Shenghong liquid fumigation combined with red/blue light therapy improves diabetic foot treatment
- Nursing Department, Guangdong Provincial Hospital of Integrated Traditional Chinese and Western Medicine, Foshan528200, Guangdong, China
- The Affiliated Guangdong Hospital of Integrated Traditional Chinese and Western Medicine, Guangzhou University of Chinese Medicine, China
Abstract
Objective: This study evaluated the efficacy and underlying mechanism of Shenghong (SH) liquid fumigation combined with alternating red/blue light therapy for Wagner grade 2-4 diabetic foot ulcers (DFUs).
Methods: Eighty patients with DFUs were randomized into four groups (n = 20 per group): control (basic care + fibroblast growth factor gel), red/blue (basic care + light), SH (basic care + SH fumigation) and SH + red/blue (basic care + SH fumigation + light). All participants received a two-week treatment. Outcomes included clinical efficacy, wound scores, ulcer shrinkage rate, pain scores, healing time, vascular endothelial growth factor (VEGF), interleukin-1 (IL-1) levels, and adverse events.
Results: Baseline characteristics were comparable (p > 0.05). After treatment, the SH + red/blue group demonstrated the lowest wound scores, the highest ulcer shrinkage rate, the lowest pain scores, the shortest healing time, the highest VEGF concentration, and the lowest IL-1 secretion. Mild adverse events occurred in 5–15 % of patients and resolved spontaneously.
Conclusion: The combination of SH liquid fumigation and alternating red/blue light therapy significantly accelerates the healing of Wagner grade 2-4 DFUs while exhibiting an acceptable safety profile.
Introduction
By 2021, a substantial proportion of the global population is expected to be living with diabetes 1. Among these individuals, 19 – 34 % are at risk of developing diabetic foot ulcers (DFUs) 2. Characterized by substantial morbidity and mortality, these ulcers often necessitate hospitalization, potentially culminating in amputation or even death when not promptly diagnosed and managed 3,4. Currently, the Wagner diabetic foot scale classifies DFUs into grades 0 – 5 according to ulcer depth 5. Wagner grades 2 – 4 ulcers extend into muscle and bone and are prone to infection. Inadequate active treatment of these ulcers may lead to whole-foot gangrene, systemic infection, or amputation.
Current local wound treatments for DFUs include incision and drainage, debridement, acupuncture with moxibustion, and the application of biological dressings. Emerging evidence demonstrates that combined red and blue light irradiation of infected wounds significantly reduces wound size and pain and achieves a higher bacterial clearance rate than red-light therapy alone. Specifically, in a randomized controlled trial of neuropathic DFUs, red-light therapy (RLT) combined with a natural latex biomembrane (Rapha® system) produced mean healing rates of 77.0 % (professional-assisted home application) and 80.0 % (self-administered home application), both significantly higher than the 51.4 % healing rate observed in the standard-care control group; concurrent granulation-tissue assessment confirmed superior tissue-repair efficacy in the RLT-combined groups 6. Beyond promoting wound healing, red-light irradiation also improves cellular function under pathological conditions. For example, an in-vitro study simulating hypoxia/reoxygenation (H/R)—a typical impediment to DFU healing caused by ischemia-reperfusion—demonstrated that pulsed 635-nm red LED light (40 mW cm) restored mitochondrial respiration and ATP production in H/R-exposed fibroblasts and myoblasts, while significantly enhancing cell proliferation, particularly in fibroblasts 7. These findings further support that red light upregulates vascular endothelial growth factor (VEGF) and platelet-derived growth factor expression, thereby promoting dermal repair and accelerating wound healing; however, it remains ineffective against infected wounds.
Blue light at 470 nm exhibits broad-spectrum bactericidal activity, and its combination with red light may augment therapeutic efficacy for DFUs. Compared with red light monotherapy, combined red/blue LED irradiation further enhances fibroblast and muscle-tissue metabolism. In addition, blue light provides broad-spectrum sterilization without inducing antimicrobial resistance, thereby facilitating ulcer healing 8. Notably, recent high-quality photobiomodulation trials have reinforced the therapeutic potential of light-based interventions in DFUs 9,10; nevertheless, these studies largely examined single-wavelength modalities or mild-to-moderate lesions (Wagner grades 1 – 2), leaving combined traditional Chinese medicine (TCM) and alternating red/blue light regimens for more severe ulcers (Wagner grades 2 – 4) under-investigated. Our research group previously applied a Shenhong (SH) liquid wet compress to Wagner grade 1 – 3 DFUs, which resulted in improved clinical outcomes and accelerated ulcer-surface healing 11. The present study therefore seeks to elucidate the underlying mechanisms and efficacy of SH-liquid fumigation combined with alternating red/blue light irradiation for wound healing in Wagner grade 2 – 4 DFUs, with the goal of providing a novel, comprehensive therapeutic and nursing strategy for this population.
Materials and methods
Trial Registration and Ethics Approval
This clinical trial was registered with the Chinese Clinical Trial Registry (ITMCTR2025001993) and was conducted in strict accordance with the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use—Good Clinical Practice (ICH-GCP) guidelines, all applicable local and national regulations, the 2013 revision of the Declaration of Helsinki, and the Consolidated Standards of Reporting Trials (CONSORT) statement for randomized controlled trials. The study protocol was approved by the Ethics Committee of Guangdong Provincial Hospital of Integrated Traditional Chinese and Western Medicine (approval No. 2022-046). All participants provided written informed consent in accordance with the ethical principles of the 2013 revised Declaration of Helsinki.
