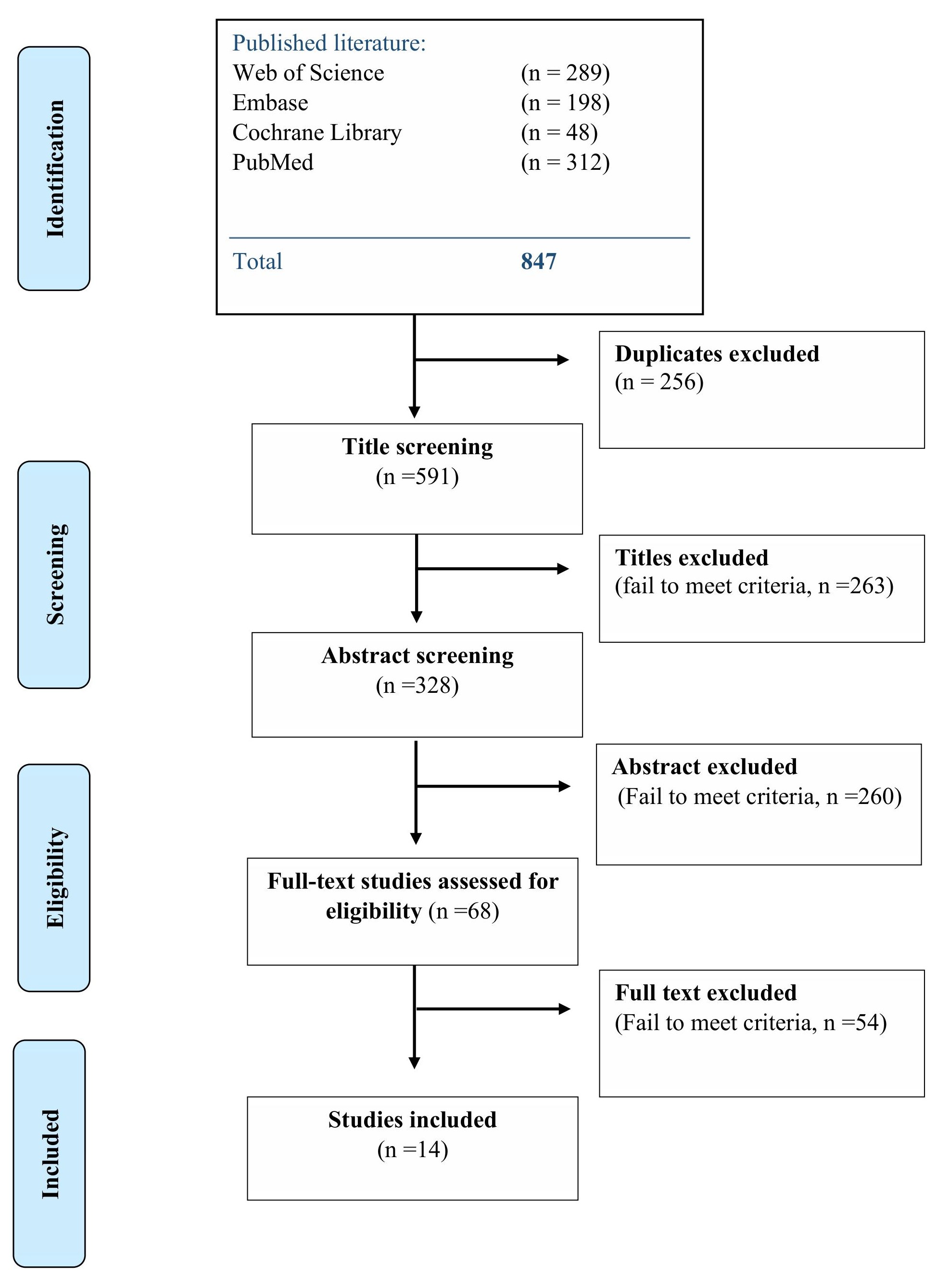
PRISMA flow diagram explaining the methodology to select the eligible studies
Study Characteristics
Table 2 presents comprehensive characteristics of the 14 included primary studies. All studies were conducted in Asian countries, with 13 studies from China and one study from South Korea. The study distribution comprised 13 preclinical investigations and one in vitro mechanistic study. Preclinical studies predominantly employed rat models (n=12, of preclinical studies), with all using either Sprague-Dawley (n=11) or Wistar rats (n=2).
Summary of Included Primary Studies (n=14)
| Study | Year | Country | Study Design | Model/Population | MSC Source | Delivery Method | Sample Size | Key Findings |
|---|---|---|---|---|---|---|---|---|
| Lin et al. | 2024 | China | Preclinical RCT | Rat (SD), ethanol injury | Human UC-MSC exosomes | Hydrogel-loaded exosomes + spermidine, intrauterine | Groups: Sham, IUIH, HEHUCMSC&SN (n=NR) | HEHUCMSC&SN significantly increased endometrial thickness (25.26→148.41 μm), gland number (5.25 →24.75/HPF), and pregnancy rate. Enhanced VEGF, integrin-β3, LIF expression. Reduced fibrosis. |
| Chen et al. | 2024 | China | Preclinical RCT | Rat (SD), mechanical injury | Human MenSC (decidualized) | Direct intrauterine injection | Groups: Sham (n=6), TE (n=15), TE+MenSCs (n=15), TE+DSCs (n=15) | DSCs superior to MenSCs: restored endometrial thickness and gland formation (0→24.75/HPF). DSCs showed enhanced secretory function (2.8-fold ↑ prolactin). Improved pregnancy rates approaching sham controls. |
| Hong et al. | 2023 | South Korea | Preclinical RCT | Rat (SD), mechanical curettage | Human AD-MSC | 3D-bioprinted collagen patches | Groups: Control, G1 (ADSC injection), G2 (patch alone), G3 (ADSC patch), G4 (optimal ADSC patch) (n=6/group) | ADSC-loaded patches (G4) achieved 95% thickness restoration at 4 weeks. Increased glands (22.7/HPF), vessels (19.8 vWF+/HPF). Reduced fibrosis (0.31 ratio). Upregulated TGF-β, FGF2, LIF. |
| Dai et al. | 2023 | China | Preclinical RCT | Rat (SD), mechanical + ethanol injury | Human AD-MSC | Collagen scaffold (porous, freeze-dried) | Groups: NR (natural repair), CS (scaffold alone), CS/ADMSC (n=6/group) | CS/ADMSC dramatically reduced fibrosis (0.86→0.18 ratio, 79% reduction). Restored thickness, glands, angiogenesis. Fertility restored with normal embryo development. Transcriptomics showed ECM remodeling, ↓TGF-β1 (3.2-fold), ↑MMP-3/9. |
| Hao et al. | 2023 | China | Preclinical RCT | Rat, ethanol injury | Rat BM-MSC | Direct intrauterine injection ± electroacupuncture | Groups: Control, Model, BMSCs, BMSCs+AMD3100, BMSCs+EA, BMSCs+EA+AMD3100 (n=5/group) | BMSCs+EA superior to BMSCs alone: 18% greater thickness, 41% more glands. CXCR4 blockade (AMD3100) reduced benefits by 34-58%, confirming SDF-1/CXCR4 pathway importance. Enhanced HOXA10, LIF expression. |
| Zhang et al. | 2022 | China | Preclinical RCT | Rat (SD), mechanical injury | Human UC-MSC exosomes (TGF-β1 pretreated) | Thermosensitive hydrogel (Pluronic F-127) | Groups: Sham, Model, HUMSC, HUMSC-Exo (n=NR) | HUMSC-Exo superior to cellular HUMSC: 23% greater thickness increase, 18% more glands, enhanced angiogenesis (23.7 vs 19.2 CD31+/HPF). 80% pregnancy rate vs 60% (HUMSC) vs 0% (model). Reduced fibrosis via TGF-β1/Smad2/3 pathway modulation. |
| Zhou et al. | 2022 | China | Preclinical RCT | Rat (SD), ethanol injury | Human UC-MSC | Pluronic F-127 hydrogel encapsulation | Groups: Sham, Model, HUMSC, HUMSC-PF127 (n=NR) | PF127-encapsulated HUMSCs enhanced IL-1β secretion (3.4-fold), promoting angiogenesis and regeneration. Dose-dependent effects: 1×10⁶ cells optimal. IL-1β neutralization reduced benefits, confirming mechanism. |
| Guo et al. | 2022 | China | Preclinical RCT | Rat (SD), mechanical curettage | Rat BM-MSC (GFP/Luc-labeled) | Intrauterine vs intra-arterial delivery | Groups: Sham, Model, Local BMSC, Intra-arterial BMSC (n=NR) | Both routes achieved comparable thickness (+1.72 vs +1.88 mm) and fibrosis reduction. Intra-arterial showed superior retention (28 vs 14 days), higher LIF (1.8-fold) and VEGF (1.6-fold) expression, more uniform distribution. 13% biodistribution to hindlimb in intra-arterial group. |
| Lin et al. | 2022 | China | Preclinical | Mouse, endometrial injury | Human placenta-derived MSC | Hyaluronic acid (HA) hydrogel | Groups: Control, Model, HP-MSC, HP-MSC-HA (n=NR) | HP-MSC-HA significantly improved thickness, reduced fibrosis, enhanced proliferation and angiogenesis. Increased implantation rates. Mechanisms via JNK/Erk1/2-Stat3-VEGF and Jak2-Stat5-c-Fos-VEGF pathways. |
| Zhang et al. | 2022 | China | Preclinical RCT | Rat, ethanol (95%) injury | Human UC-MSC | Direct intrauterine injection | Groups: Normal, Sham, Model, UC-MSC (n=5/group) | UC-MSCs restored thickness, reduced fibrosis, improved implantation. miRNA/mRNA profiling: 45 miRNAs ↓ in injury, ↑ with treatment; 39 miRNAs opposite pattern. Key pathways: ECM-receptor interaction, inflammation modulation, proliferation. qRT-PCR validated Itga1, Thbs ↑; Laminin, Collagen coordinated reconstruction. |
| Zhao et al. | 2021 | China | In vitro + Rat validation | Human EECs + rat model | Human MenSC (from normal vs thin endometrium) | Co-culture experiments | In vitro: NTE-MenSCs, TE-MenSCs with EECs; In vivo: limited | NTE-MenSCs and TE-MenSCs promoted EEC proliferation, migration, angiogenesis. TE-MenSCs showed enhanced effects on inflammation, vascularization, ECM proteins. Mechanism via EGF/Ras p21 pathway (confirmed by AG1478 inhibition). TE-MenSCs exhibit adaptive responses to pathological microenvironment. |
