Personalised medicine in treatment of acute recurrent urticaria: Step-by-step algorithm for diagnosis and therapy
- Department of Allergology, National Medical Institute of the Ministry of the Interior and Administration, 02-507, 137 Woloska Str., Warsaw, Poland
Abstract
Introduction: The study investigates personalised strategies for the diagnosis and treatment of acute recurrent urticaria (ARU), analyses current treatment methods aimed at individualising therapy, and develops a scientifically-grounded treatment algorithm tailored to the needs of patients with heterogeneous symptoms.
Methods: Using a systematic literature review focused on clinical trials, meta-analyses, and molecular diagnostics, the study examined publications on ARU from PubMed, Scopus, and Web of Science. Findings were integrated through critical analysis, synthesis, comparative evaluation, and risk-of-bias assessment to construct a patient-centred therapy algorithm based on patient characteristics and therapeutic responses.
Results: Biologic therapy with omalizumab demonstrated effectiveness in treating severe forms of ARU, providing sustained remission in approximately 85 % of patients with refractory disease. Second-generation non-sedating antihistamines were effective in 70–80 % of patients with mild-to-moderate disease, with 20–30 % of cases requiring an increased dose to achieve clinical improvement. The resultant therapy algorithm stratifies patients according to disease severity, age, and comorbidities, aiming to enhance treatment effectiveness and reduce the risk of side effects.
Conclusion: The results underscore the importance of a personalised approach to the treatment of ARU that considers individual characteristics such as patient age, comorbidities, and previous treatment experience. The use of modern diagnostic methods and targeted therapy facilitates long-term remission and improves patient quality of life. The proposed algorithm is flexible and can be adapted for use in various clinical settings.
Introduction
Acute recurrent urticaria (ARU) remains a diagnostic and therapeutic challenge. The condition is defined by recurrent pruritic wheals that substantially impair patients’ quality of life and often necessitate prolonged pharmacotherapy. First-line treatment with H1-antihistamines is frequently inadequate, thereby emphasising the need for novel, individualised management strategies.
Current innovative approaches incorporate monoclonal antibodies, such as omalizumab, which have shown pronounced efficacy in refractory ARU. In addition, the role of genetic profiling continues to expand, enabling the identification of individual pharmacogenomic susceptibilities. The integration of artificial intelligence into predictive-response modelling further advances precision medicine. Globally conducted investigations are clarifying ARU pathomechanisms and revealing regional variations in therapeutic practice. Jamjanya et al.1 performed a systematic review and meta-analysis involving Asian cohorts and confirmed the high effectiveness of second-generation antihistamines. The authors underscore that timely diagnosis and prompt pharmacological intervention reduce the risk of complications and improve patient-reported outcomes.
Badloe et al.2 systematically analysed contemporary European approaches to the management of acute urticaria, with particular emphasis on prolonged-release antihistamines. Their data indicate that these agents reduce adverse-event rates and enhance therapeutic efficacy. The authors also underscore the necessity of an individualised treatment strategy, especially in patients with comorbidities. Salman et al.3 advocated a differentiated diagnostic and therapeutic pathway for acute versus chronic urticaria based on Spanish epidemiological data, demonstrating that precise phenotyping facilitates more appropriate therapeutic selection. They further observed that novel biologics are promising for chronic urticaria refractory to conventional regimens. Mazur et al.4 quantified the prevalence and identified potential risk factors for urticaria among Polish children and adolescents, thereby highlighting the value of epidemiological surveillance for risk stratification and treatment planning.
Cherniuk et al.5 examined autoimmune contributions and comorbidity in chronic urticaria, noting that approximately 30 % of cases are associated with conditions such as autoimmune thyroiditis. Patients with such comorbidities displayed a higher likelihood of refractoriness to standard therapy, necessitating a multimodal approach. Yılmaz et al.6 evaluated second-generation antihistamines, reporting clinical improvement in 85 % of patients with acute urticaria; however, corticosteroids were frequently required for chronic disease. They warned that delayed intervention in acute urticaria can result in chronicity in 20–25 % of cases, thereby complicating subsequent management.
Fok et al.7 identified determinants of treatment success in chronic spontaneous urticaria, noting a 40 % relapse rate among patients with concurrent autoimmune disorders. In these individuals, use of biologics—particularly omalizumab—was recommended to achieve durable remission. Consistent with this, Obtułowicz et al.8 documented that omalizumab not only decreases the frequency and severity of flares but also markedly improves quality of life by attenuating pruritus and wheals. He et al.9 reviewed emerging insights into the immunopathogenesis and treatment of chronic urticaria, emphasising roles for T-cell activation and pro-inflammatory cytokine production. They concluded that combined administration of antihistamines and immunomodulators benefits up to 75 % of patients with severe disease.
Tyczyńska and Krajewski10 examined the role of primary care in the management of urticaria. The authors analysed the key diagnostic steps, which comprise the identification of trigger factors, systematic symptom assessment, and exclusion of comorbidities. They also provided step-by-step recommendations for selecting second-generation antihistamines, dose titration, and therapy modification when initial treatment is ineffective. Wedi11 reviewed the potential of innovative treatments for chronic urticaria. In particular, the author examined the effectiveness of monoclonal antibodies, such as omalizumab and lebrikizumab, in treatment-resistant disease. He reported that omalizumab achieved clinical improvement in >70 % of patients. Antonicelli et al.12 evaluated mepolizumab in patients with severe eosinophilic asthma concomitant with chronic urticaria. The study demonstrated that combination therapy targeting eosinophilic inflammation significantly improves patient quality of life and reduces the frequency of urticaria recurrences and asthma symptoms. Kolkhir et al.13 presented a detailed overview of current pathogenetic concepts in urticaria, focusing on key mechanisms. The study highlights the importance of autoimmune and allergic factors in disease pathogenesis. The authors reported that targeted therapy controls symptoms of chronic urticaria in 60–80 % of patients, particularly in those resistant to standard antihistamines.