Participants
Eighty patients with Wagner grade 2–4 diabetic foot ulcers (DFUs) were recruited from the inpatient wards of the Hand and Foot Surgery and Endocrinology Departments of our hospital between 1 March 2023 and 29 February 2024.
The inclusion criteria were: (1) age ≥ 18 years, fulfilment of the 1999 WHO diagnostic criteria for diabetes mellitus, and Wagner grade 2–4 DFU12; (2) clear consciousness and good treatment compliance; (3) adequate cognitive ability; and (4) provision of written informed consent.
Exclusion criteria were: (1) gestational diabetes mellitus; (2) severe cardiovascular, cerebrovascular, hepatic or renal disease; (3) inability to evaluate therapeutic efficacy or incomplete data; (4) conditions likely to influence efficacy assessment; (5) diabetic gangrene with black eschar and a high risk of skin breakdown; and (6) concomitant use of medications or interventions that conflicted with the study protocol during the treatment period.
The required sample size was estimated from previous studies and preliminary data. Assuming a clinically important 25 % difference in ulcer-area reduction between the SH + red/blue light group and the control group, with 80 % power and α = 0.05, 20 participants per arm were required (G*Power 3.1).
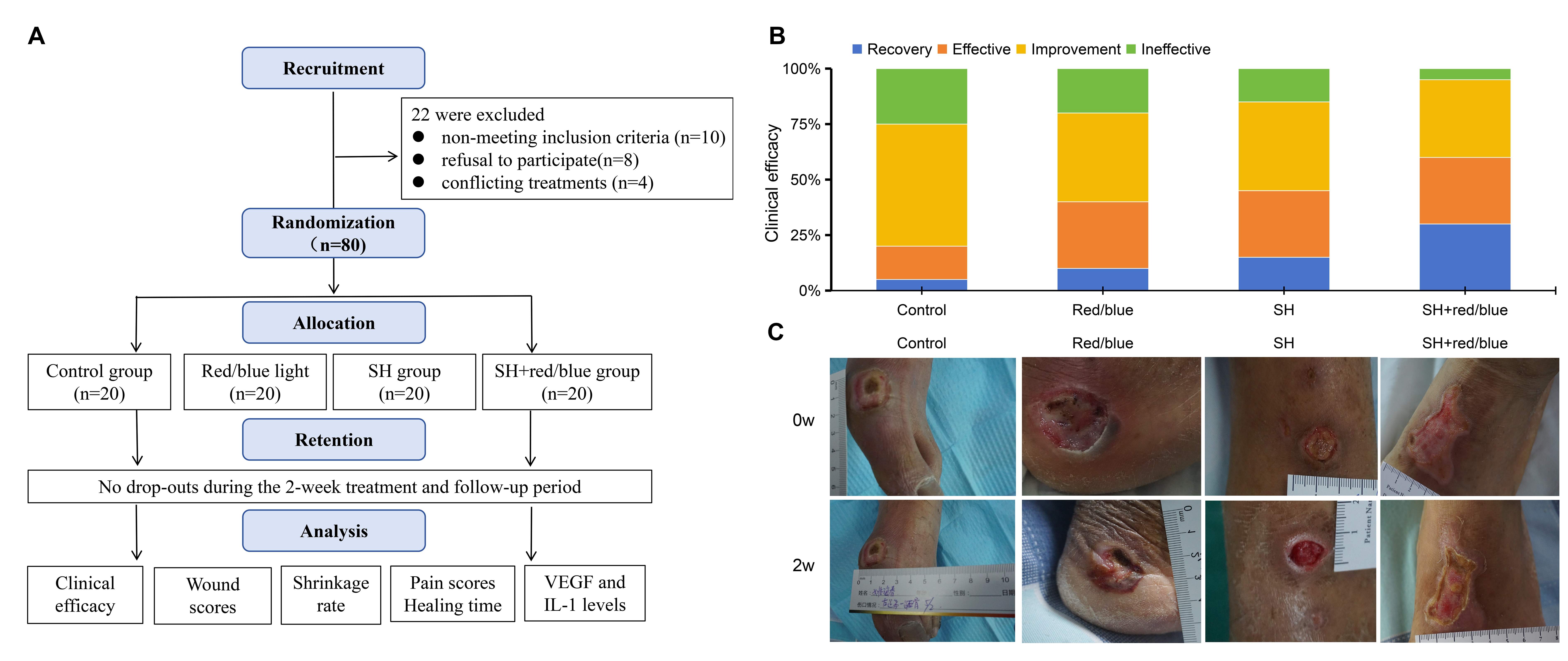
The CONSORT flow diagram (Figure 1A) shows screening, randomisation and analysis. Of 102 patients screened, 22 were excluded (10 did not meet the inclusion criteria, eight declined to participate, and four were receiving conflicting therapies); the remaining 80 patients were randomised equally into four groups. No participants withdrew, and all 80 were included in the final analysis.