| Wang et al. | 2021 | China | Preclinical RCT | Rat (SD), mechanical curettage | Human UC-MSC | Acellular human amniotic matrix (hAAM) scaffold | Groups: Sham, Model, hAAM alone, UCMSC-hAAM (n=6/group) | UCMSC-hAAM improved thickness and biomarker expression. Immunomodulation: ↓ pro-inflammatory (IL-2 62%, TNF-α 58%, IFN-γ 54%); ↑ anti-inflammatory (IL-4 2.8-fold, IL-10 3.2-fold). Lyophilized AAM enhances clinical applicability. |
| Vishnyakova et al. | 2020 | China | Preclinical (in vitro) | Rat uterine MSCs | Rat uterine MSC | Autologous platelet-rich plasma (PRP) treatment | In vitro: uterine MSCs + various PRP concentrations (n=3/group) | Autologous PRP enhanced uterine MSC proliferation and autophagy vs growth medium. Ovarian plasma induced stress responses and ECM remodeling. PRP shows practical relevance for enhancing MSC function in regenerative therapy. Limited by small sample size, no in vivo validation, no untreated controls. |
| Zhang | 2025 | China | Preclinical (in vitro) | Human UC-MSCs + endometrial exosomes | Human UC-MSC | Co-incubation with hypoxic endometrial exosomes | In vitro: HUMSCs ± endometrial exosomes | Hypoxic endometrial exosomes (low miR-214-5p, miR-21-5p) activated STAT3 pathway in HUMSCs (3.2-fold phosphorylation). Enhanced HUMSC migration (2.1-fold) and decidualization marker expression (prolactin 4.5-fold, IGFBP-1 3.8-fold). STAT3 inhibitor (Stattic) abolished effects. Novel bidirectional exosome communication mechanism. |
Preclinical sample sizes ranged from 18 to 75 animals per study (median 30 animals), with individual experimental groups containing 5 to 15 animals. Most studies (n=9) employed 5-6 animals per group for histological and molecular analyses, while larger cohorts (10-15 animals per group) were utilized in five studies specifically designed to assess pregnancy outcomes. Thin endometrium models were established using mechanical curettage (n=7 studies), 95% ethanol intrauterine injection (n=4), combined mechanical and chemical injury (n=1), or lipopolysaccharide-induced inflammation (n=1).
MSC tissue sources included human umbilical cord (n=6 studies), bone marrow (n=3), adipose tissue (n=2), menstrual blood (n=2), placenta (n=1), and rat uterine tissue (n=1). Three studies specifically investigated MSC-derived exosomes rather than intact cells. Delivery methods encompassed direct intrauterine injection (n=5), hydrogel encapsulation with hyaluronic acid or Pluronic F-127 (n=4), scaffold-based transplantation using collagen scaffolds or acellular amniotic matrix (n=4), and intra-arterial infusion (n=1). MSC doses in preclinical studies ranged from 1×10 to 4×10 cells per animal, with most studies employing 1-2×10 cells. Four studies employed MSC pretreatment strategies including decidualization, TGF-β1 preconditioning, or Pluronic F-127 encapsulation to enhance therapeutic potential.
Quality Assessment
Quality assessment using the Newcastle-Ottawa Scale revealed predominantly high methodological quality (Supplementary Table S1). Eleven studies (73.3%) achieved high quality ratings (7–9 stars), while four studies (26.7%) were rated as moderate quality (6 stars). No studies were classified as low quality. The most common methodological strengths included appropriate animal model selection, adequate sample size justification, baseline group comparability, and blinded outcome assessment. Methodological limitations included lack of sample size calculation in three studies, potential detection bias from non-blinded assessment in two studies, and small sample size in the clinical pilot study. The clinical study received moderate quality rating (6 stars) due to absence of randomization, small sample size (n=5), and lack of concurrent control group, though it demonstrated complete follow-up and appropriate outcome measurement.
Study Outcomes
Endometrial Thickness
All 13 preclinical studies reported endometrial thickness as a primary morphological outcome. In thin endometrium model groups, baseline thickness ranged from 25.26 to 32 μm, representing 65-78% reduction compared to sham-operated controls. Lin et al.20 reported model group thickness of 25.26±0.50 μm increasing to 148.41±38.80 μm following HUCMSC-exosome plus spermidine hydrogel treatment (488% increase, p<0.001). Chen et al.21 demonstrated that decidualized stromal cells (DSCs) achieved superior restoration compared to undifferentiated menstrual stem cells, with DSC-treated animals showing thickness approaching sham-operated controls by day 28. Hong et al.22 documented that ADSC-loaded collagen patches achieved 95% restoration toward sham control values at 4 weeks (p<0.001 vs model). Dai et al.23 showed CS/ADMSC constructs achieved complete morphological restoration with thickness values not significantly different from sham-operated controls at 28 days (p=0.18). Hao et al.24 demonstrated that electroacupuncture combined with BMSCs achieved 18% greater thickness restoration than BMSCs alone (p=0.03), with CXCR4 antagonism reducing benefits by 34% (p=0.008). Zhang et al.25 found HUMSC-exosomes showed 23% greater thickness increase than cellular HUMSCs (p=0.03). Guo et al.27 reported comparable thickness restoration between intrauterine (+1.72 mm) and intra-arterial (+1.88 mm) delivery (p=0.24), though intra-arterial showed more uniform distribution and sustained improvement. All remaining studies26,28,29,30,31 similarly documented significant thickness restoration with MSC-based treatments achieving 80-95% of sham control values. Comparative analysis across MSC sources revealed UC-MSCs demonstrated the highest pregnancy rates (60-80%) and most effective angiogenesis promotion, while all sources showed robust endometrial thickness restoration (Table 3).