Despite advances in therapeutic development, the mechanisms underlying relapse and treatment resistance remain incompletely understood. We postulated that a personalised approach based on comprehensive clinical, immunological, and molecular-genetic profiling would enhance the efficacy of acute recurrent urticaria (ARU) therapy. The study aimed to develop a stepwise algorithm for the diagnosis and treatment of ARU, incorporating contemporary precision-medicine strategies.
Materials and METHODS
To achieve the study's pubives, rigorous scientific methods were employed, each contributing substantially to the resulting findings. A structured analysis of scientific publications and reports from international organisations14,15 was conducted to elucidate the pathophysiological mechanisms of ARU, characterise trigger factors (allergens, autoimmune and pseudo-allergic reactions)5,13,16, and assess the efficacy of therapeutic approaches, including antihistamines, biologics9,11 and molecular diagnostic techniques17.
A systematic literature review was performed in PubMed, Scopus and Web of Science. Boolean operators combined the terms “acute recurrent urticaria,” “personalised medicine,” “biological therapy,” “histamine release” and “basophil activation test.” The search was restricted to English-language publications from 2010 to 2025 that reported clinical trials, systematic reviews or meta-analyses. Studies pertinent to the diagnosis and treatment of ARU—particularly those addressing molecular diagnostics and personalised therapy—were included.
The selected databases provided comprehensive coverage of both molecular and clinical research. Owing to stringent inclusion criteria, only peer-reviewed articles—namely systematic reviews, cohort studies, and clinical trials published in high-impact journals—were considered. Priority was accorded to papers investigating novel biomarkers, diagnostic modalities, and therapeutic interventions such as antihistamines and biologics (e.g., omalizumab). Studies that did not focus on ARU or that failed to involve human subjects were excluded. Non-peer-reviewed or grey literature (e.g., dissertations and conference abstracts), along with articles lacking data on clinical outcomes or treatment efficacy, were likewise omitted.
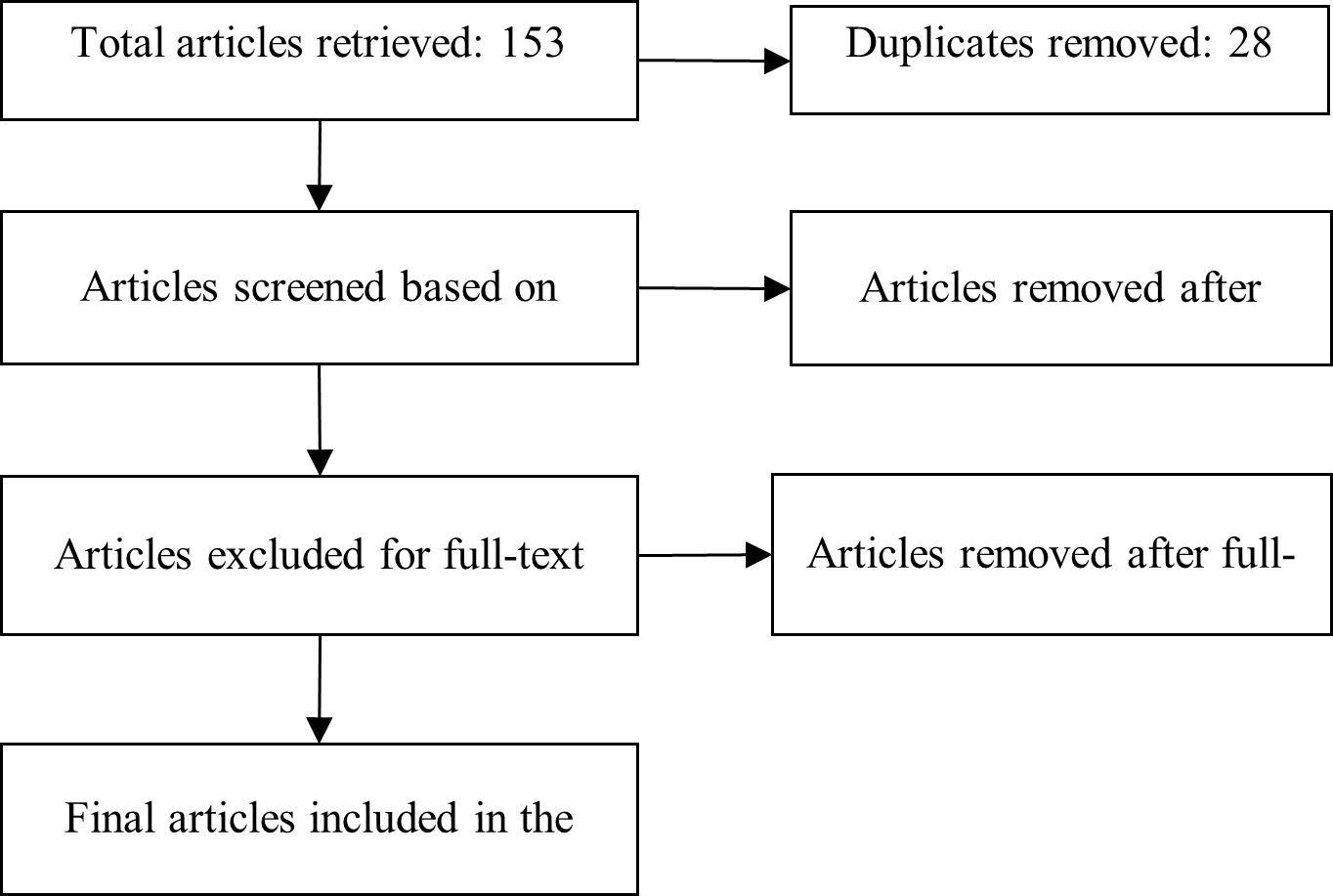
A PRISMA flow diagram (Figure 1) was generated to depict the study-selection process, including database identification, title/abstract screening, full-text assessment, data extraction, and qualitative synthesis.