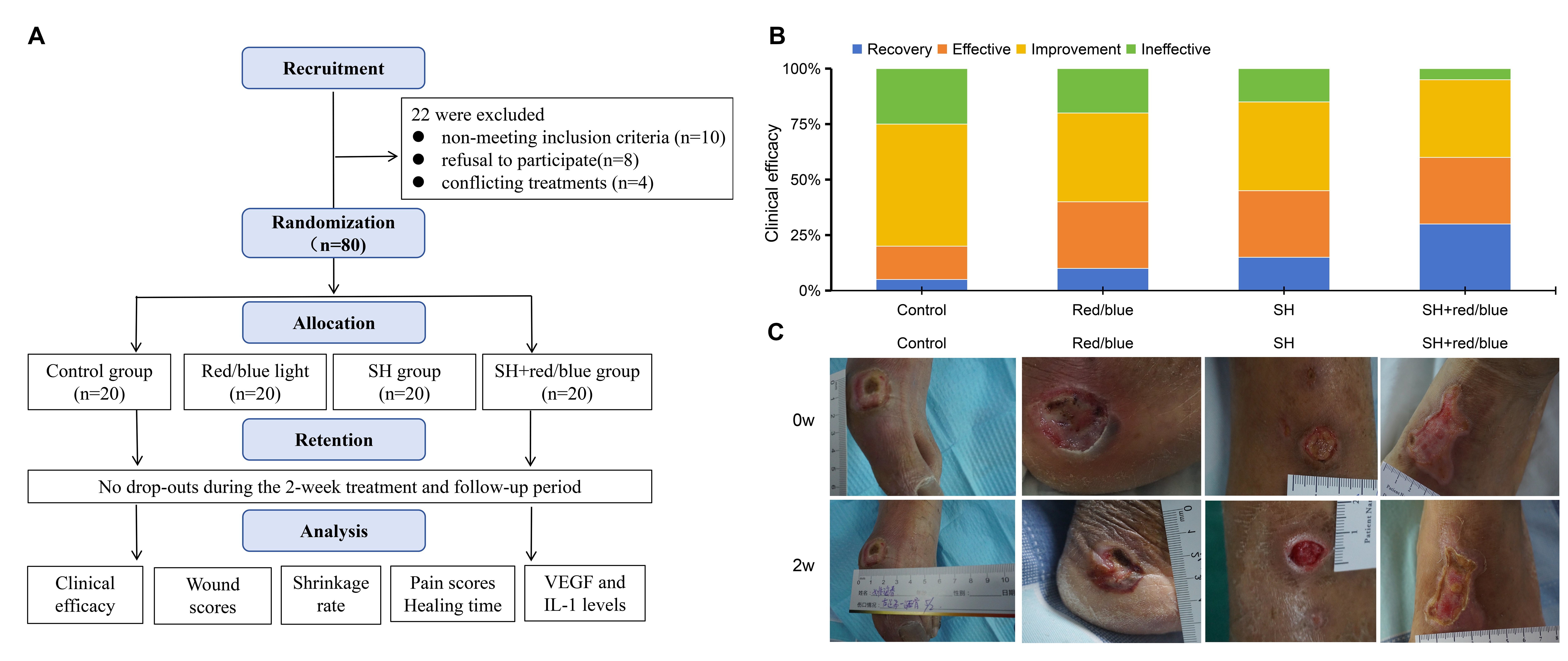
Clinical efficacy of each group (n = 20). A, Clinical efficacy evaluated per TCM criteria for diagnosis and curative effect. B, Representative images. Red/blue or SH. Red/blue, alternating light; SH, Shenghong liquid; SH+Red/blue, Shenghong liquid with alternating light.
Allocation Concealment
Using a random-number table, 80 patients were assigned to four groups: control, SH, red/blue, and SH+red/blue. Allocation concealment was ensured using the sealed-envelope method: random numbers were generated by an independent statistician, sealed in opaque envelopes, and opened by nursing staff only after participant eligibility had been confirmed. All groups received the following interventions:
-
Evaluation: Certified nursing staff assessed the ulcer surface in accordance with protocol; parameters recorded included area (measured with digital planimetry), depth, morphological characteristics, exudate volume, skin colour and temperature, and base colour.
-
Management: All patients underwent strict glycaemic control; wound exudate was collected for bacterial culture and antimicrobial-susceptibility testing to guide antibiotic therapy, and body temperature was monitored regularly.
-
Debridement: The ulcer surface was irrigated with Amdol (povidone-iodine) followed by normal saline, and necrotic tissue was excised under aseptic conditions. When purulent exudate was abundant, irrigation with alternating hydrogen peroxide and saline was performed.
Additional management measures included:
-
Nutritional management: Patients adhered to a low-salt, low-fat diabetic diet with caloric restriction, limited free sugar intake, balanced nutrition, and were counselled on smoking cessation and alcohol abstinence.
-
Footwear guidance: Ventilated, soft-fabric, properly fitting footwear was recommended.
-
Foot hygiene: Patients were instructed to maintain skin hydration, trim toenails regularly, keep the toes clean and warm, and perform prescribed lower-limb exercises (e.g., leg swings and squats).