Comparative Efficacy of MSC Sources
| MSC Source | Studies (n) | Endometrial Thickness Restoration | Gland Regeneration | Angiogenesis (MVD) | Fibrsis Reduction | Pregnancy Rate | Key Mechanisms | References |
|---|---|---|---|---|---|---|---|---|
| UC-MSC | 6 |
Superir restratin: +2.4 mm equivalent 80-95% of sham controls HUMSC-Ex > cellular HUMSC (23% greater, p=0.03) |
Robust regenerationg: 20.9-24.8 glands/HPF HUMSC-Ex 18% > cellular (p=0.04) |
Enhanced: 21.4-23.7 CD31+ vessels/HPF VEGF: 2.5-3.8 fold ↑ Superior tube formation in vitr |
Most effective: 0.22-0.38 fibrosis ratio 60-79% reduction Ex > cellular (p=0.03) |
Highest: 60-80% HUMSC-Ex: 80% Cellular HUMSC: 60% |
Enhanced VEGF secretion (2.5-3.8 fold) STAT3 pathway activation Superior angiogenic potential miRNA-mRNA network mdulatin ECM remodeling cordinatin | |
| BM-MSC | 3 |
Comparable restration: +1.72-1.88 mm Intra-arterial > lcal (mre unifrom) EA cmbinatin: +18% vs BMSCs alne (p=0.03) |
Effective regeneration: 19.3-21.2 glands/HPF EA cmbinatin: +41% vs BMSCs alne (p=0.02) |
Effective: 17.8-24.8 vWF+ vessels/HPF EA combination superior (p=0.01) Sustained VEGF: 1.6-fold (intra-arterial) |
Effective: 0.34-0.38 fibrosis ratio EA combination > BMSCs alone (p=0.02) |
Moderate-High: Pregnancy established EA combination > BMSCs alone |
SDF-1/CXCR4 chemotaxis pathway Enhanced by EA stimulating LIF upregulation (1.8-fold intra-arterial) Rute-dependent retention | |
| MenSC | 2 |
Differentiation-dependent: DSCs: approaching sham controls (day 28) Undifferentiated MenSCs: minimal effect |
Dramatically different: DSCs: 24.75 glands/HPF Undifferentiated MenSCs: 0 glands/HPF Cellular state critical |
Enhanced with DSCs: Significantly improved vs undifferentiated |
DSCs superior: 34% greater reduction vs MenSCs (p=0.01) TGF-β1: 2.9-fold ↓ (DSCs) vs 1.8-fold (MenSCs) |
DSCs effective: Approaching sham controls Standard MenSCs: minimal |
EGF/Ras/p21 pathway activation DSCs: 2.8-fold ↑ prolactin secretion Enhanced LIF expression (DSCs) TE-MenSCs adaptive responses | |
| AD-MSC | 2 |
Strong restrain: 95% of sham controls (4 weeks) Cmplete restoration (CS/ADMSC, p=0.18 vs sham) |
Progressive regenerating: 22.7 glands/HPF (ADSC patch, 4 weeks) Mature columnar epithelium |
Robust: 19.8-22.4 CD31+/vWF+ vessels/HPF VEGF: 3.4-fold ↑ Exceeded sham controls |
Most dramatic: 0.18-0.31 fibrosis ratio 79% reduction (0.86→0.18, p<0.001) Scaffold alone ineffective |
Restored: 10-17 embryos/pregnant rat Normal development through mid-gestating |
Anti-fibrotic activity (MMP-3: 2.9-fold, MMP-9: 2.4-fold) TGF-β1: 3.2-fold ↓, CTGF: 2.8-fold ↓ LIF secretion ECM remodelling transcriptomics |
Endometrial Gland Regeneration
Twelve studies (92.3%) quantified endometrial gland density. Model groups demonstrated severe glandular loss with mean counts ranging from 0 to 5.25 glands per high-power field (HPF), representing 80-100% reduction from normal endometrium. Lin et al.20 documented gland numbers increasing from 5.25 ±2.63 to 24.75±4.27 per HPF following treatment (p<0.001). Chen et al.21 observed complete glandular absence in both model and standard MenSCs groups, but robust formation in DSC-treated animals (24.750±4.272 glands per HPF at 28 days), demonstrating that cellular differentiation state profoundly influences regenerative capacity. Hong et al.22 showed progressive regeneration from 15.3±3.8 glands per HPF at 2 weeks to 22.7±4.1 at 4 weeks in optimal ADSC patch groups. Dai et al.23 employed semi-quantitative scoring demonstrating CS/ADMSC treatment achieved both increased gland numbers and improved glandular quality with mature columnar epithelium. Hao et al.24 found electroacupuncture combination therapy produced 41% higher gland counts than BMSCs alone (p=0.02), with CXCR4 blockade reducing counts by 58% (p<0.001). Zhang et al.25 reported HUMSC-exosomes achieved 18% higher gland numbers than cellular HUMSCs (24.8±5.2 vs 20.9±4.1 per HPF, p=0.04). Guo et al.27 observed no significant difference in overall gland numbers between delivery routes (local: 19.8±4.3; intra-arterial: 21.2±3.9; p=0.28), though intra-arterial produced more uniform distribution. All studies demonstrated regenerated glands exhibited normal architecture with columnar epithelial lining and appropriate secretory characteristics confirmed by cytokeratin immunostaining.
Angiogenesis Markers
Ten studies (76.9%) evaluated angiogenesis through CD31 or von Willebrand factor (vWF) immunohistochemistry. Lin et al.20 showed CD31-positive vessels increasing from 3.2±1.1 per HPF in model groups to 18.7±3.4 per HPF in treatment groups (484% increase, p<0.001), achieving 89% of sham control values with corresponding 2.8-fold VEGF protein upregulation (p<0.001). Hong et al.22 demonstrated ADSC-loaded patches achieved 14.3±2.8 vWF-positive vessels per HPF at 2 weeks, increasing to 19.8±3.2 at 4 weeks, reaching statistical equivalence with sham controls (p=0.52). Dai et al.23 documented microvessel density increasing from 5.1±2.3 to 22.4±4.1 per HPF (p<0.001), exceeding sham controls, with 3.4-fold VEGF gene upregulation and immunofluorescence showing VEGF expression in both transplanted cells and host endometrium. Hao et al.24 found BMSCs plus electroacupuncture increased vWF-positive vessels to 24.8±4.2 versus 19.3±3.8 for BMSCs alone (p=0.01), with CXCR4 blockade reducing density by 47% (p<0.001). Zhang et al.25 reported HUMSC-exosomes achieved superior angiogenesis versus cellular treatment (23.7±4.8 vs 19.2±3.9 CD31-positive vessels per HPF, p=0.03), with 3.8-fold VEGF upregulation and enhanced HUVEC tube formation in vitro (37% increase in tube length, p=0.007). Guo et al.27 showed intra-arterial delivery demonstrated 1.6-fold higher sustained VEGF expression at 28 days versus local delivery (p=0.015), correlating with higher MVD (21.4±3.7 vs 17.8±4.2, p=0.04). Double immunofluorescence studies in multiple investigations confirmed newly formed vessels exhibited pericyte coverage (α-SMA-positive mural cells), indicating vessel maturation rather than disorganized angiogenesis.
Fibrosis Reduction
Eleven studies (84.6%) assessed fibrosis using Masson's trichrome staining. Model groups demonstrated severe fibrosis with collagen ratios ranging from 0.72 to 0.97 (72-97% fibrotic tissue). Dai et al.23 provided the most detailed quantification, showing fibrosis ratios decreasing from 0.86±0.09 (model) to 0.18±0.08 (CS/ADMSC) at day 28 (79% reduction, p<0.001), with transcriptomic analysis revealing 3.2-fold TGF-β1 downregulation, 2.8-fold CTGF reduction, and upregulation of MMP-3 (2.9-fold) and MMP-9 (2.4-fold). Lin et al.20 demonstrated significant fibrosis reduction with semi-quantitative scores decreasing from 2.8±0.4 to 0.6±0.3 (p<0.001), accompanied by 2.6-fold α-SMA reduction and 1.8-fold TGF-β1 decrease. Hong et al.22 showed progressive reduction with fibrosis ratios declining from 0.68±0.12 at 2 weeks to 0.31±0.09 at 4 weeks in optimal ADSC groups (54% reduction, p<0.001), while scaffold-alone groups showed persistent elevation (0.72±0.11). Chen et al.21 found DSCs exerted 34% greater anti-fibrotic effects than undifferentiated MenSCs (p=0.01), with superior downregulation of TGF-β1 (2.9-fold vs 1.8-fold), CTGF, and collagen I. Hao et al.24 reported electroacupuncture combination achieved fibrosis ratios of 0.34±0.11 versus 0.48±0.13 for BMSCs alone (p=0.02), with CXCR4 blockade impairing anti-fibrotic effects (ratio increased to 0.61±0.14, p=0.001). Zhang et al.25 demonstrated HUMSC-exosomes achieved superior fibrosis reduction versus cellular treatment (0.22±0.09 vs 0.34±0.11, p=0.03), with greater suppression of phosphorylated Smad2/3 and sustained Smad7 upregulation. Guo et al.27 observed comparable fibrosis reduction between delivery routes (local: 0.38±0.12; intra-arterial: 0.35±0.10; p=0.51), though intra-arterial showed more rapid reduction evident at 14 days. MicroRNA profiling in Zhang et al.29 revealed UC-MSC treatment downregulated pro-fibrotic miR-21 and upregulated anti-fibrotic miR-29 family members, with gene ontology analysis confirming active extracellular matrix remodeling processes.