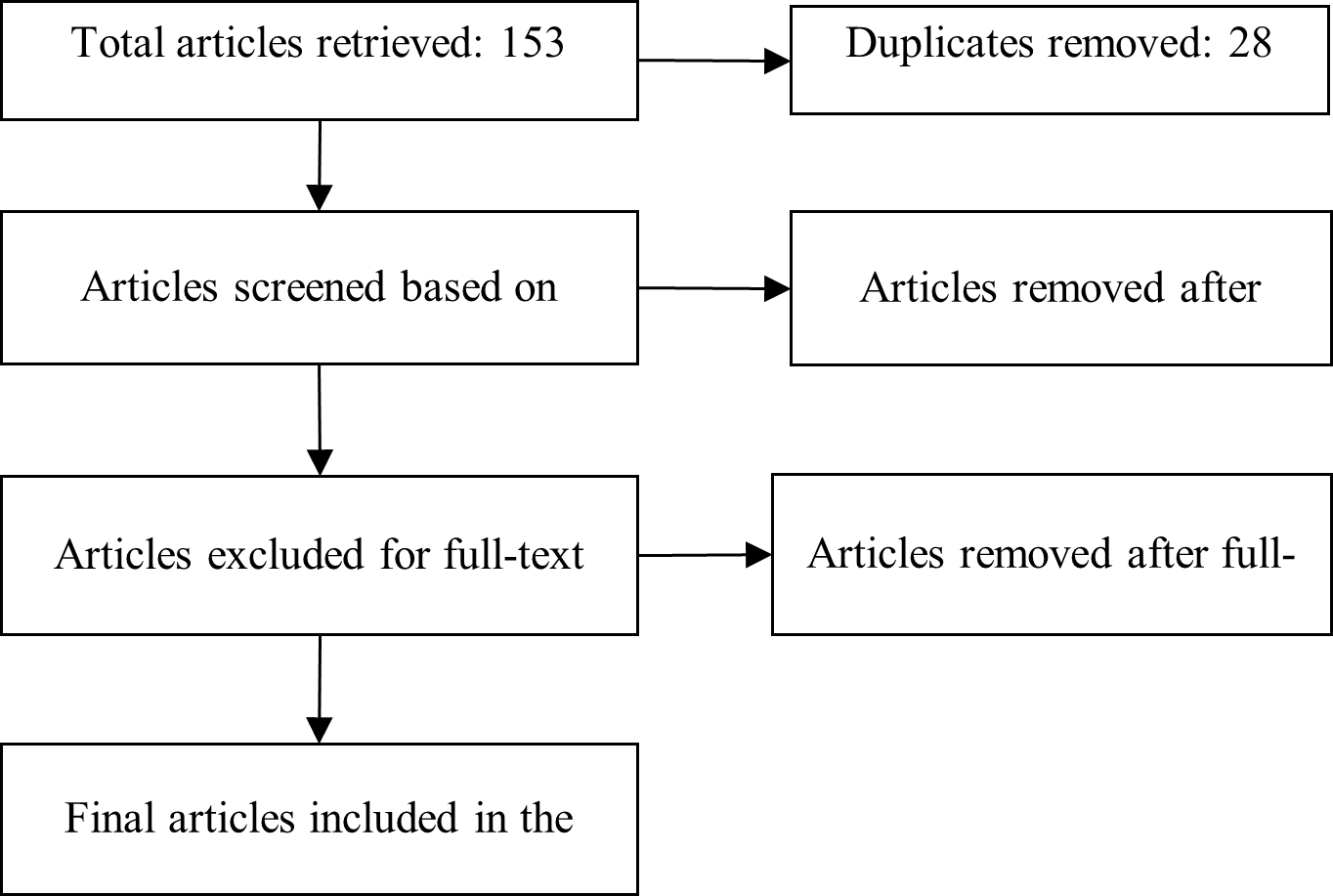
PRISMA flowchart of study selection process.
During data extraction, key variables were recorded: study design, patient demographics (age, comorbidities), therapeutic regimens (omalizumab, antihistamines), diagnostic techniques (basophil activation test, cytokine profiling), and outcomes (quality-of-life metrics, clinical efficacy). The synthesis strategy integrated heterogeneous evidence into a coherent framework, combining case reports, biologic-drug trials, and international guideline recommendations to construct an adaptive ARU-management model. This approach linked pathophysiological mechanisms to therapeutic strategies, thereby delineating the principal steps of an evidence-based diagnostic and treatment algorithm.
Study quality was appraised with the Cochrane Risk of Bias tool for randomized controlled trials and the Newcastle–Ottawa Scale for observational studies. Risk of bias was judged across selection, performance, detection, and reporting domains; studies at high risk were excluded to preserve the robustness of the conclusions.