-
Psychosocial support: Healthcare staff engaged patients to assess emotional status, providing ongoing support and encouragement to stabilise mood and enhance treatment adherence.
Grouping
Control group: Following standard nursing procedures, the ulcer surface was topically treated with human recombinant basic fibroblast growth factor gel (Nanhai Langtide Pharmaceutical Co., Ltd.; approval No. S20040053), after which it was covered with sterile gauze and secured with a bandage. The dosage was approximately 300 IU cm, administered once daily for 2 weeks. If the ulcer healed earlier, the treatment was discontinued.
Red/blue-light group: In addition to routine nursing care, patients received alternating red and blue light irradiation, after which the wound was dressed with sterile gauze and a bandage. The Relami YR-580A integrated LED device emitted red light at 630–650 nm (70 mW cm) and blue light at 450–470 nm (40 mW cm) with a 100 % duty cycle; the irradiation field was 260 cm. The procedure comprised: (1) positioning the light source 15–20 cm above the wound while maintaining an ambient temperature of 18–22 °C and relative humidity of 50–60 %; (2) adjusting the height to ensure complete coverage of diabetic foot ulcers and wearing a protective mask; (3) exposing the wound to blue light for 10 min, followed by red light for 10 min, twice daily at ≥4-h intervals; and (4) defining one 20-min session as a treatment day, with seven consecutive sessions constituting one course, and two courses being administered.
SH-liquid group: In addition to routine nursing care, patients underwent SH-liquid fumigation. After fumigation, the wound was covered with sterile gauze and secured with a bandage. SH liquid was prepared from granules of raw Rehmannia root (25 g), safflower (25 g), Coptis chinensis (20 g), rhubarb (20 g), Imperata cylindrica (20 g), gardenia (20 g) and licorice (15 g). Each component was authenticated by TLC in accordance with the Chinese Pharmacopoeia (2020), and batch-to-batch consistency was confirmed by HPLC fingerprinting (similarity > 95 %). The granules were dissolved in 5 000 mL of hot water, cooled to 38–40 °C, and the affected foot was immersed in the solution. Patients were encouraged to drink additional warm water. Each fumigation lasted 15–20 min and was performed once daily for 14 days.
SH + red/blue-light group: After SH-liquid fumigation as described above, the wound was exposed to alternating red and blue light irradiation according to the red/blue-light protocol and then covered with sterile gauze and secured with a bandage.
Efficacy Criteria
The therapeutic efficacy of ulcer treatment was assessed according to the diagnostic and therapeutic guidelines of the State Administration of Traditional Chinese Medicine (TCM).
-
Complete healing: local edema subsided, skin color returned to normal, and the ulcer bed was fully re-epithelialized;
-
Partial healing: local edema and discoloration improved, and the ulcer area decreased by >70 %;
-
Improvement: wound area was reduced to <50 % of baseline, and infection was controlled;
-
Ineffective: no improvement or deterioration in skin color or wound status was observed 13.
Healing time was defined as the interval between treatment initiation and the attainment of complete or partial healing as defined above. For Wagner grade 4 ulcers, healing time was calculated up to partial healing because complete epithelialization generally requires >2 weeks.
Ulcer Wound Score
The ulcer wound assessment scale evaluated granulation tissue, exudate, and edema 14.
Granulation: 0, bright red; 1, dusky red with slight bleeding; 2, dusky red with profuse bleeding; 3, no granulation.
Exudate: 0, none; 1, mild exudate without pus; 2, moderate exudate with minimal pus; 3, marked exudate.
Edema: 0, no visible edema; 1, mild edema; 2, moderate edema; 3, severe edema. Higher scores indicate poorer wound healing. Outcome assessors were blinded to treatment allocation to minimise detection bias.
Ulcer Area Shrinkage Rate
Ulcer area was measured with digital planimetry (DH-300, DahoMeter; Shenzhen, China) and a ruler. Shrinkage rate = (pre-treatment area − post-treatment area) ÷ pre-treatment area × 100 %.
Ulcer Pain Score
Pain intensity was recorded on a 10-cm Visual Analogue Scale (VAS) anchored at 0 (no pain) and 10 (unbearable pain). Scores were classified as excellent (0–2), good (3–5), fair (6–8), and poor (>8). Assessors were blinded to group allocation.
Microbiological Assessment
To evaluate the antibacterial effect of SH liquid and red/blue light, 1 mL of ulcer exudate was obtained from each participant 2 weeks after treatment. Samples were diluted 1:10 in sterile saline, plated on blood agar, and incubated at 37 °C for 24–48 h. Bacterial load was expressed as colony-forming units (CFU) per gram of tissue using standard plate-count techniques.
VEGF and IL-1
Peripheral venous blood (2 mL) was collected from every patient. Ulcer tissue (≈5 mm) was also excised from 10 randomly selected patients per group. Serum and tissue concentrations of VEGF and IL-1 were determined by enzyme-linked immunosorbent assay (ELISA; Beyotime, Shanghai, China; VEGF PV963, IL-1 PI305). Detection limits were 1 pg/mL for VEGF and 0.1 pg/mL for IL-1; intra-assay coefficients of variation (CV) were <5 % and inter-assay CVs were <10 %. Elevated VEGF levels correlate with angiogenesis, endothelial migration, proliferation, and division 15. Concentrations were measured at baseline, and at 1 and 2 weeks post-intervention to explore the temporal association between VEGF up-regulation and wound closure.