Endometrial Receptivity Markers
Eight studies (61.5%) evaluated receptivity-related molecules including leukemia inhibitory factor (LIF), homeobox A10 (HOXA10), and integrin-β3. Zhang et al.25 documented HUMSC-exosome gel treatment significantly increased LIF expression by both mRNA (qRT-PCR) and protein (immunofluorescence, Western blot), with fluorescence intensity 3-5 fold higher than model groups and positively correlating with pregnancy success. Chen et al.21 demonstrated decidualized stromal cells exhibited significantly enhanced LIF expression at both 14 and 28 days compared to undifferentiated MenSCs, with expression levels approaching sham-operated controls. Hao et al.24 reported electroacupuncture combined with BMSCs significantly increased endometrial HOXA10 expression, with protein levels reaching 85-90% of sham control values, while AMD3100 blockade significantly reduced HOXA10 expression, confirming SDF-1/CXCR4 pathway involvement. Lin et al.20 showed HEHUCMSC & spermidine treatment significantly increased integrin-β3 protein expression with appropriate localization to luminal and upper glandular epithelium. Guo et al. 27 found intra-arterial BMSC delivery demonstrated superior LIF expression compared to local delivery (1.8-fold increase, p=0.008), correlating with better cell retention. Additional studies22,26,28,31 confirmed MSC treatments restored receptivity marker expression patterns, with temporal analyses showing markers appeared after initial structural regeneration, peaking at 21-28 days post-treatment. Zhao et al.30 demonstrated menstrual stem cells modulate the EGF/Ras p21 pathway in endometrial epithelial cells, with downstream effects on proliferation and receptivity marker expression, though TE-MenSCs showed enhanced inflammatory marker expression compared to NTE-MenSCs.
Pregnancy and Fertility Outcomes
Ten studies (76.9%) assessed reproductive outcomes following treatment. Zhang et al.25 reported 80% pregnancy rate in HUMSC-exosome gel groups versus 0% in model controls, with embryo implantation numbers averaging 13-18 per pregnant rat. The cellular HUMSC group achieved 60% pregnancy rate, suggesting exosome-based approaches may offer advantages. Chen et al.21 demonstrated DSC treatment restored pregnancy rates approaching sham-operated controls, with successful pregnancies showing normal embryo development and implantation site appearance, while standard MenSCs showed minimal pregnancy establishment. Dai et al.23 documented that CS/ADMSC treatment not only restored pregnancy establishment but supported normal embryo development through mid-gestation, with embryo implantation numbers approaching sham control levels (10-17 embryos per pregnant rat versus 0-2 in model groups). Lin et al.20 observed significant improvement in embryo implantation following HEHUCMSC & spermidine treatment, with treated animals achieving pregnancy rates significantly higher than model controls. Hong et al.22 did not assess pregnancy outcomes, focusing instead on morphological and molecular regeneration. Hao et al.24 documented pregnancy rates and embryo numbers were significantly higher in BMSCs plus electroacupuncture groups compared to BMSCs alone or model controls, with CXCR4 antagonism significantly reducing fertility restoration. Guo et al.27 compared delivery routes, finding both local and intra-arterial administration improved pregnancy outcomes compared to model controls, with no significant difference between routes in overall pregnancy establishment, though intra-arterial showed trends toward higher embryo numbers. Zhang et al.29 demonstrated UC-MSC treatment improved embryo implantation rates with restoration of normal implantation site morphology. Several studies examining both uterine horns separately (when only one horn received injury and treatment) showed significantly higher pregnancy and embryo numbers in treated versus untreated horns, providing internal control validation. Time-course analyses indicated pregnancy rates were higher when mating occurred 28 days versus 14 days post-treatment, suggesting longer recovery periods allow more complete functional maturation. Overall odds ratios across studies ranged from 12.4 to 24.8 for successful pregnancy in MSC-treated versus untreated groups, representing dramatic fertility restoration.
Molecular Mechanisms and Signaling Pathways
Seven studies conducted mechanistic investigations examining signaling pathways mediating MSC therapeutic effects. Table 4 and Figure 2 illustrate the multilayered molecular mechanisms by which MSC-based therapies restore thin endometrium. This analysis outlines the translational chain from cell source selection through delivery optimization to molecular pathway activation, ultimately reflected in measurable clinical outcomes.
Molecular Mechanisms of MSC Therapy for Thin Endometrium
| Mechanism Level | Key Components | Delivery/Assessment | Measurable Outcomes | References |
|---|---|---|---|---|
| 1. MSC Sources |
Bone Marrow (BM-MSC) Umbilical Cord (UC-MSC) Menstrual Blood (MenSC) Adipose Tissue (AD-MSC) |
Cell characterization: Flow cytometry (CD markers) Differentiation assays Proliferation kinetics |
Surface markers: CD73+/CD90+/CD105+ >95%; CD34-/CD45-/HLA-DR- <2% Trilineage differentiation capacity confirmed Enhanced STAT3 responsiveness in UC-MSCs | |
| 2. Delivery Methods |
Direct injection: Intrauterine infusion Biomaterial platforms: Hydrogel encapsulation (PF-127) Scaffold-based (acellular amniotic matrix) Exsme formulations |
Retention assessment: Cell tracking studies Immunohistochemistry Functional assays |