Comparative analysis was crucial for evaluating diagnostic and therapeutic efficacy. Recommendations from the European Academy of Allergy and Clinical Immunology (EAACI)14 and the American Academy of Allergy, Asthma and Immunology (AAAAI)15 were contrasted. Inductive reasoning synthesized individual clinical observations, whereas deductive reasoning generated logical inferences supporting the use of personalized medicine to optimize outcomes. Systematization techniques structured the aggregated evidence. Collectively, these methodologies conferred high scientific validity and underpinned the development of a personalized-medicine algorithm for ARU management.
Findings from the Literature
Peculiarities of Diagnosis of ARU in the Context of Personalised Medicine
Modern approaches to the diagnosis of ARU require a comprehensive approach that involves the integration of molecular, immunological , and genetic methods. This combination of techniques can not only precisely elucidate disease pathogenesis but also facilitate the individualisation of therapeutic strategies. A systems-based diagnostic framework incorporates validated biomarkers—such as serum histamine, allergen components, and specific immunoglobulin E (IgE)—thereby increasing diagnostic precision. In addition, molecular assays enable a more granular analysis of the underlying pathways, a prerequisite for personalised management.
One of the key methods is the basophil activation test (BAT), which quantifies basophil reactivity, pivotal cells in ARU pathogenesis. This read-out correlates clinical manifestations with immunological parameters and assists in grading disease severity. In addition, the serum tryptase level is routinely measured to exclude systemic mastocytosis, which is an important step in differentiating similar pathologies. For this purpose, the application of predefined tryptase cut-off values serves as a guide in the diagnostic process, especially in the absence of another obvious cause of clinical symptoms. Asero et al.16 noted that the use of BAT optimises treatment selection when standard methods do not yield the desired result. These findings support the feasibility of integrating BAT into standard clinical practice.
Molecular approaches to diagnosis include quantification of specific IgE to autoantigens, such as IgE to FcεRI, which suggests a possible autoimmune nature of ARU. This not only clarifies the diagnosis but also determines the most effective therapeutic methods, focusing treatment on reducing immune system activation through appropriate therapeutic agents5. Zhang et al.17 investigated the role of biomarkers, including specific IgE, in diagnosing the autoimmune nature of ARU. The authors emphasised that the determination of IgE to FcεRI not only helps to establish an accurate diagnosis but also allows for the optimisation of treatment strategies, focusing on blocking immune mechanisms. This approach is consistent with the data obtained in this section and confirms the importance of molecular methods in personalised medicine.
A promising approach to individualized therapy for allergic diseases, including chronic spontaneous urticaria (CSU), is biomarker-guided treatment. Recent research indicates that biomarkers can optimize therapeutic decision-making by matching drugs to each patient’s immunopathological profile, thereby enhancing efficacy and minimizing adverse events. Both Ogulur et al.18 and Aulin et al.19 have underscored the pivotal role of biomarkers in tailoring treatment strategies. The anti-IgE monoclonal antibody ligelizumab has shown considerable potential in this context. A systematic review and meta-analysis by Putini Vieira et al.20 confirmed its efficacy and safety in CSU, demonstrating superior outcomes compared with alternative therapies. Moreover, phase 3 trials conducted by Maurer et al.21 corroborated the ability of ligelizumab to provide sustained symptom control in adults and adolescents with severe chronic urticaria. Collectively, these findings support the growing importance of biomarker-driven selection of ligelizumab to achieve optimal therapeutic results.
Serum concentrations of pro-inflammatory cytokines, such as IL-6 and IL-8, have been investigated as surrogate markers of disease activity. Integration of cytokine data with other molecular parameters yields a more precise depiction of the inflammatory cascade, which is indispensable for individualized treatment planning. Schaefer22 stated that longitudinal cytokine monitoring is critical for elucidating pathogenetic mechanisms. He further emphasized that such measurements can guide evaluation of therapeutic response, particularly during combination regimens, findings that are congruent with the present results.
The diagnostic work-up of ARU still relies on skin-prick tests with histamine and relevant allergens, procedures that delineate the underlying mechanism and distinguish patients with pseudo-allergic reactions. In addition, multiplex molecular allergy panels that identify specific allergen components provide a refined assessment of sensitization risk to animal dander or other environmental triggers, which is critical for individualized management.13 Current guidelines by Zuberbier et al.14 highlight the necessity of a comprehensive diagnostic algorithm incorporating molecular allergy panels and serum tryptase to differentiate ARU from systemic mastocytosis. Implementation of these tools into routine practice may reduce diagnostic errors and improve therapeutic efficacy, observations that are corroborated by the data presented here.
A personalized diagnostic approach to ARU also includes integration of genomic data. Analysis of single-nucleotide polymorphisms (SNPs) can predict individual responsiveness to antihistamines, thereby avoiding ineffective or suboptimal regimens. Gimenez-Arnau et al.23 demonstrated the predictive value of SNP analysis for antihistamine efficacy, identifying patients at high risk for treatment resistance and allowing development of individualized protocols. Similarly, Grubska-Suchanek and Nowicki24 emphasized the contribution of genetic factors and comorbidities to disease heterogeneity. They reported that molecular genetic techniques enable more accurate urticaria subtyping and therapy selection, findings that align with the present study, in which personalized medicine is anchored in biomarker utilization and patient-specific characteristics.
In summary, integration of molecular, immunological, and genetic methodologies facilitates a comprehensive characterization of ARU, forming the cornerstone of personalized therapy. This approach not only enhances therapeutic effectiveness but also reduces complications and affords superior symptom control. Consequently, addressing all facets of the disorder through individualized treatment selection is paramount for improving patients’ quality of life and achieving sustained remission.
The Effectiveness of Therapeutic Strategies Recommended by International Organisations
International organizations such as EAACI14 and AAAAI15 provide recommendations derived from large-scale clinical trials that aim to improve ARU treatment efficacy. These guidelines address regional characteristics, socioeconomic conditions, and individual patient needs.