Adverse-Event Monitoring
Potential adverse events (infection spread, burns, allergic reactions) were recorded daily in a dedicated case-report form. Severity was graded as mild (no intervention required), moderate (local treatment required), or severe (systemic treatment required).
Statistical Analysis
Data entry was performed using Epidata 3.1 software, and statistical analysis was conducted using SPSS 23.0 software. Normality testing was performed via the Shapiro-Wilk test. Data conforming to a normal distribution were expressed as mean ± standard deviation (x±s); intra-group comparisons used the t-test, and inter-group comparisons used one-way analysis of variance followed by Tukey’s post-hoc test. Data with non-normal distribution were expressed as median (M) and interquartile range (IQR) comparisons used the Kruskal-Wallis H test. The chi-square test was used to compare categorical data between groups. Multiplicity adjustment was performed using the Bonferroni method to reduce type-I error risk. Statistical significance was defined as p < 0.05.
Results
Baseline Characteristics
A CONSORT flow diagram (Figure 1A) elucidates the processes of participant screening, randomization, and analysis. A total of 80 Wagner grade 2–4 diabetic foot ulcers (DFUs) were randomly assigned to four study groups (n = 20 per group). Table 1 presents the baseline characteristics—age, sex, diabetes duration, ulcer duration, infection grade, and Wagner classification. No significant differences were detected among groups (p > 0.05), confirming their comparability. No severe adverse events were recorded. Specifically, the control group experienced three cases of mild infection spread (15 %), the red/blue group reported two cases of mild burns (10 %), the SH group had one case of mild allergic reaction (5 %), and the SH + red/blue group also recorded one mild allergic reaction (5 %). All events resolved with local treatment.
Characteristics of the patients (n = 20 each group)
| Characteristic | Control | Red/blue | SH | SH+red/blue | Mean difference (95 % CI) | |
|---|---|---|---|---|---|---|
| Male | 12 | 11 | 11 | 10 | -0.50 (-2.38~1.38) | 0.921 |
| Age (y) | 62.54 ± 10.88 | 60.38 ± 10.66 | 61.27 ± 10.11 | 60.93 ± 11.23 | -1.61 (-7.52~4.30) | 0.857 |
| Diabetes duration (y) | 15.87± 8.34 | 16.22 ± 8.19 | 15.89 ± 8.37 | 16.41 ± 7.65 | 0.54 (-3.82~4.90) | 0.986 |
| Mixed ulcers | 6 | 7 | 7 | 6 | 0.25 (-1.03~1.53) | 0.945 |
| Venous ulcer | 6 | 7 | 8 | 7 | 0.50 (-0.81~1.81) | 0.782 |
| Ischemic ulcer | 8 | 6 | 5 | 7 | -0.25 (-1.53~1.03) | 0.945 |
| Wagner II | 10 | 9 | 8 | 11 | -1.00 (-3.02~1.02) | 0.678 |
| Wagner III | 8 | 9 | 9 | 7 | 0.00 (-2.01~2.01) | 0.999 |
| Wagner IV | 2 | 2 | 3 | 2 | 0.25 (-1.03~1.53) | 0.945 |
SH liquid fumigation combined with pulsed red/blue light improves clinical efficacy
Notably, the SH + red/blue group achieved a significantly higher clinical success rate than the other groups (p = 0.002 vs control; p = 0.018 vs red/blue; p = 0.043 vs SH), as shown in Figures 1B and 1C, underscoring the potential efficacy of this treatment modality.
SH liquid fumigation with alternating red/blue light accelerates wound healing
Prior to treatment, no significant differences were observed in ulcer scores among the four groups. After the intervention, scores decreased progressively in all groups, with the SH + red/blue group showing the lowest values at week 4 (Figure 2A). This group also demonstrated a significantly higher area-reduction rate (Figure 2B). For pain, the SH + red/blue group differed significantly from control (p = 0.019) but not from the red/blue (p = 0.352) or SH group (p = 0.881; Figure 2C). In addition, the SH + red/blue group exhibited a shorter healing time (Figure 2D) and, at week 2, the lowest bacterial load expressed as CFU·g of tissue (Figure 2E; Table 2).