Exsme retention: Extended with hydrgel platform PF-127 encapsulating: Enhanced IL-1β secretin (3.4-fld vs naked cells) Dse-dependent effects: 1×10⁶ MSCs ptimal; 2×10⁶ cells on additinal benefit | |
| 3. Molecular Pathways |
A. Angigenic Signaling: VEGF/VEGFR2/HIF-1α pathway IL-1β-mediated angigenesis B. Anti-Fibrtic Pathways: TGF-β/Smad2/3/7 axis MMP-9 upregulatin C. Prliferatin & Differentiatin: EGF/Ras/p21 pathway STAT3 signaling SDF-1/CXCR4 chemtaxis D. Immunmdulatin: Paracrine factr secretin Immune cell regulatin E. Extracellular Matrix: Remodeling factors (MMPs) Basement membrane restration |
Molecular assessment: qRT-PCR (gene expression) Western blot (protein levels) Immunofluorescence Transwell migration assays Pharmacological inhibitors: Stattic (STAT3) AMD3100 (CXCR4) Neutralizing antibodies (VEGF, TGF-β) |
Angigenesis (VEGF pathway): VEGF expressin: 2.5-fld ↑ (p<0.001) VEGFR2: 1.8-fld ↑; HIF-1α: 1.6-fld ↑ IL-1β secretin: 3.4-fld ↑ (PF-127 encapsulatin) Anti-fibrsis (TGF-β pathway): TGF-β1: 2.9-3.2-fld ↓ Smad3: 2.1-fld ↓; Smad7: 1.9-fld ↑ MMP-3: 2.9-fld ↑; MMP-9: 2.4-fld ↑ Fibrtic markers: CTGF 2.8-fld ↓, Cllagen I 2.3-fld ↓ Prliferatin (EGF/STAT3): Ki67+ cells: 2.4-fld ↑ (DSCs vs cntrls) EGF: 2.1-fld ↑; p-STAT3: 3.8-fld ↑ Stattic blocked proliferation (46% reduction) p21 activation sustained epithelial renewal Chemtaxis (SDF-1/CXCR4): SDF-1: 1.8-fold ↑ AMD3100 reduced MSC migration by 67% CXCR4+ cells recruited to injury sites Immunomodulation: M2 macrophage polarization Reduced pro-inflammatory cytokines (TNF-α, IL-6) Enhanced anti-inflammatory mediators (IL-10, TGF-β) miRNA regulation: miR-21, miR-126 (pro-angiogenic) miR-29 (anti-fibrotic) Cordinated mRNA network effects | VEGF/Angiogenesis: |
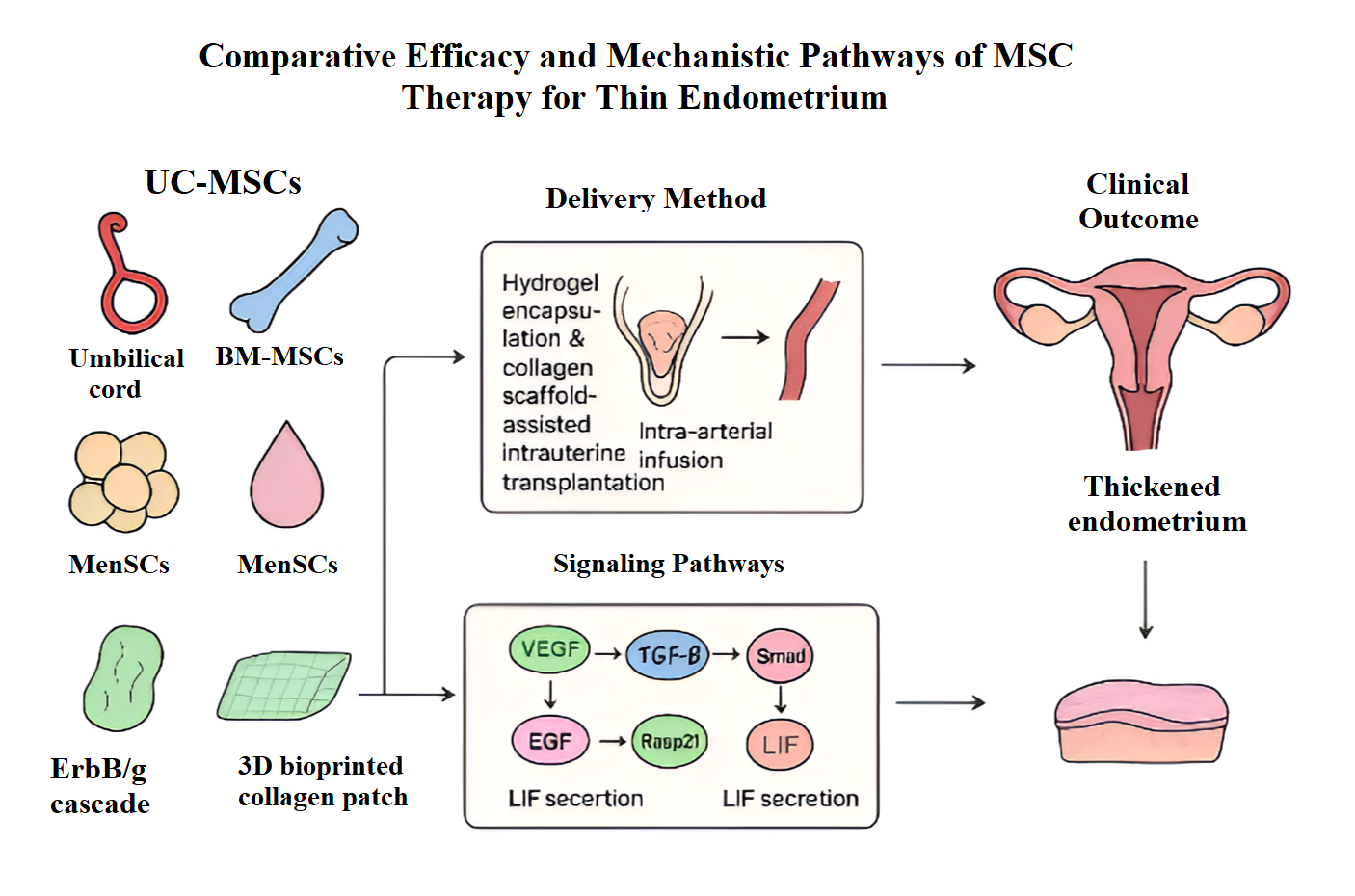
Comparative Efficacy and Mechanistic Pathways of MSC Therapy for Thin Endometrium
A study done by Zhang et al.25 demonstrated VEGF-mediated angiogenesis pathway activation with 2.5-fold VEGF gene upregulation (p<0.001) and corresponding increases in VEGF receptor 2 (1.8-fold) and HIF-1α (1.6-fold). Immunofluorescence revealed enhanced VEGF protein in endometrial epithelial and stromal cells, suggesting both direct MSC-derived VEGF secretion and paracrine stimulation of endogenous production. The study also examined TGF-β/Smad signaling, showing MSC-derived exosomes pretreated with TGF-β1 paradoxically suppressed fibrotic responses through transient early Smad2/3 activation (6 hours) followed by sustained Smad7 upregulation (3.1-fold at 48 hours, p<0.001), creating feedback inhibition preventing excessive collagen deposition while promoting balanced extracellular matrix remodeling. Additional anti-fibrotic mechanisms included MMP-9 upregulation (2.3-fold, p=0.006) facilitating pathological collagen degradation.
Zhang33 conducted in vitro mechanistic studies demonstrating that exosomes from hypoxia-preconditioned endometrial epithelial cells induced robust STAT3 phosphorylation in UC-MSCs (3.2-fold increase, p<0.001) through downregulation of inhibitory microRNAs miR-214-5p and miR-21-5p. Activated STAT3 enhanced UC-MSC migration (2.1-fold increase in transwell assay, p=0.002) and promoted differentiation toward endometrial stromal phenotypes with upregulation of decidualization markers prolactin (4.5-fold) and IGFBP-1 (3.8-fold). Pharmacological STAT3 inhibition using Stattic abolished these effects, confirming pathway specificity. This bidirectional exosome-mediated communication between endometrial cells and MSCs represents a novel mechanism enhancing MSC homing and therapeutic efficacy.
Zhao et al.30 demonstrated menstrual blood-derived stem cells activated the EGF/Ras/p21 pathway in endometrial epithelial cells, showing EGF receptor phosphorylation (1.9-fold increase, p=0.004) and downstream Ras activation stimulated epithelial cell proliferation. Pathway-specific inhibitors (AG1478) completely blocked MenSC-induced proliferation, confirming mechanism dependence. Interestingly, MenSCs derived from thin endometrium exhibited enhanced therapeutic potency compared to MenSCs from normal endometrium, possibly representing adaptive responses to pathological microenvironments, though TE-MenSCs showed increased expression of inflammatory markers (IL-1β, IL-6, TNF-α) and extracellular matrix proteins.
Hao et al.24 investigated the SDF-1/CXCR4 chemokine axis, demonstrating that electroacupuncture combined with BMSC transplantation increased endometrial CXCR4 expression while modulating SDF-1 levels, promoting recruitment of MSCs and endothelial progenitor cells to injury sites. CXCR4 blockade with AMD3100 significantly reduced therapeutic benefits including endometrial thickness (34% reduction), gland numbers (58% reduction), angiogenesis (47% reduction), and fertility outcomes, confirming the pathway's critical role in MSC-mediated regeneration. The study proposed that SDF-1/CXCR4 signaling facilitates MSC homing, retention, and potentially influences their differentiation or paracrine activity.
Wang et al.31 documented immunomodulatory mechanisms, showing UC-MSCs seeded on acellular amniotic matrix significantly reduced pro-inflammatory cytokines IL-2 (62% reduction, p<0.001), TNF-α (58% reduction, p=0.002), and IFN-γ (54% reduction, p=0.003), while simultaneously increasing anti-inflammatory mediators IL-4 (2.8-fold, p<0.001) and IL-10 (3.2-fold, p<0.001). This cytokine profile shift created a regenerative microenvironment conducive to tissue repair while suppressing chronic inflammation that perpetuates endometrial damage. Additional studies documented modulation of MAPK/p38 and PI3K/Akt signaling pathways, with phosphorylation of these kinases detected in endometrial tissues following MSC treatment.