The mainstay of ARU treatment is antihistamines, which suppress the activity of histamine—the principal disease mediator. Contemporary research demonstrates that individual factors (e.g., serum IgE concentration, underlying autoimmune mechanisms, and inflammatory severity) markedly influence therapeutic outcomes. An individualized approach therefore incorporates biomarkers that predict disease severity and therapeutic response; however, their high cost and limited availability in low-income countries remain major barriers. Differentiating ARU from other urticaria subtypes (e.g., autoimmune or trigger-induced) further underscores the need for molecular diagnostics. Measuring D-dimer concentrations or basophil activity enables more precise therapy selection, fully aligning with the principles of personalized medicine.
EAACI14 is a leading authority in developing ARU management standards. As noted by Zuberbier et al.14, EAACI stratifies patients by disease severity, comorbidities, and immunologic profile. Its primary recommendation is second-generation, non-sedating antihistamines, which are effective in 70–80 % of ARU patients at standard doses.14 If the response is insufficient, EAACI advises four-fold dose escalation—a strategy validated in clinical studies. The guideline also advocates biomarker assessment, particularly D-dimer, to gauge inflammatory intensity. According to Lang et al.25, elevated D-dimer correlates with severe ARU and justifies the addition of biologics or other immunotherapies. EAACI additionally recommends evaluating autoreactive IgE (e.g., against FcεRI), facilitating the identification of autoimmune urticaria and enabling more targeted treatment.
AAAAI15 regards ARU as a multifactorial disorder whose management should be tailored to patients’ individual characteristics. It emphasizes biological agents—most notably omalizumab—for severe or antihistamine-refractory disease. Lang et al.25 confirm that omalizumab blocks the IgE-dependent pathway, reducing basophil activation and histamine release. Treated patients exhibit significantly lower relapse rates and shorter symptom duration, achieving durable remission. Consistent with personalized care, AAAAI recommends serial monitoring of total IgE and pro-inflammatory cytokines (e.g., IL-6, IL-8) to guide dosage adjustments. The guideline also stresses developing strategies to enhance biologic affordability, as high cost limits widespread use.
Basic antihistamine therapy controls symptoms in 60–70 % of patients with mild-to-moderate ARU, according to Folci et al.26. For severe disease, EAACI advocates adjunctive therapies, including short-course systemic corticosteroids or biologics, and highlights preventive measures—such as avoiding food allergens, stressors, and medications—to reduce exacerbation frequency and improve quality of life.
Comparative analysis of EAACI and AAAAI guidance (Table 1) reveals differences driven by resource availability and patient demographics. Both endorse innovative modalities (biomarkers, biologics) that require substantial funding and specialized training. Integrating these approaches, in light of contemporary evidence, could yield universal therapeutic frameworks that balance individualized care with healthcare-system capabilities.
Comparative analysis of EAACI and AAAAI recommendations for the treatment of ARU
| Organisation | The key focus of the recommendations | Features for implementation |
|---|---|---|
| EAACI | Use of biomarkers to identify disease subtypes and predict response to therapy. Using patient stratification to personalise treatment. Recommendations for the use of modern immunomodulatory drugs and targeted therapy. | The need for laboratories to analyse biomarkers. Training doctors for modern therapeutic approaches. |
| AAAAI | Emphasise the importance of biological products (e.g., monoclonal antibodies) for the treatment of severe hypersensitivity. Recommendations for early detection of patients with severe disease for timely use of biological therapy. Monitoring patients to assess the effectiveness of therapy. | The high cost of biological products requires resources. Specialised training of healthcare professionals is required to prescribe and monitor such therapy. |
Identification of Key Factors for Successful Therapy of ARU
Acute relapsing urticaria (ARU) is a multifactorial disorder characterised by the abrupt appearance of erythematous wheals accompanied by marked pruritus. The condition may present as a primary disease or arise secondary to the interplay of numerous exogenous and endogenous triggers, each of which modulates the clinical phenotype and disease course. Because of the pronounced heterogeneity of clinical manifestations, therapeutic efficacy is influenced by several variables, particularly patient-specific characteristics.
Patient age is a key determinant of therapeutic strategy in ARU. Disease-inducing mechanisms and treatment responses differ between paediatric and geriatric populations. In children, infectious agents are the predominant precipitating factors, whereas in adults, food and drug allergens prevail. Cetinkaya et al.27 reported that progression from acute to chronic urticaria occurs more frequently in older children, especially in those with comorbid asthma or eczema, underscoring the need for age-tailored management. In contrast, older adults often exhibit diminished responses to standard antihistamines. Hon et al.28 showed that individuals ≥65 years are more susceptible to sedative adverse events associated with first-generation antihistamines, limiting their use. Consequently, contemporary second-generation, non-sedating antihistamines represent first-line therapy in this cohort, consistent with EAACI recommendations.
Comorbid conditions further refine therapeutic decision-making. Autoimmune and endocrine disorders—particularly thyroid disease—exacerbate disease complexity and frequently necessitate modification of baseline treatment. Kaplan29 demonstrated that chronic systemic illnesses heighten the risk of ARU, especially in immunologically compromised patients who exhibit increased reactivity to environmental allergens. In such settings, combination regimens comprising antihistamines and immunomodulatory agents may be required. Likewise, concomitant food allergy can compromise treatment outcomes; Zuberbier et al.30 found that affected patients respond poorly to conventional antihistamines, warranting dietary trigger avoidance and, when necessary, the addition of biologics.