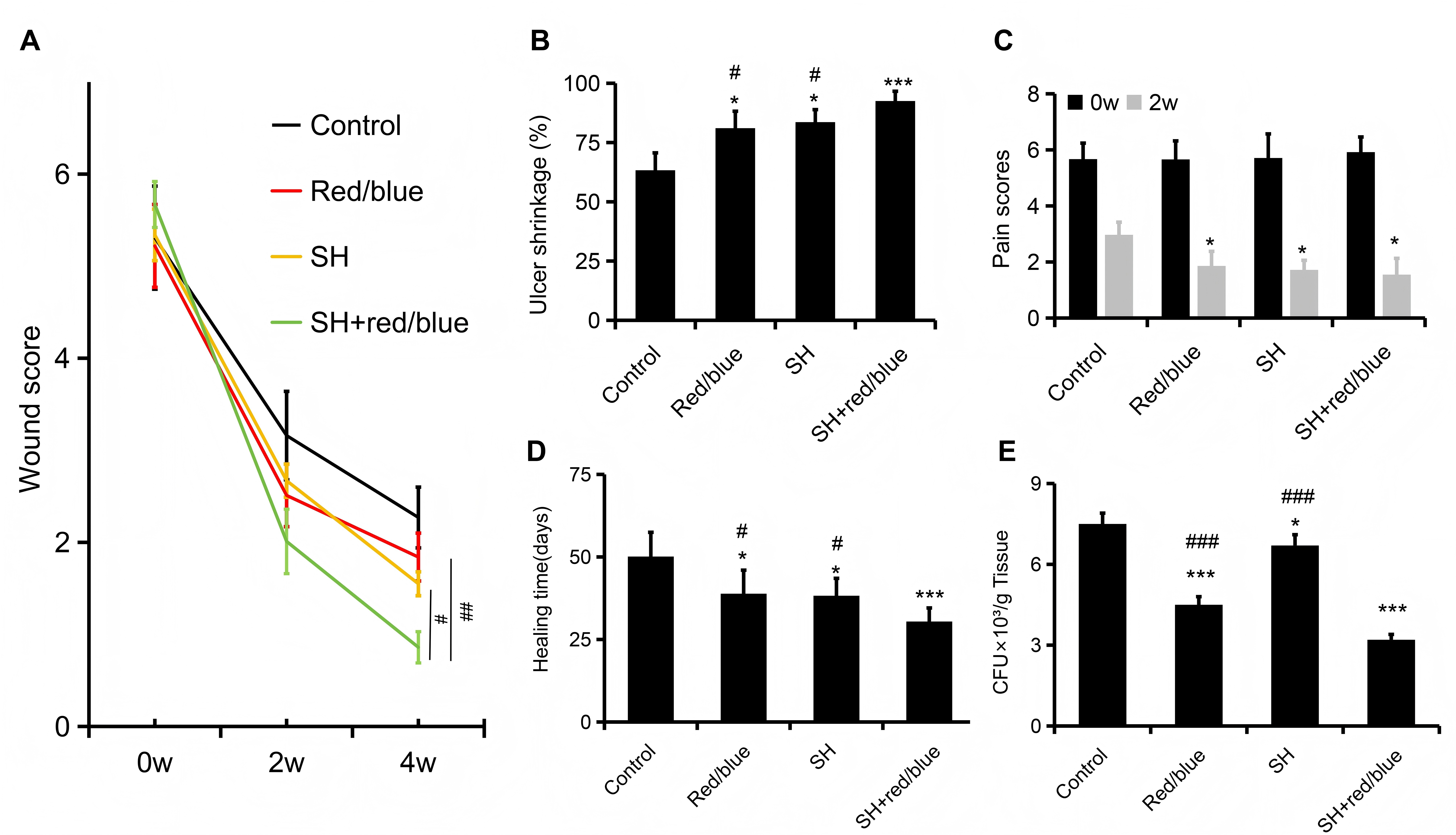
The ulcer wound scores, Shrinkage rate, pain scores and healing time pre- and post-treatment in all groups (n=20). A, Wound scores; B, Shrinkage rate; C, Pain scores; D, Healing time. *
SH + red/ blue light accelerates wound healing (n = 20)
| Groups | Control | Red/blue | SH | SH+red/blue | |
|---|---|---|---|---|---|
| Wound scores(0 w) | 5.3±0.6 | 5.2±0.5 | 5.3±0.3 | 5.7±0.3 | |
| p value | vs. control | 0.912 | 0.999 | 0.587 | |
| 0.493 | 0.587 | vs. SH+red/blue | |||
| Wound scores(2w) | 3.2±0.4 | 2.5±0.3 | 2.7±0.2 | 2.0±0.4 | |
| p value | vs. control | 0.028 | 0.045 | <0.001 | |
| 0.042 | 0.021 | vs. SH+red/blue | |||
| Wound scores(4w) | 2.3±0.3 | 1.8±0.3 | 1.6±0.1 | 0.9±0.2 | |
| p value | vs. control | 0.035 | 0.018 | <0.001 | |
| 0.008 | 0.015 | vs. SH+red/blue | |||
| Shrinkage (%) | 63.3±7.3 | 81.1±7.1 | 83.6±5.3 | 92.5±4.1 | |
| p value | vs. control | 0.022 | 0.015 | <0.001 | |
| 0.038 | 0.047 | vs. SH+red/blue | |||
| Pain scores | 3.0±0.5 | 1.9±0.5 | 1.7±0.3 | 1.6±0.6 | |
| p value | vs. control | 0.031 | 0.025 | 0.019 | |
| 0.352 | 0.881 | vs. SH+red/blue | |||
| Healing time (d) | 50.1±11.9 | 38.9±11.4 | 38.3±10.4 | 30.4±8.9 | |
| p value | vs. control | 0.027 | 0.023 | <0.001 | |
| 0.032 | 0.039 | vs. SH+red/blue | |||
| CFU×103/g Tissue (2w) | 7.5±0.4 | 4.5±0.3 | 6.7±0.4 | 3.2±0.2 | |
| p value | vs. control | <0.001 | 0.041 | <0.001 | |
| <0.001 | <0.001 | vs. SH+red/blue | |||
SH liquid fumigation combined with alternating red/blue light stimulates VEGF and suppresses IL-1 release
Before intervention, VEGF and IL-1 levels did not differ significantly between groups. Time-course analysis revealed that VEGF concentrations increased in the SH + red/blue group at week 2 (p < 0.001 vs control; p < 0.001 vs red/blue; p < 0.001 vs SH; Figure 3A), whereas IL-1 concentrations decreased (p < 0.001 vs control; p < 0.001 vs red/blue; p = 0.002 vs SH; Figure 3B). These changes paralleled the reduction in ulcer area, suggesting a mechanistic link between VEGF up-regulation, IL-1 down-regulation, and wound healing (Table 3).