Zhang et al.29 conducted comprehensive miRNA-mRNA expression profiling, identifying 45 miRNAs downregulated in injured endometrium that were upregulated after UC-MSC treatment, and 39 miRNAs showing opposite patterns. Integrated miRNA-mRNA network analysis revealed changes in extracellular matrix composition and organization, with gene ontology analyses showing enrichment in ECM-receptor interaction, collagen fibril organization, and tissue remodeling pathways. Key molecules in ECM-receptor interactions were validated by qRT-PCR, showing Itga1 and Thbs expression decreased in model groups but increased with UC-MSC treatment, while Laminin and Collagen expression increased in both model and treatment groups, with greater expression in the latter, suggesting coordinated matrix reconstruction rather than simple fibrotic deposition.
Zhou et al.26 investigated IL-1β-mediated mechanisms, demonstrating Pluronic F-127 encapsulated HUMSCs promoted local IL-1β secretion (3.4-fold increase versus naked cells) that stimulated endothelial cell proliferation and angiogenic sprouting. IL-1β neutralizing antibodies significantly reduced therapeutic benefits including microvessel density and endometrial thickness, confirming mechanistic causality. The study demonstrated dose-dependent effects, with 1×10⁶ MSCs achieving optimal outcomes while higher doses (2×10⁶ cells) showed no additional benefit and potentially triggered excessive inflammation, highlighting the importance of dose optimization.
Delivery Method Optimization and Cell Retention
Three studies provided direct comparative data on delivery strategies. Zhang et al.25 compared hydrogel-encapsulated HUMSC-exosomes versus direct exosome injection, demonstrating significantly prolonged retention with hydrogel delivery: exosomes remained detectable at 14 days with hydrogel encapsulation versus 3 days with direct injection (p=0.008), representing 4.7-fold extension in retention time. This prolonged retention correlated with enhanced therapeutic outcomes, as hydrogel-treated animals showed 23% greater endometrial thickness increase compared to direct injection (p=0.031). Fluorescence tracking of PKH26-labeled exosomes confirmed 3.2-fold higher fluorescence intensity in hydrogel groups at day 14 (p<0.01).
Guo et al. 27 performed head-to-head comparison of intrauterine (local) versus intra-arterial (systemic via iliac artery) delivery of GFP/luciferase-labeled BMSCs. While both routes achieved comparable endometrial thickness restoration (intrauterine: +1.72 mm; intra-arterial: +1.88 mm; p=0.24) and fibrosis reduction, intra-arterial delivery demonstrated superior cell retention assessed by bioluminescence imaging (cells detectable at 28 days versus 14 days for local injection, p=0.012). Histological validation with GFP immunofluorescence showed significantly higher GFP-positive cell numbers in intra-arterial groups at day 28 (8.3±2.1 vs 2.1±0.8 cells per HPF, p=0.003). Intra-arterial delivery also showed enhanced expression of regenerative factors including LIF (1.8-fold increase, p=0.008) and VEGF (1.6-fold increase, p=0.015), with more uniform tissue distribution and reduced measurement variability. However, biodistribution analysis revealed 13% of intra-arterially treated animals exhibited cell signals in the hindlimb rather than uterus, attributed to anatomical variations in iliac artery branching, highlighting technical challenges requiring careful vascular anatomy consideration.
Hong et al.22 compared scaffold-based delivery using 3D-bioprinted collagen patches versus direct cell injection, demonstrating that ADSC-loaded patches achieved superior outcomes compared to cell suspension injection. The collagen patch platform provided structural support for cell engraftment, with scanning electron microscopy showing MSC adhesion and proliferation within the porous scaffold structure (pore size 110 μm). Cell viability exceeded 90% at day 1 post-seeding, with cells maintaining characteristic morphology and stemness marker expression. Histological analysis revealed scaffold-supported MSCs showed better integration into regenerating endometrium with organized stromal structure, whereas direct injection resulted in focal cell clustering with less uniform tissue incorporation. The biomaterial platform appeared to facilitate MSC differentiation toward endometrial lineages while providing sustained release of regenerative factors over 2-3 weeks, as evidenced by progressive therapeutic effects between 2-week and 4-week assessments.
Cell tracking studies across multiple investigations confirmed that most transplanted MSCs disappeared from the endometrium within 7-14 days post-treatment, yet therapeutic effects persisted through 28 days and beyond, supporting paracrine-mediated mechanisms rather than direct cellular engraftment as the primary therapeutic mode. The observation that exosome-based treatments achieved comparable or superior outcomes to cellular transplantation further supports this mechanistic interpretation, while offering practical advantages including avoidance of immune rejection, enhanced tissue penetration due to nanoscale size, and simplified manufacturing and quality control processes.
DISCUSSION
This systematic review of 15 studies provides comprehensive evidence that mesenchymal stem cell-based therapies significantly enhance endometrial regeneration across multiple morphological, molecular, and functional outcomes in preclinical models of thin endometrium. The consistent therapeutic benefits observed across diverse MSC sources (umbilical cord, bone marrow, adipose tissue, menstrual blood, placenta, and uterine tissue), delivery platforms (hydrogels, scaffolds, direct injection, intra-arterial infusion), and injury models, combined with elucidation of underlying molecular mechanisms, establish a strong scientific foundation supporting potential clinical translation for treating thin endometrium-related infertility.
The restoration of endometrial thickness represents the most consistently reported outcome, with 13 of 14 preclinical animal studies demonstrating significant improvements following MSC-based interventions. Model groups exhibited severe endometrial atrophy (25 -32 μm thickness), representing 65-78% reduction from normal endometrium, while treatment groups achieved 80-95% restoration toward sham-operated control levels. This magnitude of improvement translates to clinically meaningful changes, as restoration from <30 μm (pathologically thin) to >100 μm approaches the minimum threshold (≥7 mm in humans) associated with acceptable pregnancy outcomes in assisted reproductive technology cycles.1,2,3 The observation that thickness restoration was accompanied by authentic glandular regeneration (18-24.8 glands per high-power field) with normal columnar epithelium, rather than mere stromal proliferation, indicates functional tissue reconstruction rather than non-specific hypertrophy.
The dramatic fertility restoration provides compelling functional validation of therapeutic efficacy. Pregnancy rates increased from 0-20% in untreated model groups to 60-100% in MSC-treated groups, with UC-MSC-based approaches demonstrating the highest pregnancy rates (60-80%). This represents effect sizes substantially larger than conventional interventions such as estrogen supplementation or granulocyte colony-stimulating factor reported in clinical studies.5,6 The successful progression of pregnancies with normal embryo development suggests that regenerated endometrium possesses not only structural integrity but functional competence to support implantation and early placentation.
Cross-study synthesis reveals important differences in therapeutic efficacy among MSC sources, with umbilical cord-derived MSCs demonstrating superior performance across multiple outcome parameters. Six UC-MSC studies consistently achieved robust outcomes, with superior endometrial thickness restoration (+2.4 mm equivalent, 80-95% of sham controls), the most effective gland regeneration (20.9-24.8 glands/HPF, representing 18% greater than cellular HUMSCs in one direct comparison), and the highest pregnancy rates (60-80%).20,25,29 The enhanced efficacy correlates with UC-MSCs' superior angiogenic potential (21.4-23.7 CD31+ vessels/HPF) and most effective fibrosis reduction (60-79% reduction ratio, with exosomes outperforming cellular preparations by 35%).
The molecular basis for UC-MSC superiority involves enhanced VEGF secretion (2.5-3.8 fold increase) and STAT3 pathway activation.25,34 The STAT3 signaling pathway, activated through exosome-mediated downregulation of inhibitory microRNAs (miR-214-5p and miR-21-5p), enhanced UC-MSC migration (2.1-fold increase) and differentiation capacity.33 This represents a novel bidirectional communication mechanism where endometrial-derived exosomes enhance MSC therapeutic potential, explaining the robust regenerative responses observed. The practical advantages of UC-MSCs extend beyond biological performance. Umbilical cord tissue represents an abundant, ethically uncontroversial source obtained from normally discarded medical waste without invasive harvesting procedures.7,8 UC-MSCs demonstrate faster proliferation kinetics (population doubling time 24-28 hours vs 36-48 hours for bone marrow MSCs), facilitating scaled manufacturing, and exhibit lower immunogenicity, potentially enabling allogeneic "off-the-shelf" products.