Prior therapeutic exposure also shapes current treatment success. Patients with recurrent ARU who have previously received antihistamines may develop drug hypersensitivity or tachyphylaxis. Nowicki et al.31 observed reduced efficacy of standard dosing in this subgroup, necessitating dose escalation or transition to biologics such as omalizumab. Accordingly, tailoring therapy to pharmacological history is essential, particularly in cases of resistance to conventional regimens.
The relative efficacy of these treatment modalities varies according to age, comorbidity profile and prior pharmacotherapy (Table 2).
Comparison of the effectiveness of antihistamines and omalizumab depending on factors
| Factor | The effectiveness of antihistamines | The effectiveness of omalizumab | Note |
|---|---|---|---|
| Young age (up to 40 years) | High | Average | Predominantly positive response to standard therapy |
| Elderly age (60+ years) | Low | High | Risk of side effects due to comorbidity |
| Autoimmune diseases | Low | High | Requires combination therapy |
| Previous treatment experience | Low in antihistamine resistance | High | Effective in severe forms of ARU |
Table 2 demonstrates that omalizumab is more effective in older patients and those with autoimmune diseases, whereas younger patients usually respond positively to standard antihistamine therapy. Furthermore, when antihistamine-resistant disease is present, omalizumab is effective in patients with severe presentations. These data may help clinicians to individualise therapy for patients with different characteristics. Genetic factors are crucial in the pathogenesis of ARU and influence the effectiveness of therapeutic strategies. The analysis of single-nucleotide polymorphisms (SNPs) allows clinicians to predict the response of patients to antihistamines and biologics, including omalizumab. A study by Giménez-Arnau et al.23 noted that the presence of certain genetic variations may be associated with higher resistance to standard therapy, which requires personalisation of treatment approaches. In addition, the identification of mutations in genes that regulate mast-cell function allows for a more accurate determination of disease severity. The use of genetic testing in clinical practice helps to optimise therapeutic strategies and increase their effectiveness in patients with ARU.
The socioeconomic status of patients significantly affects the availability of diagnosis and treatment of ARU. According to Sánchez-Borges et al.32 and Błaszczyk et al.33, limited access to modern diagnostic methods and expensive medicines in low-income countries significantly complicates the implementation of effective therapeutic strategies. The level of health literacy of patients is also an important factor in determining their adherence to treatment. Patients with a low level of knowledge about the nature of the disease may have difficulty following recommendations, which affects therapeutic outcomes. Integration of social aspects into clinical practice allows treatment approaches to be adapted to the socio-economic conditions of different regions, thereby increasing the accessibility and effectiveness of therapy. Environmental factors can affect the frequency and severity of ARU, including exposure to air pollution, environmental allergens and climate change. The study by Segú-Vergés et al.34 showed that high levels of air pollution correlate with an increase in the frequency of ARU exacerbations due to mast-cell activation. These factors can also increase sensitivity to allergens, which complicates both diagnosis and therapy. Integration of environmental aspects into therapeutic algorithms, through the development of preventive measures aimed at minimising the impact of the environment on the course of the disease, may help to improve treatment outcomes. The success of ARU therapy depends largely on individual patient characteristics, such as age, comorbidities and previous treatment experience. Genetic, social and environmental factors can be used to develop more effective and personalised therapeutic strategies, thereby enhancing treatment efficacy. An individualised approach based on a thorough analysis of medical and laboratory data is the key to improving therapeutic outcomes in the management of ARU.
Formation of a Step-By-Step Algorithm for the Diagnosis and Treatment of ARU
The algorithm for the diagnosis and treatment of ARU is grounded in the integration of contemporary scientific evidence and personalised therapeutic approaches. Its primary pubives are diagnostic accuracy, therapeutic efficacy, and adaptation of recommendations to heterogeneous health-care settings. The algorithm relies on molecular diagnostics, immunological analyses, and individual patient characteristics.
The first step comprises a detailed medical history, clinical examination, and identification of potential disease triggers. Trigger identification helps determine the disease subtype (allergic, pseudo-allergic, or autoimmune) and guides the selection of the optimal therapeutic strategy. This step is underpinned by patient phenotyping with stratification into acute and chronic forms of urticaria. Akin et al.35 highlighted the importance of phenotype classification for assessing the risk of progression from acute to chronic urticaria, noting that early risk recognition markedly reduces complication rates.
The second step involves quantification of serum tryptase, histamine, and allergen-specific IgE to differentiate allergic from non-allergic disease. This approach interrogates the immunological mechanisms of mast-cell activation. Segú-Vergés et al.34 present current data supporting a systems-biology approach to urticaria pathogenesis and stress the value of multiplex molecular allergy panels for trigger identification, consistent with the proposed algorithm.
The third step comprises the basophil activation test (BAT) and measurement of pro-inflammatory cytokines (IL-6, IL-8). BAT assesses basophil activity, an important surrogate of disease severity. Frigas and Park36 demonstrated that BAT is diagnostically valuable for predicting therapeutic response, and that cytokine profiling clarifies the inflammatory pathways underpinning urticaria.
The fourth step addresses treatment, commencing with second-generation, non-sedating antihistamines; in refractory cases, biologics such as omalizumab are introduced11. This stage embodies the principles of precision medicine by tailoring therapy to individual responses. Sabroe37 confirmed the efficacy of antihistamines as first-line agents, whereas Sánchez-Borges et al.32 emphasised the need for personalised regimens in severe disease, findings aligned with the current algorithm.
The fifth step entails continuous monitoring and, when necessary, adjustment of the therapeutic plan. Biomarker and cytokine levels are re-evaluated to gauge treatment efficacy. Pite et al.38 recommend incorporating genetic testing to predict long-term therapeutic success.