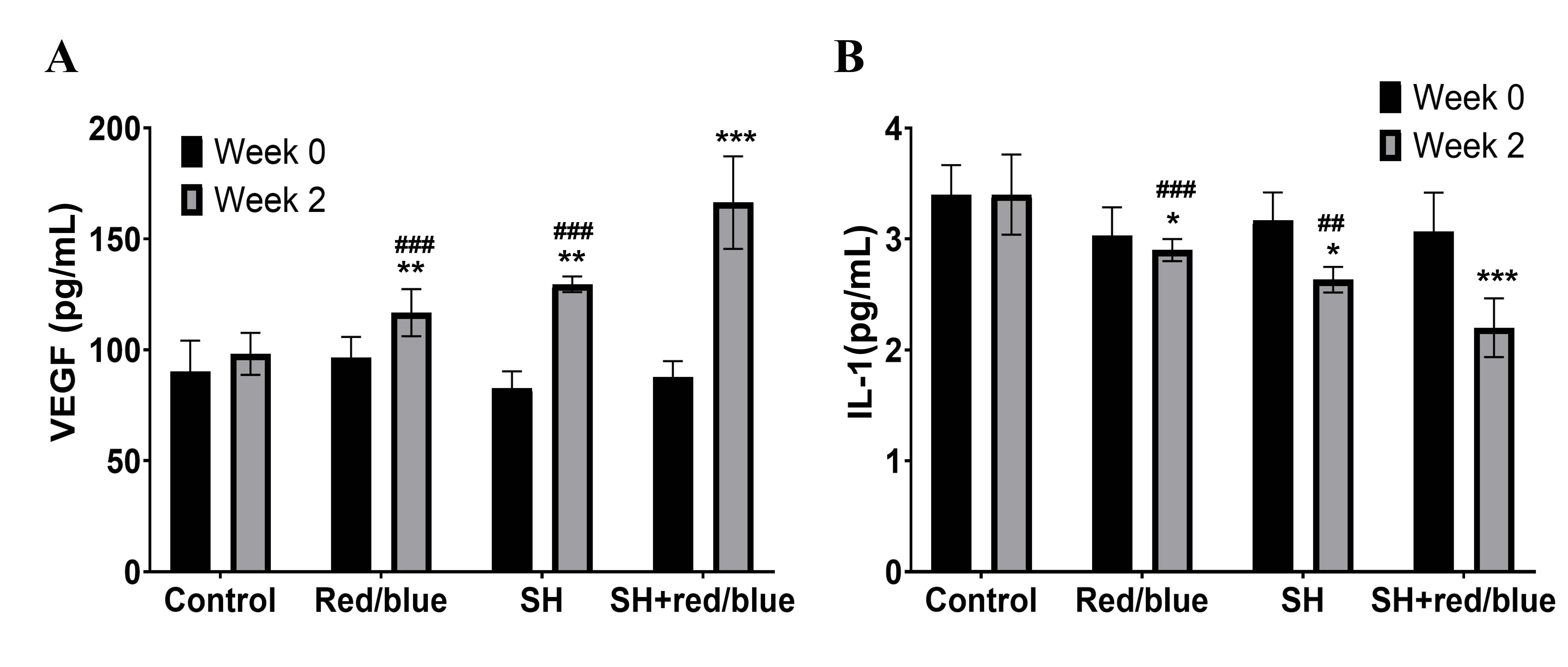
VEGF and IL-1 levels pre- and post-treatment in all groups (n=20). A, VEGF; B, IL-1; *
SH + red/ blue light stimulates VEGF and suppresses IL-1 release (n = 20)
| Groups (pg/mL) | Control | Red/blue | SH | SH+red/blue | |
|---|---|---|---|---|---|
| VEGF (0w) | 89.6±28.7 | 95.0±25.4 | 81.2±20.2 | 86.4±16.3 | |
| p value | vs. control | 0.725 | 0.489 | 0.801 | |
| 0.476 | 0.733 | vs. SH+red/blue | |||
| VEGF (2w) | 108.7±24.8 | 126.4±22.6 | 130.4±10.5 | 165.7±21.3 | |
| p value | vs. control | 0.008 | 0.005 | <0.001 | |
| <0.001 | <0.001 | vs. SH+red/blue | |||
| IL-1 (0w) | 3.6±0.7 | 3.0±0.2 | 3.2±0.9 | 3.4±0.1 | |
| p value | vs. control | 0.412 | 0.587 | 0.793 | |
| 0.576 | 0.805 | vs. SH+red/blue | |||
| IL-1 (2w) | 3.7±0.3 | 3.1±0.6 | 2.8±0.3 | 2.1±0.2 | |
| p value | vs. control | 0.042 | 0.018 | <0.001 | |
| <0.001 | 0.002 | vs. SH+red/blue | |||
Discussion
Diabetic foot, a severe complication of diabetes, is associated with high mortality, disability, and recurrence rates, and it imposes substantial financial burdens on patients and healthcare systems16,17. Epidemiological data indicate that up to 25 % of patients with diabetic foot ulcers (DFUs) will require amputation18, and the case-fatality rate is approximately 11 %19. The etiological and pathological mechanisms underlying DFUs are multifactorial. Current therapeutic strategies include debridement, infection control (predominantly with antibiotics), vascular reconstruction, wound dressings, oxygen therapy, and biological agents; however, antibiotic therapy may have under-appreciated adverse effects. Recent evidence demonstrates that different antibiotics differentially alter gut microbiota composition, some disrupting beneficial taxa, exacerbating systemic inflammation, and impairing metabolic regulation in people with diabetes20. This situation creates a paradox: while antibiotics control local infection, they may simultaneously compromise systemic conditions essential for DFU healing. In contrast, the integrative regimen evaluated in the present study—Traditional Chinese Medicine (TCM) SH liquid fumigation combined with alternating red/blue light—targets local infection while minimizing antibiotic exposure, thereby reducing the risk of gut-microbiota dysbiosis, a problem that remains insufficiently addressed in contemporary DFU management21.
Emerging evidence suggests that photobiomodulation enhances wound repair by modulating cellular metabolism and inflammatory pathways22. Mechanistic studies show that red light (620–760 nm) stimulates cell proliferation and expression of genes involved in tissue repair, augments VEGF secretion, reduces pro-inflammatory mediators, dilates deep-tissue blood vessels, accelerates inflammation resolution, and promotes both wound closure and analgesia23,24. Blue light (435–480 nm), another component of the visible spectrum, possesses bactericidal activity25,26. According to Wang et al., its antibacterial mechanism relies on the excitation of endogenous microbial photosensitizers, leading to the production of highly cytotoxic reactive oxygen species (ROS) and consequent oxidative damage to bacterial DNA, proteins, and lipids27.