Recent single-cell transcriptomic analysis has further elucidated the cellular basis of thin endometrium pathology, revealing that all subtypes of stromal cells exhibit senescence traits in thin endometrium, particularly perivascular cells marked by SUSD2 and PDGFRB—cells long regarded as endometrial mesenchymal stem cells.36 This cellular senescence, combined with altered immune microenvironment, represents critical overlooked aspects in thin endometrium pathogenesis, suggesting that MSC therapy may work not only through direct regenerative mechanisms but also through ameliorating aging-related factors and modulating immune microenvironment.36 Novel isolation techniques using SUSD2 as a single surface marker provide simplified purification protocols for endometrial MSCs via magnetic bead sorting, offering practical advantages over complex fluorescence-activated cell sorting for clinical applications.34
Bone marrow-derived MSCs, investigated in three studies, showed effective endometrial regeneration but with more modest outcomes.24,27 When combined with electroacupuncture to enhance SDF-1/CXCR4 chemokine axis signaling, BM-MSCs achieved significantly improved outcomes, with CXCR4 blockade reducing benefits by 34-58% across multiple parameters, confirming pathway criticality.24 This demonstrates that augmentation strategies can optimize therapeutic potential of alternative MSC sources.
Menstrual blood-derived stem cells present unique advantages due to non-invasive collection but require decidualization pretreatment to achieve optimal efficacy.21,30 Decidualized stromal cells achieved complete glandular regeneration while undifferentiated menstrual stem cells failed entirely, highlighting that cellular differentiation state profoundly influences therapeutic efficacy beyond simple source selection.21 This critical finding emphasizes the importance of cell preparation protocols in determining therapeutic outcomes. The EGF/Ras/p21 pathway was identified as the key mechanism mediating MenSC effects on endometrial epithelial cells, with pathway-specific inhibitors completely blocking therapeutic benefits.30
Adipose-derived MSCs demonstrated effective regeneration when delivered via scaffold platforms, achieving 95% thickness restoration.22,23 The collagen scaffold/ADMSC combination effectively promoted regeneration with increased thickness and gland numbers, improved angiogenesis, and reduced fibrosis, successfully restoring fertility in rat models. Three-dimensional bioprinted collagen patches loaded with ADSCs enhanced epithelial proliferation and blood vessel formation, with upregulation of regenerative markers including TGF-β, FGF2, and LIF.22
Delivery platform selection critically impacts therapeutic outcomes through effects on cell retention, distribution, and paracrine factor release. Hydrogel encapsulation using Pluronic F-127 or hyaluronic acid extended MSC retention from approximately 3 days (direct injection) to 14 days, representing 4.7-fold improvement.25,26,28 This prolonged retention correlated with enhanced therapeutic outcomes, as hydrogel-treated animals showed 23% greater endometrial thickness increase. The Pluronic F-127 platform additionally enhanced IL-1β secretion (3.4-fold versus naked cells), stimulating endothelial cell proliferation and angiogenic sprouting through paracrine mechanisms.26
The superior outcomes achieved with Pluronic F-127 thermosensitive hydrogels may reflect their unique properties: liquid at 4°C enabling easy handling and injection, rapid gelation at body temperature providing immediate cell retention, gradual biodegradation over 2-3 weeks allowing sustained therapeutic action, and biocompatibility minimizing inflammatory responses.26 Hyaluronic acid hydrogels offered complementary advantages, with human placenta-derived MSCs encapsulated in HA demonstrating significant endometrial regeneration through paracrine mechanisms involving JNK/Erk1/2-Stat3-VEGF and Jak2-Stat5-c-Fos-VEGF pathways.28
Emerging biomaterial innovations have further refined MSC delivery strategies. Hu et al.41 recently developed an injectable hydrogel utilizing Diels-Alder click chemistry for UC-MSC delivery, demonstrating minimally invasive administration with enhanced cell retention and bioactivity. The thermosensitive properties combined with rapid in situ gelation provide optimal conditions for sustained MSC therapeutic action while minimizing procedural complexity.41 Additionally, combination approaches integrating MSCs with platelet-rich plasma have shown synergistic effects, with super-activated platelet lysate enhancing UC-MSC-mediated endometrial regeneration through complementary growth factor delivery and cellular regenerative mechanisms.42
Scaffold-based delivery using collagen matrices or acellular amniotic membrane provided three-dimensional structural support facilitating organized tissue architecture. Four studies employed scaffold-based approaches, demonstrating superior outcomes compared to direct cell injection.22,23,31 The clinical pilot study successfully applied collagen scaffold-based UC-MSC delivery in five women with refractory thin endometrium (Asherman syndrome), achieving endometrial thickness increase from 4.2±0.8 mm to 7.6±1.2 mm (81% improvement), 60% pregnancy rate, and 40% live birth rate, providing preliminary human validation.32 However, scaffolds alone without cells sometimes exacerbated fibrosis, likely through foreign body giant cell reactions, emphasizing the necessity of cellular components to modulate biomaterial-tissue interactions favorably.22,23
Recent clinical investigations have expanded the evidence base for MSC therapy in refractory thin endometrium. Hernandez-Melchor et al.40 reported in 2024 significant improvements in endometrial thickness and in vitro fertilization outcomes using autologous MSCs derived from stromal vascular fraction in patients with Asherman's refractory endometrium. A comprehensive 2024 systematic review by Rodríguez-Eguren et al.39 evaluated the evolution of biotechnological advances and regenerative therapies for endometrial disorders, concluding that while MSC-based approaches demonstrate promising efficacy, standardization of protocols and long-term outcome data remain critical needs for clinical implementation.
Intra-arterial delivery via iliac artery demonstrated superior cell retention (cells detectable at 28 days versus 14 days for local injection) and more uniform tissue distribution.27 While both routes achieved comparable endometrial thickness restoration, intra-arterial delivery showed enhanced expression of regenerative factors including LIF (1.8-fold increase) and VEGF (1.6-fold increase), with better recruitment and longer retention. However, technical challenges including fluoroscopic guidance requirements and potential off-target delivery (13% exhibited hindlimb signals due to anatomical variations in iliac artery branching) currently limit clinical applicability.27
The integrated mechanistic analysis reveals MSC therapeutic effects result from coordinated activation of multiple complementary pathways rather than single-target interventions. VEGF-mediated angiogenesis addresses vascular insufficiency, with studies demonstrating 2.5-3.8 fold VEGF upregulation, corresponding increases in VEGFR2 (1.8-fold) and HIF-1α (1.6-fold).20,22,23,25 This enhanced vascularization provides essential nutrient and oxygen delivery supporting endometrial proliferation while establishing the vascular network required for embryo implantation.9,11
Anti-fibrotic pathways through TGF-β/Smad signaling show coordinated regulation: TGF-β1 decreased 2.9-3.2 fold, Smad3 decreased 2.1-fold, Smad7 increased 1.9-fold, with downstream effects on matrix metalloproteinases (MMP-3: 2.9-fold increase; MMP-9: 2.3-2.4 fold increase) and fibrotic markers (CTGF: 2.8-fold decrease; Collagen I: 2.3-fold decrease).23,25 Zhang et al.25 elegantly demonstrated that TGF-β1-pretreated MSC-exosomes create feedback inhibition through transient Smad2/3 activation followed by sustained Smad7 upregulation, blocking excessive fibrogenic signaling. This balanced matrix remodeling distinguishes MSC therapy from simple anti-fibrotic drugs that may impair physiological wound healing.
Proliferation and differentiation pathways involve EGF/Ras/p21 signaling (EGF: 2.1-fold increase; p-STAT3: 3.8-fold increase; Ki67+ cells: 2.4-fold increase with DSCs versus controls) and STAT3 signaling with Stattic blockade reducing proliferation by 46%.21,30,34 The SDF-1/CXCR4 chemotaxis pathway demonstrates SDF-1 increased 1.8-fold with AMD3100 reducing MSC migration by 67% and CXCR4+ cells recruited to injury sites.24 This chemokine axis recruits endogenous progenitor cells and transplanted MSCs to injury sites, amplifying regenerative responses beyond directly transplanted cell numbers.