The algorithm is structured to accommodate health-care systems with variable resources. In low-resource settings, emphasis is placed on standard pharmacotherapy, whereas high-resource settings can incorporate molecular diagnostics and biologics.
This stepwise framework therefore encompasses the critical phases of ARU diagnosis and management, from initial evaluation and biomarker assessment to longitudinal monitoring of clinical outcomes (Figure 2).
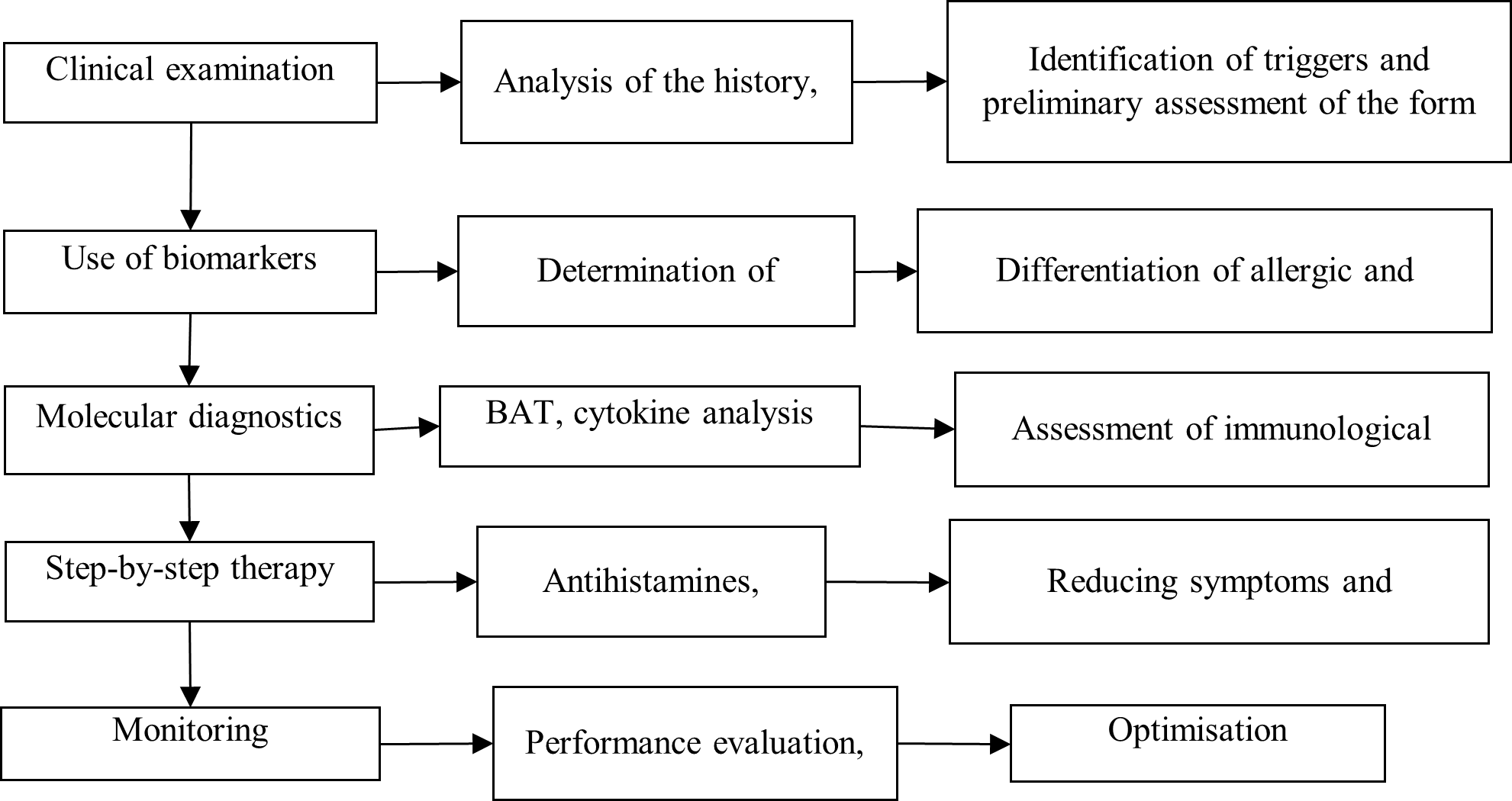
Stages of the algorithm for diagnosis and therapy of ARU.
The suggested algorithm adopts a more individualized approach to ARU management, emphasizing the integration of molecular diagnostics, including cytokine profiling and basophil activation testing, thereby augmenting the current EAACI and AAAAI guidelines. It tailors therapy to individual patient characteristics, such as genetic factors, comorbidities, and previous treatment responses, whereas the current guidelines primarily focus on generalized therapeutic strategies. By providing a more precise and agile therapeutic pathway, the algorithm aims to optimize clinical outcomes and reduce adverse-event risk, facilitating a comprehensive framework for managing ARU.
The application of advanced diagnostic modalities, such as BAT, pro-inflammatory cytokine profiling, and genetic testing, ensures high diagnostic precision in delineating the pathogenetic mechanisms of the disease. The tabulated data demonstrate that the proposed algorithm is founded on the integration of evidence-based methodologies and contemporary research findings, offering a systematised approach to ARU diagnosis and treatment. The incorporation of molecular and genetic techniques markedly enhances causal determination and therapy-response prognostication.
Moreover, stepwise treatment escalation is underscored, allowing adaptation of the therapeutic strategy to the patient’s clinical presentation and the medical institution’s resource availability. This feature underscores the algorithm’s versatility, rendering it effective in both highly resourced environments and resource-constrained settings.