In the present trial, therapeutic efficacy improved in all four groups after two weeks, ranking SH + red/blue > SH > red/blue > control. The superior effect of the combined therapy is presumably attributable to synergy: SH liquid fumigation improves local perfusion and mitigates inflammation, whereas alternating red/blue light promotes angiogenesis via VEGF and eliminates bacteria, jointly accelerating wound healing. Moreover, blinded outcome assessment minimized detection bias, and adjustment for multiplicity limited type I error, thereby strengthening the validity of the findings. These results provide preliminary evidence supporting the combined regimen for DFU management.
A key limitation of this study is the small sample size (20 participants per group) despite an a priori calculation. Larger, multicentre trials are required to confirm these observations. A further limitation is the short follow-up period; long-term endpoints such as ulcer recurrence and amputation rates warrant evaluation in future research.
Conclusion
In conclusion, the combined application of SH liquid and alternating red/blue light significantly accelerates the healing of Wagner grade 2–4 diabetic foot ulcers (DFUs), alleviates ulcer-related pain, up-regulates VEGF expression, attenuates inflammatory responses, and represents a promising therapeutic strategy for this patient population.
Abbreviations
CFU: Colony-forming units; CONSORT: Consolidated Standards of Reporting Trials; CV: Coefficient of variation; DFUs: Diabetic foot ulcers; ELISA: Enzyme-linked immunosorbent assay; H/R: Hypoxia/reoxygenation; ICH-GCP: International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use—Good Clinical Practice; IL-1: Interleukin-1; IQR: Interquartile range; RLT: Red-light therapy; ROS: Reactive oxygen species; SH: Shenghong; TCM: Traditional Chinese Medicine; VAS: Visual Analogue Scale; VEGF: Vascular endothelial growth factor; WHO: World Health Organization
Acknowledgments
None
Author’s contributions
CD, LL and YZ conducted the experiments and analysis and wrote initial draft. YC contributed to discussions. XX reviewed and edited the paper. All authors read and approved the final manuscript.
Funding
None.
Availability of data and materials
Data utilized in this study are available upon reasonable request are available from the corresponding author upon reasonable request.
Ethics approval and consent to participate
This project was reviewed and approved by the Medical Ethics Committee of Guangdong Integrated Traditional Chinese and Western Medicine Hospital (No.2022-046). All participants provided written informed consent.
Consent for publication
Not applicable.
Declaration of generative AI and AI-assisted technologies in the writing process
The authors declare that they have not used generative AI (a type of artificial intelligence technology that can produce various types of content including text, imagery, audio and synthetic data. Examples include ChatGPT, NovelAI, Jasper AI, Rytr AI, DALL-E, etc) and AI-assisted technologies in the writing process before submission.
Competing interests
The authors declare that they have no competing interests.
Trial registration: Chinese Clinical Trial Registry, ITMCTR2025001993.