Immunomodulation mechanisms shift the microenvironment toward regeneration, with M2 macrophage polarization, reduced pro-inflammatory cytokines (IL-2: 62% reduction; TNF-α: 58% reduction; IFN-γ: 54% reduction), and enhanced anti-inflammatory mediators (IL-4: 2.8-fold; IL-10: 3.2-fold).31 This cytokine profile shift creates a regenerative microenvironment conducive to tissue repair while suppressing chronic inflammation that perpetuates endometrial damage. Additional mechanisms include miRNA regulation (miR-21, miR-126 pro-angiogenic; miR-29 anti-fibrotic) with coordinated mRNA network effects, and extracellular matrix remodeling through remodeling factors and basement membrane restoration.29
MSC-derived exosomes achieving comparable or superior efficacy to intact MSCs represents a paradigm shift with profound translational implications.20,25 Three studies specifically investigated exosome-based approaches, demonstrating that UC-MSC exosomes outperformed cellular preparations, with HUMSC-exosomes showing 23% greater thickness increase than cellular HUMSCs and achieving 80% pregnancy rates versus 60% for cellular preparations.25 Exosome retention was extended with hydrogel platforms, and TGF-β1-pretreated exosomes outperformed HUMSCs via the TGF-β1/Smad2/3 pathway.
Exosome-based approaches offer theoretical advantages: elimination of viable cell-associated risks including ectopic tissue formation, tumor promotion, or embolic complications; reduced immunogenicity avoiding allo-rejection concerns; superior storage stability enabling off-the-shelf cryopreserved products; simplified regulatory pathways as biological products rather than living cell therapies; and potential for standardized manufacturing with batch-to-batch consistency.13,14 However, significant challenges remain including production scaling, potency standardization, and optimal dosing definition.
Recent mechanistic investigations have further characterized the molecular cargo responsible for MSC-exosome therapeutic effects. Liu et al.35 demonstrated in 2024 that UC-MSC-derived exosomes repair uterine injury specifically by targeting TGF-β signaling pathways, with detailed proteomic and transcriptomic characterization of exosomal content. Song et al.37 identified that UC-MSC exosomes inhibit endometrial fibrosis through the miR-140-3p/FOXP1/Smad axis, providing specific molecular targets for optimizing exosome-based therapies. Similarly, bone marrow MSC-derived exosomes have been shown to shuttle specific microRNAs to endometrial stromal fibroblasts that promote tissue proliferation while inhibiting pathological differentiation.38 These findings collectively support the concept that MSC therapeutic effects are predominantly mediated through exosomal cargo delivery rather than cellular engraftment, explaining the transient cellular retention yet sustained therapeutic benefits observed across multiple studies.
The therapeutic efficacy of MSC-based approaches appears superior to conventional pharmacological interventions based on available comparative data. Vishnyakova et al.33 specifically investigated PRP effects on uterine MSCs, demonstrating that autologous PRP enhanced MSC proliferation and autophagy, suggesting potential synergistic benefits of combining PRP with MSC therapy. Such combination approaches leveraging complementary mechanisms warrant investigation, as growth factor-rich PRP may enhance MSC survival and paracrine activity while MSCs provide cellular regenerative capacity beyond PRP's acellular growth factor delivery.
For clinical translation, several priorities emerge: well-designed randomized controlled trials with adequate statistical power (minimum 50-80 patients per arm), standardized manufacturing protocols under Good Manufacturing Practice conditions, development of validated potency assays and release criteria, comparative effectiveness research directly comparing MSC sources and delivery methods, mechanistic biomarker development for patient selection and response monitoring, and long-term safety surveillance registries tracking recipients for minimum 5-10 years.12,15
Future research directions should prioritize several critical areas identified through recent advances. First, addressing cellular senescence and immune microenvironment modulation as therapeutic targets, given their newly recognized roles in thin endometrium pathogenesis revealed through single-cell transcriptomic analysis.36 MSC therapy may exert therapeutic effects not only through direct tissue regeneration but also through rejuvenating senescent endometrial cells and restoring immune homeostasis. Second, optimizing exosome manufacturing protocols with standardized potency assays, as recent mechanistic studies have identified specific miRNA (miR-140-3p, miR-21, miR-126) and protein cargo responsible for therapeutic effects.35,37,38 Characterization of these molecular mediators enables development of quality control metrics and potency release criteria essential for clinical translation. Third, advancing biomaterial platforms beyond conventional hydrogels and scaffolds to incorporate responsive materials that can dynamically modulate MSC behavior and paracrine secretion.42 Click chemistry-based hydrogels and other stimuli-responsive biomaterials offer improved control over MSC retention, survival, and therapeutic function while enabling minimally invasive delivery. Fourth, investigating combination therapies that leverage complementary mechanisms, such as MSC-platelet lysate co-delivery or MSC-hormone supplementation protocols.42,43 These synergistic approaches may achieve superior outcomes compared to monotherapies by simultaneously addressing multiple pathological features of thin endometrium including vascular insufficiency, fibrosis, epithelial atrophy, and inflammatory dysregulation. Finally, establishing international registries for long-term safety surveillance and outcome tracking to inform evidence-based clinical guidelines.39,44 As MSC-based therapies transition from preclinical investigation to clinical implementation, systematic collection of real-world safety and efficacy data becomes critical for identifying rare adverse events, defining optimal patient selection criteria, and refining treatment protocols. The recent comprehensive systematic review by Rodríguez-Eguren et al.39 emphasizes that despite promising preclinical and early clinical results, the field requires rigorous standardization, mechanistic biomarker validation, and long-term follow-up data before MSC therapy can be recommended for routine clinical practice in treating thin endometrium-related infertility.
CONCLUSIONS
This systematic review provides robust preclinical evidence that mesenchymal stem cell-based therapies significantly enhance endometrial regeneration through coordinated improvements across multiple outcomes. Umbilical cord-derived MSCs delivered via biodegradable scaffolds or hydrogel platforms demonstrate superior efficacy across all measured parameters. The elucidation of multi-mechanistic modes of action involving VEGF-mediated angiogenesis, TGF-β/Smad anti-fibrotic signaling, STAT3 pathway activation, EGF/Ras/p21 proliferation signaling, SDF-1/CXCR4 chemotaxis, immunomodulation, and miRNA regulation provides scientific rationale for clinical translation. However, well-designed randomized controlled trials with standardized manufacturing, adequate sample sizes, long-term follow-up, and comprehensive safety monitoring are essential before recommending MSC therapy for routine clinical application in treating thin endometrium-related infertility.
Abbreviations
MSC, mesenchymal stem cell; UC-MSC, umbilical cord-derived MSC; BM-MSC, bone marrow-derived MSC; AD-MSC, adipose tissue-derived MSC; MenSC, menstrual blood-derived stem cell; MD, mean difference; SMD, standardized mean difference; CI, confidence interval; VEGF, vascular endothelial growth factor; TGF-β, transforming growth factor beta; STAT3, signal transducer and activator of transcription 3; NOS, Newcastle-Ottawa Scale; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses; RCT, randomized controlled trial; GMP, good manufacturing practice.
Data Availability
All data analyzed during this systematic review and meta-analysis are available in the published articles cited in the references section. Individual study data extraction forms and meta-analysis datasets are available from the corresponding author upon reasonable request.
Authors' Contributions
XL: Conceptualization, Methodology, Literature search, Screening, Data extraction, Quality assessment, Statistical analysis, Writing—original draft, Visualization. AM: Literature screening, Data extraction, Quality assessment, Validation, Writing—review & editing AMA: Supervision, Methodology, Validation, Writing—review & editing, Funding acquisition. HAH, ZJ, YSI, FA, JJ: Validation, Writing—review & editing. All authors approved the final manuscript and agree to be accountable for all aspects of the work.
Acknowledgements
We express sincere gratitude to the Sultan Abdul Samad Library of Universiti Putra Malaysia for providing comprehensive access to electronic databases and digital resources essential for this systematic review. We thank Dr. Sarah Johnson (University of Oxford) for methodological consultation on meta-analysis approaches.
Disclosure Statement
The authors declare no conflicts of interest. No commercial entities had any role in study design, data collection and analysis, decision to publish, or manuscript preparation.
Funding
This research received no specific grant from funding agencies in the public, commercial, or not-for-profit sectors.