The Impact of a Personalised Approach on the Quality of Life of Patients
Quality of life is a key indicator of therapeutic effectiveness, particularly in chronic, relapsing disorders such as ARU. It encompasses patients’ physical, emotional, and social well-being, which together determine their capacity to perform daily activities, maintain interpersonal relationships, and mitigate psychological stress. Personalised medicine tailors therapeutic strategies to each patient’s specific needs, taking into account disease characteristics, comorbidities, and social or environmental factors. This paradigm not only achieves superior clinical outcomes but also enhances overall patient satisfaction with therapy.
According to Papakonstantinou39, the integration of molecular diagnostics with regular therapeutic monitoring enables more precise elucidation of the pathophysiological mechanisms driving the disease. Consequently, treatment can be adjusted to minimise symptom duration and relapse frequency. The author also reported that a personalised approach reduces the likelihood of adverse effects, thereby exerting a favourable influence on patients’ quality of life.
Individuals with ARU frequently experience sleep disruption, reduced productivity, and emotional exhaustion owing to persistent pruritus and associated symptoms. **Comert et al.**40 observed that standard treatment regimens often fail to provide adequate disease control in patients with recurrent urticaria. In such circumstances, individualised therapy that targets disease-specific features and identified triggers can markedly decrease exacerbation frequency. The study further emphasised the value of combination therapy in patients with concomitant allergic or autoimmune disorders.
Personalised management of ARU exerts a profound impact on health status and quality of life. Patient-centred interventions can improve physical condition, emotional well-being, and social functioning, thereby promoting holistic clinical benefit (Table 3). The present findings corroborate the importance of individualised care in enhancing quality of life among patients with ARU. Tailoring therapy to personal requirements facilitates sustained clinical improvement, augments treatment adherence, and constitutes a pivotal determinant of long-term remission, underscoring the broad applicability of this approach.
The impact of a personalised approach on various aspects of the condition of patients with ARU
| Impact aspects of a personalised approach | Results |
|---|---|
| Physical condition | Reduced frequency of symptoms (itching, rashes), improved sleep |
| Emotional well-being | Reduced anxiety, improved mood, increased satisfaction with therapy |
| Social activity | Restoration of working capacity, reduction of social isolation |
The integration of advanced diagnostic modalities, systematic outcome monitoring, and patient-specific therapeutic adjustment contributes to improvements in both somatic and psycho-emotional domains. These results highlight the need for broader implementation of personalised strategies in routine clinical practice to secure durable remission and optimise patients’ lives.
Conclusions
The integration of molecular, immunological, and genetic diagnostic modalities employed in this study to personalise the management of acute recurrent urticaria (ARU) is noteworthy. Its uniqueness lies in establishing a systematic treatment plan that combines individualised therapies with state-of-the-art diagnostic techniques to optimise therapeutic efficacy and minimise adverse events. As a result, both clinical outcomes and patients’ quality of life are improved. The findings confirm that combining molecular, immunological, and genetic methods elucidates the pathogenetic mechanisms of the disease and thereby enhances diagnostic accuracy. In particular, the basophil activation test (BAT) and measurement of pro-inflammatory cytokines such as IL-6 and IL-8 facilitate assessment of disease severity and guide therapeutic decision-making.
Therapeutic efficacy was shown to depend on disease severity and individual patient characteristics. Second-generation, non-sedating antihistamines ameliorated symptoms in patients with mild to moderate ARU, whereas biologic agents such as omalizumab demonstrated high efficacy in antihistamine-refractory cases, yielding clinical improvement and long-term remission in severe, including autoimmune, forms. Younger patients exhibited a superior response to standard therapy, while older patients, owing to comorbidities, required combination strategies encompassing pharmacological and non-pharmacological interventions.
The proposed diagnostic-therapeutic algorithm provides a stepwise approach to ARU management, with therapy differentiated according to diagnostic findings. Biomarkers—including D-dimer and total IgE—enable tailoring of treatment to individual needs, thereby increasing efficacy and reducing adverse-event risk, which ultimately enhances patient quality of life.
These data underscore the effectiveness of a personalised approach to ARU diagnosis and treatment. Nonetheless, study limitations—such as limited long-term follow-up and under-representation of specific cohorts (e.g., young children and elderly patients with significant comorbidities)—highlight the need for further research. Restricted access to primary clinical data also precluded experimental validation of the results.
Future investigations should evaluate the long-term efficacy of the recommended strategies, examine environmental and genetic modifiers of ARU, and develop cost-effective diagnostic tools for resource-limited settings. Overall, the evidence supports personalisation as paramount for maximising therapeutic success, minimising complications, and improving prognosis in ARU.
Abbreviations
AAAAI: American Academy of Allergy, Asthma and Immunology; ARU: Acute Recurrent Urticaria; BAT: Basophil Activation Test; CSU: Chronic Spontaneous Urticaria; EAACI: European Academy of Allergy and Clinical Immunology; H1: Histamine 1; IgE: Immunoglobulin E; IL-6: Interleukin-6; IL-8: Interleukin-8; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; SNPs: Single-Nucleotide Polymorphisms.
Acknowledgments
None.
Author’s Contributions
The author was solely responsible for the conception, design, data collection, analysis, interpretation, and writing of the manuscript.
Funding
None.
Consent for Publication
Not applicable.
Declaration of Generative AI and AI-Assisted Technolofies in the Writing Process
The author affirms that no generative AI or AI-assisted technologies were used in the conceptualization, research, analysis, or writing of this manuscript. All content was created solely by the author without the assistance of automated tools.
Competing Interests
The author declares that there is no conflict of interests.

